Abstract
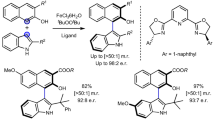
The ready availability, low price and environmentally benign character of iron mean that it is an ideal alternative to precious metals in catalysis. Recent growth in the number of iron-catalysed reactions reported reflects an increasing demand for sustainable chemistry. Only a limited number of chiral iron catalysts have been reported and these have, in general, proven less enantioselective than other transition-metal catalysts, thus limiting their appeal. Here, we report that iron complexes of spiro-bisoxazoline ligands are highly efficient catalysts for asymmetric O–H bond insertion reactions. These complexes catalyse insertions into the O–H bond of a wide variety of alcohols and even water, with exceptional enantioselectivities under mild reaction conditions. The selectivities surpass those obtained with other transition-metal catalysts. This study should inspire and encourage the use of iron instead of traditional precious metals in the development of greener catalysts for catalytic asymmetric synthesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Jacobson, E. N., Pfaltz, A. & Yamamoto, H. (eds) Comprehensive Asymmetric Catalysis and supplement 1 and 2 (Springer, 1999, 2004).
Yoon, T. P. & Jacobsen, E. N. Privileged chiral catalysts. Science 299, 1691–1693 (2003).
Bolm, C. A new iron age. Nature Chem. 1, 420 (2009).
Plietker, B. (ed.) Iron Catalysis in Organic Chemistry (Wiley-VCH, 2008).
Bolm, C., Legros, J., Le Paih, J. & Zani, L. Iron-catalyzed reaction in organic synthesis. Chem. Rev. 104, 6217–6254 (2004).
Enthaler, S., Junge, K. & Beller, M. Sustainable metal catalysis with iron: from rust to a rising star? Angew. Chem. Int. Ed. 47, 3317–3321 (2008).
Sherry, B. D. & Fürstner, A. The promise and challenge of iron-catalyzed cross coupling. Acc. Chem. Res. 41, 1500–1511 (2008).
Corey, E. J., Imai, N. & Zhang, H.-Y. Designed catalyst for enantioselective Diels–Alder addition from a C2-symmetric chiral bis(oxazoline)–Fe(III) complex. J. Am. Chem. Soc. 113, 728–729 (1991).
Suzuki, K., Oldenburg, P. D. & Que, L. Jr. Iron-catalyzed asymmetric olefin cis-dihydoxylation with 97% enantiomeric excess. Angew. Chem. Int. Ed. 47, 1887–1889 (2008).
Shaikh, N. S., Enthaler, S., Junge, K. & Beller, M. Iron-catalyzed enantioselective hydrosilylation of ketones. Angew. Chem. Int. Ed. 47, 2497–2501 (2008).
Langlotz, B. K., Wadepohl, H. & Gade, L. H. Chiral bis(pyridylimino)isoindoles: a highly modular class of pincer ligands for enantioselective catalysis. Angew. Chem. Int. Ed. 47, 4670–4674 (2008).
Mikhailine, A., Lough, A. J. & Morris, R. H. Efficient asymmetric transfer hydrogenation of ketones catalyzed by an iron complex containing a P–N–N–P tetradentate ligand formed by template synthesis. J. Am. Chem. Soc. 131, 1394–1395 (2009).
Egami, H. & Katsuki, T. Iron-catalyzed asymmetric aerobic oxidation: oxidative coupling of 2-naphthols. J. Am. Chem. Soc. 131, 6082–6083 (2009).
Crabtree, R. H. The Organometallic Chemistry of the Transition Metals 4th edn, vol. 11 (Wiley-Interscience, 2005).
Doyle, M. P., McKervey, M. A. & Ye, T. Modern Catalytic Methods for Organic Synthesis with Diazo Compounds (Wiley, 1998).
Davies, H. M. L. & Manning, J. R. Catalytic C–H functionalization by metal carbenoid and nitrenoid insertion. Nature 451, 417–424 (2008).
Ge, M. & Corey, E. J. A method for the catalytic enantioselective synthesis of 6-silylated 2-cyclohexenones. Tetrahedron Lett. 47, 2319–2321 (2006).
Maier, T. C. & Fu, G. C. Catalytic enantioselective O–H insertion reaction. J. Am. Chem. Soc. 128, 4594–4595 (2006).
Liu, B., Zhu, S.-F., Zhang, W., Chen, C. & Zhou, Q.-L. Highly enantioselective insertion of carbenoids into N–H bonds catalyzed by copper complexes of chiral bisoxazolines. J. Am. Chem. Soc. 129, 5834–5835 (2007).
Lee, E. C. & Fu, G. C. Copper-catalyzed asymmeric N–H insertion reactions: coupling of diazo compounds with carbamates to generate α-amino acids. J. Am. Chem. Soc. 129, 12066–12067 (2007).
Chen, C., Zhu, S.-F., Liu, B., Wang, L.-X. & Zhou, Q.-L. Highly enantioselective insertion of carbenoids into O–H bonds of phenols: an efficient approach to chiral α-aryloxycarboxylic esters. J. Am. Chem. Soc. 129, 12616–12617 (2007).
Zhu, S.-F., Chen, C., Cai, Y. & Zhou, Q.-L. Catalytic asymmetric reaction with water: enantioselective synthesis of α-hydroxyesters by a copper-carbenoid O–H insertion reaction. Angew. Chem. Int. Ed. 47, 932–934 (2008).
Jolly, P. W. & Pettit, R. Evidence for a novel metal–carbene system. J. Am. Chem. Soc. 88, 5044–5045 (1966).
Redlich, M. & Hossain, M. M. Synthesis of asymmetric iron–pybox complexes and their application to aziridine forming reaction. Tetrahedron Lett. 45, 8987–8990 (2004).
Liu, B., Zhu, S.-F., Wang, L.-X. & Zhou, Q.-L. Preparation and application of bisoxazoline ligands with a chiral spirobiindane skeleton for asymmetric cyclopropanation and allylic oxidation. Tetrahedron Asymm. 17, 634–641 (2006).
Schmidt, B. & Wildemann, H. Diastereoselective ring-closing metathesis in the synthesis of dihydropyrans. J. Org. Chem. 65, 5817–5822 (2000).
Schmidt, B. Ruthenium-catalyzed olefin metathesis double-bond isomerization sequence. J. Org. Chem. 69, 7672–7687 (2004).
De Benassuti, L., Garanti, L. & Molteni, G. Stereoselective intramolecular cycloaddition of homochiral nitrilimines: synthesis of enantiopure (6S)-substituted-2,3,3a,4,5,6-hexahydrofuro[3,4-c]pyrazoles. Tetrahedron Asymm. 15, 1127–1131 (2004).
Cierpucha, M., Solecka, J., Frelek, J., Szczukiewicz, P. & Chmielewski, M. Synthesis, biological and chiroptical active of 3-phenyl-clavams. Bioorg. Med. Chem. 12, 405–416 (2004).
Schmidt, B. & Nave, S. Palladium-catalyzed O-allylation of α-hydroxy carbonyl compounds. Adv. Synth. Catal. 348, 531–537 (2006) and references therein.
Grimley, J. Pharma challenged: market shifts and generic competition create dynamic environment for drug developers. Chem. Eng. News 84, 17 (2006).
Castaldi, G., Barreca, G. & Bologna, A. A process for the preparation of clopidogrel. WO 03093276 (2003).
Acknowledgements
The authors wish to thank the National Natural Science Foundation of China, the National Basic Research Program of China (973 Program, nos 2006CB806106, 2010CB833300), the Ministry of Health (grant no. 2009ZX09501-017) and the ‘111’ Project of the Ministry of Education of China (grant no. B06005) for financial support.
Author information
Authors and Affiliations
Contributions
S.-F.Z., J.-H.X. and Q.-L.Z. designed the project. C.Y. and H.-X.M. carried out the experimental work. S.-F.Z. and Q.-L.Z. wrote the paper. All authors analysed the data, discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 3861 kb)
Rights and permissions
About this article
Cite this article
Zhu, SF., Cai, Y., Mao, HX. et al. Enantioselective iron-catalysed O–H bond insertions. Nature Chem 2, 546–551 (2010). https://doi.org/10.1038/nchem.651
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.651
This article is cited by
-
Chemoselective carbene insertion into the N−H bonds of NH3·H2O
Nature Communications (2022)
-
Mixture of clopidogrel bisulfate and magnesium oxide tablets reduces clopidogrel dose administered through a feeding tube
Journal of Pharmaceutical Health Care and Sciences (2021)
-
Asymmetric O-propargylation of secondary aliphatic alcohols
Nature Catalysis (2020)
-
Ligands with 1,10-phenanthroline scaffold for highly regioselective iron-catalyzed alkene hydrosilylation
Nature Communications (2018)
-
Recent progress in insertion and cyclopropanation reactions of metal carbenoids from α-diazocarbonyl compounds
Research on Chemical Intermediates (2017)