Abstract
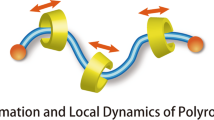
Molecular mobility has attracted considerable attention in supramolecular chemistry and biochemistry, but the simple question of whether a small molecule can move directly between different binding sites of a multitopic host without intermediate dissociation has not been addressed so far. To study such processes, we consider hydrogen/deuterium exchange experiments on a model system comprising complexes formed between 18-crown-6 and oligolysine peptides. Because direct binding-site hopping is indistinguishable in solution from a dissociation/reassociation mechanism, here we show that the high vacuum of a mass spectrometer offers a unique environment for probing such processes. The highly dynamic motion of crown ethers along oligolysine peptide chains proceeds mechanistically by a simultaneous transfer of the crown ether from its ammonium ion binding site to a nearby amino group together with a proton. Furthermore, the exchange experiments unambiguously reveal the zwitterionic structure of the 18-crown-6/oligolysine complexes, highlighting the versatility and potential of gas-phase experiments for investigating non-covalent interactions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
23 October 2009
At roughly the same time, the authors of this Article independently submitted a manuscript to Angewandte Chemie, in which the method presented here was applied to polyamino propylene amine dendrimers. The authors forgot to mention this publication and would like to apologize for their mistake. The missing reference is: Winkler, H. D. F., Weimann, D. P., Springer, A. and Schalley, C. A. Dynamic motion in crown ether dendrimer complexes: A "spacewalk" on the molecular scale. Angew. Chem. Int. Ed. 48, 7246-7250 (2009).
References
Meot-Ner (Mautner), M. The ionic hydrogen bond. Chem. Rev. 105, 213–284 (2005).
Lifshitz, C. A review of gas-phase H/D exchange experiments: the protonated arginine dimer and bradykinin nonapeptide systems. Int. J. Mass Spectrom. 234, 63–70 (2004).
Kellersberger, K. A., Desjupa, C., Liang, Y. J., Pope, R. M. & Dearden, D. V. Gas phase studies of ammonium-cyclodextrin compounds using Fourier transform ion cyclotron resonance. Int. J. Mass Spectrom. 193, 181–195 (1999).
Ventola, E., Rissanen, K. & Vainiotalo, P. Mass spectrometric investigation of noncovalent complexation between a tetratosylated resorcarene and alkyl ammonium ions. Chem. Eur. J. 10, 6152–6162 (2004).
Ventola, E., Hyyryläinen, A. & Vainiotalo, P. Complex formation between a tetramesityl sulfonated resorcarene and alkylammonium ions: a mass spectrometric study of noncovalent interactions. Rapid Commun. Mass Spectrom. 20, 1218–1224 (2006).
Kalenius, E., Moiani, D., Dalcanale, E. & Vainiotalo, P. Measuring H-bonding supramolecular complexes by gas phase ion-molecule reactions. Chem. Commun. 3865–3867 (2007).
Wood, T. D. et al. Gas-phase folding and unfolding of cytochrome c cations. Prod. Natl Acad. Sci. USA 92, 2421–2454 (1995).
Wyttenbach, T. & Bowers, M. T. Gas phase conformations of biological molecules: the hydrogen/deuterium exchange mechanism. J. Am. Soc. Mass Spectrom. 10, 9–14 (1999).
Lee, S.-W., Lee, H.-N., Kim, H. S. & Beauchamp, J. L. Selective binding of crown ethers to protonated peptides can be used to probe mechanisms of H/D exchange and collision-induced dissociation reactions in the gas phase. J. Am. Chem. Soc. 120, 5800–5805 (1998).
Lias, S. G. Thermoneutral isotope exchange-reactions in proton-bound complexes of water with organic-molecules — correlations with energetics of formation of the corresponding association ions. J. Phys. Chem. 88, 4401–4407 (1984).
Cram, D. J. & Cram, J. M. Design of complexes between synthetic hosts and organic guests. Acc. Chem. Res. 11, 8–14 (1978).
Rüdiger, V. et al. Crown ether-ammonium complexes: binding mechanisms and solvent effects. Eur. J. Org. Chem. 1847–1856 (1999).
Maleknia, S. & Brodbelt, J. Cavity-size-dependent dissociation of crown ether/ammonium ion complexes in the gas phase. J. Am. Chem. Soc. 115, 2837–2843 (1993).
Dearden, D. V., Liang, Y. J., Nicoll, J. B. & Kellersberger, K. A. Study of gas-phase molecular recognition using Fourier transform ion cyclotron resonance mass spectrometry (FTICR/MS). J. Mass Spectrom. 36, 989–997 (2001).
Dearden, D. V., Desjupa, C., Liang, Y. J., Bradshaw, J. S. & Izatt, R. M. Intrinsic contributions to chiral recognition: Discrimination between enantiomeric amines by dimethyldiketopyridino-18-crown-6 in the gas phase. J. Am. Chem. Soc. 119, 353–359 (1997).
Wu, H. F. & Brodbelt, J. S. Comparison of the orders of gas-phase basicities and ammonium ion affinities of polyethers by the kinetic method and ligand-exchange technique. J. Am. Soc. Mass Spectrom. 4, 718–722 (1993).
Liou, C.-C. & Brodbelt, J. S. Comparison of gas-phase proton and ammonium ion affinities of crown ethers and related acyclic analogs. J. Am. Chem. Soc. 114, 6761–6764 (1992).
Dearden, D. V. & Chu, I.-H. Relative ammonium ion affinities of 18-crown-6 and the isomers of dicyclohexano-18-crown-6. J. Incl. Phenomena Mol. Recogn. 29, 269–282 (1997).
Julian, R. R. & Beauchamp, J. L. The unusually high proton affinity of aza-18-crown-6 ether: Implications for the molecular recognition of lysine in peptides by lariat crown ethers. J. Am. Soc. Mass. Spectrom. 13, 493–498 (2002).
Julian, R. R., May, J. A., Stoltz, B. M. & Beauchamp, J. L. Biomimetic approaches to gas phase peptide chemistry: combining selective binding motifs with reactive carbene precursors to form molecular mousetraps. Int. J. Mass Spectrom. 228, 851–864 (2003).
Fenn, J. B., Mann, M., Meng, C. K., Wong, S. F. & Whitehouse, C. M. Electrospray ionization for mass spectrometry of large biomolecules. Science 246, 64–71 (1989).
Marshall, A. G., Hendrickson, C. S. & Jackson, G. S. Fourier transform ion cyclotron resonance mass spectrometry: a primer. Mass Spectrom. Rev. 17, 1–35 (1998).
Hofstadler, S. A., Sannes-Lowery, K. A. & Griffey, R. H. A gated-beam electrospray ionization source with an external ion reservoir. A new tool for the characterization of biomolecules using electrospray ionization mass spectrometry Rapid Commun. Mass. Spectrom. 13, 1971–1979 (1999).
Hofstadler, S. A., Sannes-Lowery, K. A. & Griffey, R. H. Enhanced gas-phase hydrogen-deuterium exchange of oligonucleotide and protein ions stored in an external multipole ion reservoir. J. Mass. Spectrom. 35, 62–70 (2000).
Campbell, S., Rodgers, M. T., Marzluff, E. M. & Beauchamp, J. L. Deuterium exchange reactions as a probe of biomolecule structure. Fundamental studies of gas phase H/D exchange reactions of protonated glycine oligomers with D2O, CD3OD, CD3CO2D, and ND3 . J. Am. Chem. Soc. 117, 12840–12854 (1995).
Cox, H. A., Julian, R. R., Lee, S. W. & Beauchamp, J. L. Gas-Phase H/D exchange of sodiated glycine oligomers with ND3: exchange kinetics do not reflect parent ion structures. J. Am. Chem. Soc. 126, 6485–6490 (2004).
Freitas, M. A. & Marshall, A. G. Rate and extent of gas-phase hydrogen/deuterium exchange of bradykinins: evidence for peptide zwitterions in the gas phase. Int. J. Mass Spectrom. 182–183, 221–231 (1999).
Polfer, N. C., Dunbar, R. C. & Oomens, J. Observation of zwitterion formation in the gas-phase H/D-exchange with CH3OD: solution-phase structures in the gas phase. J. Am. Soc. Mass Spectrom. 18, 512–516 (2007).
Acknowledgements
We thank Andreas Springer for valuable scientific advice and acknowledge funding from the Deutsche Forschungsgemeinschaft and the Fonds der Chemischen Industrie.
Author information
Authors and Affiliations
Contributions
C.A.S., H.D.F.W. and D.P.W. conceived, designed and performed the mass spectrometric experiments and co-wrote the paper. J.A.F. and B.K. contributed the peptides. All authors discussed the results and commented on the manuscript.
Corresponding author
Supplementary information
Supplementary information
Supplementary information (PDF 1540 kb)
Rights and permissions
About this article
Cite this article
Weimann, D., Winkler, H., Falenski, J. et al. Highly dynamic motion of crown ethers along oligolysine peptide chains. Nature Chem 1, 573–577 (2009). https://doi.org/10.1038/nchem.352
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.352
This article is cited by
-
Protein camouflage in cytochrome c–calixarene complexes
Nature Chemistry (2012)
-
Recognition-mediated activation of therapeutic gold nanoparticles inside living cells
Nature Chemistry (2010)
-
Tracking molecular mobility
Nature Chemistry (2009)
-
Erratum: Highly dynamic motion of crown ethers along oligolysine peptide chains
Nature Chemistry (2009)