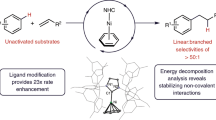
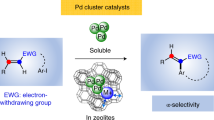
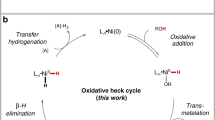
Abstract
Although the cleavage of C–C bonds in unactivated hydrocarbons by soluble transition-metal complexes remains a challenge, such reactions hold the potential to provide access to previously inconceivable skeletal transformations. For instance, one can imagine the dismantling and reassembly of C–C and C–H bonds commonly observed in surface catalysis, but with the increased control innate to homogeneous catalysis. Here we report a pentanuclear nickel cluster that is unreactive to functional groups, such as alcohols, amines and even water, but selectively cleaves the C=C bonds of simple alkenes, such as ethylene, styrene and isobutylene, at temperatures as low as −30 °C and in near-quantitative yields. The isolation of intermediates in reactions with styrene and isobutylene demonstrates that the five nickel centres cooperatively activate three C–H bonds of the alkene substrate before cleaving the C–C bond in the core of the cluster to give a pentanuclear nickel carbide. The net organic product transformation is the abstraction of a carbon atom from an alkene.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Takemori, T., Inagaki, A. & Suzuki, H. A novel type of carbon–carbon double bond cleavage of 1,1-disubstituted alkenes on a triruthenium polyhydrido cluster. J. Am. Chem. Soc. 123, 1762–1763 (2001).
Hu, S., Shima, T. & Hou, Z. Carbon–carbon bond cleavage and rearrangement of benzene by a trinuclear titanium hydride. Nature 512, 413–415 (2014).
Suzuki, H., Takaya, Y., Takemori, T. & Tanaka, M. Selective carbon–carbon bond cleavage of cyclopentadiene on a trinuclear ruthenium pentahydride complex. J. Am. Chem. Soc. 116, 10779–10780 (1994).
Matsubara, K., Inagaki, A., Tanaka, M. & Suzuki, H. Regioselective C–H bond activation of alkanes by a trinuclear ruthenium trihydride complex having a μ3-sulfido ligand. J. Am. Chem. Soc. 121, 7421–7422 (1999).
Buchowicz, W. et al. Triple C–H bond activation of a nickel-bound methyl group: synthesis and X-ray structure of a carbide cluster (NiCp)6(μ6-C). Inorg. Chem. 51, 8292–8297 (2012).
Chamberlin, R. L., Rosenfeld, D. C., Wolczanski, P. T. & Lobkovsky, E. B. Ethylene and alkyne carbon–carbon bond cleavage across tungsten–tungsten multiple bonds. Organometallics 21, 2724–2735 (2002).
Calle-Vallejo, F. et al. Finding optimal surface sites on heterogeneous catalysts by counting nearest neighbors. Science 350, 185–189 (2015).
Van Santen, R. A. Complementary structure sensitive and insensitive catalytic relationships. Acc. Chem. Res. 42, 57–66 (2008).
Siaj, M. & McBreen, P. H. Creating, varying, and growing single-site molecular contacts. Science 309, 588–590 (2005).
Vang, R. T. et al. Ethylene dissociation on flat and stepped Ni(111): a combined STM and DFT study. Surf. Sci. 600, 66–77 (2006).
Guo, X. et al. Direct, nonoxidative conversion of methane to ethylene, aromatics, and hydrogen. Science 344, 616–619 (2014).
Gozin, M., Weisman, A., Ben-David, Y. & Milstein, D. Activation of a carbon–carbon bond in solution by transition-metal insertion. Nature 364, 699–701 (1993).
Rybtchinski, B. & Milstein, D. Metal insertion into C−C bonds in solution. Angew. Chem. Int. Ed. 38, 870–883 (1999).
Eisch, J., Piotrowski, A., Han, K., Krüger, C. & Tsay, Y. Oxidative addition of nickel(0) complexes to carbon−carbon bonds in biphenylene: formation of nickelole and 1,2-dinickelecin intermediates. Organometallics 4, 224–231 (1985).
Gunay, A. & Jones, W. D. Cleavage of carbon–carbon bonds of diphenylacetylene and its derivatives via photolysis of Pt complexes: tuning the C–C bond formation energy toward selective C–C bond activation. J. Am. Chem. Soc. 129, 8729–8735 (2007).
Xi, Z., Sato, K., Gao, Y., Lu, J. & Takahashi, T. Unprecedented double C=C bond cleavage of a cyclopentadienyl ligand. J. Am. Chem. Soc. 125, 9568–9569 (2003).
Sattler, A. & Parkin, G. Cleaving carbon–carbon bonds by inserting tungsten into unstrained aromatic rings. Nature 463, 523–526 (2010).
Ohki, Y. et al. Co6H8(PiPr3)6: a cobalt octahedron with face-capping hydrides. Angew. Chem. 128, 16053–16057 (2016).
Shima, T. et al. Dinitrogen cleavage and hydrogenation by a trinuclear titanium polyhydride complex. Science 340, 1549–1552 (2013).
Camacho-Bunquin, J., Ferguson, M. J. & Stryker, J. M. Hydrocarbon-soluble nanocatalysts with no bulk phase: coplanar, two-coordinate arrays of the base metals. J. Am. Chem. Soc. 135, 5537–5540 (2013).
Goodfellow, R. J., Hamon, E. M., Howard, J. A., Spencer, J. L. & Turner, D. G. Cationic platinum hydride clusters: X-ray crystal structures of [Pt4H2(PtBu3)4][BF4]2[HBF4]2 and [Pt4H7(PtBu3)4][BPh4]. J. Chem. Soc. Chem. Commun. 1604–1606 (1984).
Brayshaw, S. K. et al. Holding onto lots of hydrogen: a 12-hydride rhodium cluster that reversibly adds two molecules of H2 . Angew. Chem. Int. Ed. 44, 6875–6878 (2005).
Brayshaw, S. K. et al. Sequential reduction of high hydride count octahedral rhodium clusters [Rh6(PR3)6H12][BArF4]2: redox-switchable hydrogen storage. J. Am. Chem. Soc. 129, 1793–1804 (2007).
Araake, R., Sakadani, K., Tada, M., Sakai, Y. & Ohki, Y. [Fe4] and [Fe6] hydride clusters supported by phosphines: synthesis, characterization, and application in N2 reduction. J. Am. Chem. Soc. 139, 5596–5606 (2017).
Tsui, E. Y., Tran, R., Yano, J. & Agapie, T. Redox-inactive metals modulate the reduction potential in heterometallic manganese–oxido clusters. Nat. Chem. 5, 293–299 (2013).
Kanady, J. S., Tsui, E. Y., Day, M. W. & Agapie, T. A synthetic model of the Mn3Ca subsite of the oxygen-evolving complex in photosystem II. Science 333, 733–736 (2011).
Fout, A. R., Zhao, Q., Xiao, D. J. & Betley, T. A. Oxidative atom-transfer to a trimanganese complex to form Mn6 (μ6-E)(E = O, N) clusters featuring interstitial oxide and nitride functionalities. J. Am. Chem. Soc. 133, 16750–16753 (2011).
Lee, Y. et al. Dinitrogen activation upon reduction of a triiron(II) complex. Angew. Chem. 127, 1519–1523 (2015).
Lee, Y. et al. Reactivity of hydride bridges in high-spin [3M–3(μ-H)] clusters (M = FeII, CoII). J. Am. Chem. Soc. 137, 10610–10617 (2015).
Murray, L. J., Weare, W. W., Shearer, J., Mitchell, A. D. & Abboud, K. A. Isolation of a (dinitrogen) tricopper (I) complex. J. Am. Chem. Soc. 136, 13502–13505 (2014).
Powers, T. M. & Betley, T. A. Testing the polynuclear hypothesis: multi-electron reduction of small molecules by triiron reaction sites. J. Am. Chem. Soc. 135, 12289 (2013).
Ishikawa, Y. et al. Modulation of benzene or naphthalene binding to palladium cluster sites by the backside-ligand effect. Angew. Chem. 127, 2512–2516 (2015).
Beck, R., Shoshani, M. & Johnson, S. A. Catalytic hydrogen/deuterium exchange of unactivated carbon–hydrogen bonds by a pentanuclear electron-deficient nickel hydride cluster. Angew. Chem. 124, 11923–11926 (2012).
Shoshani, M. M. & Johnson, S. A. Facile deep and ultradeep hydrodesulfurization by the [(iPr3P)Ni]5H6 cluster compared to mononuclear Ni sources. Inorg. Chem. 54, 11977–11985 (2015).
Ervin, K. M. et al. Bond strengths of ethylene and acetylene. J. Am. Chem. Soc. 112, 5750–5759 (1990).
Kalyani, D., Deprez, N. R., Desai, L. V. & Sanford, M. S. Oxidative C−H activation/C−C bond forming reactions: synthetic scope and mechanistic insights. J. Am. Chem. Soc. 127, 7330–7331 (2005).
Bartholomew, C. H. Mechanisms of catalyst deactivation. Appl. Catal. A 212, 17–60 (2001).
Solymosi, F., Cserényi, J., Szöke, A., Bánsági, T. & Oszko, A. Aromatization of methane over supported and unsupported Mo-based catalysts. J. Catal. 165, 150–161 (1997).
Oyama, S. Preparation and catalytic properties of transition metal carbides and nitrides. Catal. Today 15, 179–200 (1992).
Nandula, A., Trinh, Q. T., Saeys, M. & Alexandrova, A. N. Origin of extraordinary stability of square-planar carbon atoms in surface carbides of cobalt and nickel. Angew. Chem. Int. Ed. 54, 5312–5316 (2015).
Acknowledgements
Acknowledgement is made to the Natural Sciences and Engineering Research Council (NSERC) of Canada for funding. M.M.S. acknowledges the NSERC scholarship support.
Author information
Authors and Affiliations
Contributions
M.M.S. conducted the experiments and analysed the data. M.M.S. and S.A.J. conceived and designed the project and prepared the manuscript
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 2034 kb)
Supplementary information
Crystallographic data for compound 2 (CIF 3129 kb)
Supplementary information
Crystallographic data for compound 3 (CIF 3070 kb)
Supplementary information
Crystallographic data for compound 4 (CIF 49 kb)
Rights and permissions
About this article
Cite this article
Shoshani, M., Johnson, S. Cooperative carbon-atom abstraction from alkenes in the core of a pentanuclear nickel cluster. Nature Chem 9, 1282–1285 (2017). https://doi.org/10.1038/nchem.2840
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2840
This article is cited by
-
Skeletal Ni electrode-catalyzed C-O cleavage of diaryl ethers entails direct elimination via benzyne intermediates
Nature Communications (2022)