Abstract
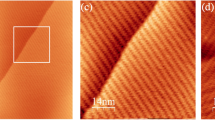
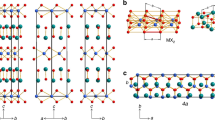
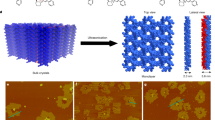
Two-dimensional (2D) supramolecular arrays provide a route to the spatial control of the chemical functionality of a surface, but their deposition is in almost all cases limited to a monolayer termination. Here we investigated the sequential deposition of one 2D array on another to form a supramolecular heterostructure and realize the growth—normal to the underlying substrate—of distinct ordered layers, each of which is stabilized by in-plane hydrogen bonding. For heterostructures formed by depositing terephthalic acid or trimesic acid on cyanuric acid/melamine, we have determined, using atomic force microscopy under ambient conditions, a clear epitaxial arrangement despite the intrinsically distinct symmetries and/or lattice constants of each layer. Structures calculated using classical molecular dynamics are in excellent agreement with the orientation, registry and dimensions of the epitaxial layers. Calculations confirm that van der Waals interactions provide the dominant contribution to the adsorption energy and registry of the layers.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Elemans, J. A. A. W., Lei, S. & De Feyter, S. Molecular and supramolecular networks on surfaces: from two-dimensional crystal engineering to reactivity. Angew. Chem. Int. Ed. 48, 7298–7333 (2009).
Macleod, J. M. & Rosei, F. Molecular self-assembly on graphene. Small 10, 1038–1049 (2014).
Slater (née Phillips), A. G., Beton, P. H. & Champness, N. R. Two-dimensional supramolecular chemistry on surfaces. Chem. Sci. 2, 1440–1448 (2011).
Wang, D., Wan, L.-J. & Bai, C.-L. Formation and structural transition of molecular self-assembly on solid surface investigated by scanning tunneling microscopy. Mater. Sci. Eng. Rep. 70, 169–187 (2010).
Theobald, J. A., Oxtoby, N. S., Phillips, M. A., Champness, N. R. & Beton, P. H. Controlling molecular deposition and layer structure with supramolecular surface assemblies. Nature 424, 1029–1031 (2003).
Baris, B. et al. Robust and open tailored supramolecular networks controlled by the template effect of a silicon surface. Angew. Chem. Int. Ed. 50, 4094–4098 (2011).
Barth, J. V. et al. Building supramolecular nanostructures at surfaces by hydrogen bonding. Angew. Chem. Int. Ed. 39, 1230–1234 (2000).
Barth, J. V., Costantini, G. & Kern, K. Engineering atomic and molecular nanostructures at surfaces. Nature 437, 671–679 (2005).
Maier, S. et al. Nanoscale engineering of molecular porphyrin wires on insulating surfaces. Small 4, 1115–1118 (2008).
Burke, S., Mativetsky, J., Hoffmann, R. & Grütter, P. Nucleation and submonolayer growth of C60 on KBr. Phys. Rev. Lett. 94, 096102 (2005).
Rahe, P. et al. Tuning molecular self-assembly on bulk insulator surfaces by anchoring of the organic building blocks. Adv. Mater. 25, 3948–3956 (2013).
Li, B. et al. Self-assembled air-stable supramolecular porous networks on graphene. ACS Nano 7, 10764–10772 (2013).
Korolkov, V. V. et al. Bimolecular porous supramolecular networks deposited from solution on layered materials: graphite, boron nitride and molybdenum disulphide. Chem. Commun. 50, 8882–8885 (2014).
Korolkov, V. V. et al. Van der Waals-induced chromatic shifts in hydrogen-bonded two-dimensional porphyrin arrays on boron nitride. ACS Nano 9, 10347–10355 (2015).
Ludwig, C., Gompf, B., Petersen, J., Strohmaier, R. & Eisenmenger, W. STM investigations of PTCDA and PTCDI on graphite and MoS2. A systematic study of epitaxy and STM image contrast. Z. Phys. B 93, 365–373 (1994).
Blunt, M. O. et al. Guest-induced growth of a surface-based supramolecular bilayer. Nat. Chem. 3, 74–78 (2011).
Ciesielski, A. et al. Self-templating 2D supramolecular networks: a new avenue to reach control over a bilayer formation. Nanoscale 3, 4125 (2011).
Xu, B., Tao, C., Cullen, W. G., Reutt-Robey, J. E. & Williams, E. D. Chiral symmetry breaking in two-dimensional C60–ACA intermixed systems. Nano Lett. 5, 2207–2211 (2005).
Wei, Y. & Reutt-Robey, J. E. Directed organization of C70 kagome lattice by titanyl phthalocyanine monolayer template. J. Am. Chem. Soc. 133, 15232–15235 (2011).
Skomski, D. & Tait, S. L. Interfacial organic layers for chemical stability and crystalline ordering of thiophene and carboxyl films on a metal surface. J. Phys. Chem. C 118, 1594–1601 (2014).
Chen, W. et al. Orientationally ordered C60 on p-sexiphenyl nanostripes on Ag(111). ACS Nano 2, 693–698 (2008).
Huang, H. et al. Molecular orientation of CuPc thin films on C60/Ag(111). Appl. Phys. Lett. 94, 163303–163304 (2009).
Yoshimoto, S., Sawaguchi, T., Su, W., Jiang, J. & Kobayashi, N. Superstructure formation and rearrangement in the adlayer of a rare-earth-metal triple-decker sandwich complex at the electrochemical interface. Angew. Chem. Int. Ed. 46, 1071–1074 (2007).
Yoshimoto, S. et al. Controlled molecular orientation in an adlayer of a supramolecular assembly consisting of an open-cage C60 derivative and ZnII octaethylporphyrin on Au(111). Angew. Chem. Int. Ed. 43, 3044–3047 (2004).
Hinderhofer, A. & Schreiber, F. Organic–organic heterostructures: concepts and applications. ChemPhysChem 13, 628–643 (2012).
Forker, R. et al. Electronic decoupling of aromatic molecules from a metal by an atomically thin organic spacer. Adv. Mater. 20, 4450–4454 (2008).
Gruenewald, M. et al. Commensurism at electronically weakly interacting phthalocyanine/PTCDA heterointerfaces. Phys. Rev. B 91, 155432 (2015).
Chen, W., Qi, D. C., Huang, H., Gao, X. & Wee, A. T. S. Organic–organic heterojunction interfaces: effect of molecular orientation. Adv. Funct. Mater. 21, 410–424 (2011).
Schmitz-Hübsch, T. et al. Direct observation of organic–organic heteroepitaxy: perylenetetracarboxylic-dianhydride on hexa-peri-benzocoronene on highly ordered pyrolytic graphite. Surf. Sci. 445, 358–367 (2000).
Schwarze, M. et al. Band structure engineering in organic semiconductors. Science 352, 1446–1449 (2016).
Orton, J. & Foxon, T. Molecular Beam Epitaxy: A Short History (Oxford Univ. Press, 2015).
Geim, A. K. & Grigorieva, I. V. Van der Waals heterostructures. Nature 499, 419–425 (2013).
Perdigão, L. M. A., Champness, N. R. & Beton, P. H. Surface self-assembly of the cyanuric acid–melamine hydrogen bonded network. Chem. Commun. 28, 538–540 (2006).
Zhang, X., Chen, T., Chen, Q., Wang, L. & Wan, L.-J. Self-assembly and aggregation of melamine and melamine–uric/cyanuric acid investigated by STM and AFM on solid surfaces. Phys. Chem. Chem. Phys. 11, 7708–7712 (2009).
Staniec, P. A., Perdigão, L. M. A., Rogers, B. L., Champness, N. R. & Beton, P. H. Honeycomb networks and chiral superstructures formed by cyanuric acid and melamine on Au(111). J. Phys. Chem. C 111, 886–893 (2007).
Xu, W. et al. Cyanuric acid and melamine on Au(111): structure and energetics of hydrogen-bonded networks. Small 3, 854–858 (2007).
Ranganathan, A., Pedireddi, V. R. & Rao, C. N. R. Hydrothermal synthesis of organic channel structures: 1:1 hydrogen-bonded adducts of melamine with cyanuric and trithiocyanuric acids. J. Am. Chem. Soc. 121, 1752–1753 (1999).
Griessl, S., Lackinger, M., Edelwirth, M., Hietschold, M. & Heckl, W. M. Self-assembled two-dimensional molecular host–guest architectures from trimesic acid. Single Mol. 3, 25–31 (2002).
Plimpton, S. Fast parallel algorithms for short-range molecular dynamics. J. Comput. Phys. 117, 1–19 (1995).
Jorgensen, W. L. & Tirado-Rives, J. The OPLS [optimized potentials for liquid simulations] potential functions for proteins, energy minimizations for crystals of cyclic peptides and crambin. J. Am. Chem. Soc. 110, 1657–1666 (1988).
Jorgensen, W. L., Maxwell, D. S. & Tirado-Rives, J. Development and testing of the OPLS All-Atom Force Field on conformational energetics and properties of organic liquids. J. Am. Chem. Soc. 118, 11225–11236 (1996).
Kannappan, K., Werblowsky, T. L., Rim, K. T., Berne, B. J. & Flynn, G. W. An experimental and theoretical study of the formation of nanostructures of self-assembled cyanuric acid through hydrogen bond networks on graphite. J. Phys. Chem. B 111, 6634–6642 (2007).
Conti, S. & Cecchini, M. Accurate and efficient calculation of the desorption energy of small molecules from graphene. J. Phys. Chem. C 119, 1867–1879 (2015).
Hutter, J., Iannuzzi, M., Schiffmann, F. & Van de Vondele, J. Cp2k: atomistic simulations of condensed matter systems. Wiley Interdiscip. Rev. Comput. Mol. Sci. 4, 15–25 (2014).
Lee, J. H. A study on a boron-nitride nanotube as a gigahertz oscillator. J. Korean Phys. Soc. 49, 172–176 (2006).
Clair, S. et al. STM study of terephthalic acid self-assembly on Au(111): hydrogen-bonded sheets on an inhomogeneous substrate. J. Phys. Chem. B 108, 14585–14590 (2004).
Horcas, I. et al. WSXM: a software for scanning probe microscopy and a tool for nanotechnology. Rev. Sci. Instrum. 78, 013705 (2007).
Acknowledgements
We are grateful to the Leverhulme Trust for providing financial support under grant RPG-2016-104 and to the Engineering and Physical Sciences Research Council for support through grant EP/N033906/1. E.B. gratefully acknowledges the receipt of a European Research Council Consolidator Grant; M.B. and E.B. acknowledge the University of Nottingham and Midplus High Performance Computing facilities for providing computational time. K.W. and T.T. acknowledge support from the Elemental Strategy Initiative conducted by MEXT, Japan, and the Japan Society for the Promotion of Science KAKENHI Grant Numbers JP26248061, JP15K21722 and JP25106006.
Author information
Authors and Affiliations
Contributions
V.V.K. and P.H.B. conceived and designed the experiments, which were carried out by V.V.K. The hBN crystals were grown by K.W. and T.T. The numerical calculations were conceived through a discussion between M.B., E.B., P.H.B. and V.V.K., and were carried out by M.B. with additional input from E.B. The paper was written by P.H.B., V.V.K., M.B. and E.B. with revisions and comments from all of the authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 7654 kb)
Rights and permissions
About this article
Cite this article
Korolkov, V., Baldoni, M., Watanabe, K. et al. Supramolecular heterostructures formed by sequential epitaxial deposition of two-dimensional hydrogen-bonded arrays. Nature Chem 9, 1191–1197 (2017). https://doi.org/10.1038/nchem.2824
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2824
This article is cited by
-
Extending on-surface synthesis from 2D to 3D by cycloaddition with C60
Nature Communications (2023)
-
Shape control in 2D molecular nanosheets by tuning anisotropic intermolecular interactions and assembly kinetics
Nature Communications (2023)
-
Ultra-high resolution imaging of thin films and single strands of polythiophene using atomic force microscopy
Nature Communications (2019)
-
Pyridine-induced interfacial structural transformation of tetraphenylethylene derivatives investigated by scanning tunneling microscopy
Nano Research (2018)
-
Supramolecular networks stabilise and functionalise black phosphorus
Nature Communications (2017)