Abstract
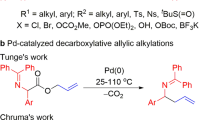
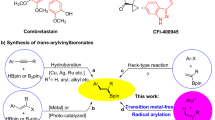
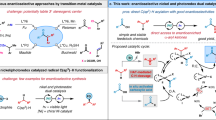
Direct C(sp3)–C(sp2) bond formation under transition-metal-free conditions offers an atom-economical, inexpensive and environmentally benign alternative to traditional transition-metal-catalysed cross-coupling reactions. A new chemo- and regioselective coupling protocol between 3-aryl-substituted-1,1-diphenyl-2-azaallyl derivatives and vinyl bromides has been developed. This is the first transition-metal-free cross-coupling of azaallyls with vinyl bromide electrophiles and delivers allylic amines in excellent yields (up to 99%). This relatively simple and mild protocol offers a direct and practical strategy for the synthesis of high-value allylic amine building blocks that does not require the use of transition metals, special initiators or photoredox catalysts. Radical clock experiments, electron paramagnetic resonance studies and density functional theory calculations point to an unprecedented substrate-dependent coupling mechanism. Furthermore, an electron paramagnetic resonance signal was observed when the N-benzyl benzophenone ketimine was subjected to silylamide base, supporting the formation of radical species upon deprotonation. The unique mechanisms outlined herein could pave the way for new approaches to transition-metal-free C–C bond formations.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Negishi, E. Magical power of transition metals: past, present, and future (Nobel Lecture). Angew. Chem. Int. Ed. 50, 6738–6764 (2011).
Suzuki, A. Cross-coupling reactions of organoboranes: an easy way to construct C–C bonds (Nobel Lecture). Angew. Chem. Int. Ed. 50, 6722–6737 (2011).
Sun, C. L. & Shi, Z. J. Transition-metal-free coupling reactions. Chem. Rev. 114, 9219–9280 (2014).
Petranyi, G., Ryder, N. & Stutz, A. Allylamine derivatives: new class of synthetic antifungal agents inhibiting fungal squalene epoxidase. Science 224, 1239–1241 (1984).
Roggen, M. & Carreira, E. M. Stereospecific substitution of allylic alcohols to give optically active primary allylic amines: unique reactivity of a (P,alkene)Ir complex modulated by iodide. J. Am. Chem. Soc. 132, 11917–11919 (2010).
Yamashita, Y., Gopalarathnam, A. & Hartwig, J. F. Iridium-catalyzed, asymmetric amination of allylic alcohols activated by Lewis acids. J. Am. Chem. Soc. 129, 7508–7509 (2007).
Patel, S. J. & Jamison, T. F. Asymmetric catalytic coupling of organoboranes, alkynes, and imines with a removable (trialkylsilyloxy)ethyl group—direct access to enantiomerically pure primary allylic amines. Angew. Chem. Int. Ed. 43, 3941–3944 (2004).
Shi, X., Kiesman, W. F., Levina, A. & Xin, Z. Catalytic asymmetric petasis reactions of vinylboronates. J. Org. Chem. 78, 9415–9423 (2013).
Overman, L. E. A general method for the synthesis of amines by the rearrangement of allylic trichloroacetimidates. 1,3 Transposition of alcohol and amine functions. J. Am. Chem. Soc. 98, 2901–2910 (1976).
Ngai, M.-Y., Barchuk, A. & Krische, M. J. Enantioselective iridium-catalyzed imine vinylation: optically enriched allylic amines via alkyne–imine reductive coupling mediated by hydrogen. J. Am. Chem. Soc. 129, 12644–12645 (2007).
Skucas, E., Kong, J. R. & Krische, M. J. Enantioselective reductive coupling of acetylene to N-arylsulfonyl imines via rhodium catalyzed C–C bond-forming hydrogenation: (Z)-dienyl allylic amines. J. Am. Chem. Soc. 129, 7242–7243 (2007).
Li, M., Berritt, S. & Walsh, P. J. Palladium-catalyzed regioselective arylation of 1,1,3-triaryl-2-azaallyl anions with aryl chlorides. Org. Lett. 16, 4312–4315 (2014).
Li, M. et al. Palladium-catalyzed C–H arylation of α,β-unsaturated imines: catalyst-controlled synthesis of enamine and allylic amine derivatives. Angew. Chem. Int. Ed. 128, 2875–2879 (2016).
Li, M., Yucel, B., Adrio, J., Bellomo, A. & Walsh, P. J. Synthesis of diarylmethylamines via palladium-catalyzed regioselective arylation of 1,1,3-triaryl-2-azaallyl anions. Chem. Sci. 5, 2383–2391 (2014).
Li, M . et al. Umpolung synthesis of diarylmethylamines via palladium-catalyzed arylation of N-benzyl aldimines. Adv. Synth. Catal. 358, 1910–1915 (2016).
Wu, Y., Hu, L., Li, Z. & Deng, L. Catalytic asymmetric Umpolung reactions of imines. Nature 523, 445–450 (2015).
Burger, E. C. & Tunge, J. A. Synthesis of homoallylic amines via the palladium-catalyzed decarboxylative coupling of amino acid derivatives. J. Am. Chem. Soc. 128, 10002–10003 (2006).
Fields, W. H. & Chruma, J. J. Palladium-catalyzed decarboxylative benzylation of diphenylglycinate imines. Org. Lett. 12, 316–319 (2009).
Niwa, T., Yorimitsu, H. & Oshima, K. Palladium-catalyzed benzylic arylation of N-benzylxanthone imine. Org. Lett. 10, 4689–4691 (2008).
Zhu, Y. & Buchwald, S. L. Ligand-controlled asymmetric arylation of aliphatic α-amino anion equivalents. J. Am. Chem. Soc. 136, 4500–4503 (2014).
Liu, Y. E. et al. Enzyme-inspired axially chiral pyridoxamines armed with a cooperative lateral amine chain for enantioselective biomimetic transamination. J. Am. Chem. Soc. 138, 10730–10733 (2016).
Ankner, T., Cosner, C. C. & Helquist, P. Palladium- and nickel-catalyzed alkenylation of enolates. Chem. Eur. J. 19, 1858–1871 (2013).
Grigalunas, M., Ankner, T., Norrby, P. O., Wiest, O. & Helquist, P. Palladium-catalyzed alkenylation of ketone enolates under mild conditions. Org. Lett. 16, 3970–3973 (2014).
Hardegger, L. A., Habegger, J. & Donohoe, T. J. Modular synthesis of highly substituted pyridines via enolate alpha-alkenylation. Org. Lett. 17, 3222–3225 (2015).
Padilla-Salinas, R., Walvoord, R. R., Tcyrulnikov, S. & Kozlowski, M. C. Nitroethylation of vinyl triflates and bromides. Org. Lett. 15, 3966–3969 (2013).
Piers, E. & Marais, P. C. A new five-membered ring annulation method based on palladium(0)-catalyzed intramolecular coupling of vinyl iodide and enolate anion functions. J. Org. Chem. 55, 3454–3455 (1990).
Solé, D., Peidró, E. & Bonjoch, J. Palladium-catalyzed intramolecular coupling of vinyl halides and ketone enolates. synthesis of bridged azabicyclic compounds. Org. Lett. 2, 2225–2228 (2000).
Yang, X., Kim, B. S., Li, M. & Walsh, P. J. Palladium-catalyzed selective alpha-alkenylation of pyridylmethyl ethers with vinyl bromides. Org. Lett. 18, 2371–2374 (2016).
Niwa, T., Suehiro, T., Yorimitsu, H. & Oshima, K. Carbon–carbon bond formations at the benzylic positions of N-benzylxanthone imines and N-benzyldi-1-naphthyl ketone imine. Tetrahedron 65, 5125–5131 (2009).
Tang, S., Park, J. Y., Yeagley, A. A., Sabat, M. & Chruma, J. J. Decarboxylative generation of 2-azaallyl anions: 2-iminoalcohols via a decarboxylative Erlenmeyer reaction. Org. Lett. 17, 2042–2045 (2015).
Yeagley, A. A., Lowder, M. A. & Chruma, J. J. Tandem C–C bond-forming processes: interception of the Pd-catalyzed decarboxylative allylation of allyl diphenylglycinate imines with activated olefins. Org. Lett. 11, 4022–4025 (2009).
Kauffmann, T., Berger, D., Scheerer, B. & Woltermann, A. Anionic 3+2 cycloaddition of a 1,2-diazaallyllithium compound. Angew. Chem. Int. Ed. 9, 961–962 (1970).
Pandiancherri, S. & Lupton, D. W. Preparation of 2-azaallyl anions and imines from N-chloroamines and their cycloaddition and allylation. Tetrahedron Lett. 52, 671–674 (2011).
Taber, D. F., Sahli, A., Yu, H. & Meagley, R. P. Efficient intramolecular C–H insertion by an alkylidene carbene generated from a vinyl chloride. J. Org. Chem. 60, 6571–6573 (1995).
Taber, D. F., Sikkander, M. I. & Storck, P. H. Enantioselective synthesis of (+)-majusculone. J. Org. Chem. 72, 4098–4101 (2007).
Bernasconi, C. F. & Rappoport, Z. Recent advances in our mechanistic understanding of SNV reactions. Acc. Chem. Res. 42, 993–1003 (2009).
Rappoport, Z. Nucleophilic vinylic substitution. A single- or a multi-step process? Acc. Chem. Res. 14, 7–15 (1981).
Rappoport, Z. The rapid steps in nucleophilic vinylic addition–elimination substitution. Recent developments. Acc. Chem. Res. 25, 474–479 (1992).
Bach, R. D., Baboul, A. G. & Schlegel, H. B. Inversion versus retention of configuration for nucleophilic substitution at vinylic carbon. J. Am. Chem. Soc. 123, 5787–5793 (2001).
Castro, E. A., Gazitua, M. & Santos, J. G. Kinetics and mechanism of the anilinolysis of aryl 4-nitrophenyl carbonates in aqueous ethanol. J. Org. Chem. 70, 8088–8092 (2005).
Castro, E. A., Ramos, M. & Santos, J. G. Concerted pyridinolysis of aryl 2,4,6-trinitrophenyl carbonates. J. Org. Chem. 74, 6374–6377 (2009).
Williams, A. Concerted mechanisms of acyl group transfer reactions in solution. Acc. Chem. Res. 22, 387–392 (1989).
Fernandez, I., Bickelhaupt, F. M. & Uggerud, E. Reactivity in nucleophilic vinylic substitution (SNV): SNVπ versus SNVσ mechanistic dichotomy. J. Org. Chem. 78, 8574–8584 (2013).
Baum, A. A. & Karnischky, L. A. Photochemical formation of oxazolidines from aryl ketones and aliphatic imines. J. Am. Chem. Soc. 95, 3072–3074 (1973).
Dannenberg, J. J. & Tanaka, K. Theoretical studies of radical recombination reactions. 1. Allyl and azaallyl radicals. J. Am. Chem. Soc. 107, 671–674 (1985).
Malassa, A., Agthe, C., Görls, H., Friedrich, M. & Westerhausen, M. Deprotonation and dehydrogenation of di(2-pyridylmethyl)amine with M[N(SiMe3)2]2 (M=Mn, Fe, Co, Zn) and Fe(C6H2-2,4,6-Me3)2 . J. Organomet. Chem. 695, 1641–1650 (2010).
Pallagi, I., Toró, A. & Horváth, G. Mechanism of the Gibbs reaction. Part 4.1 Indophenol formation via N-chlorobenzoquinone imine radical anions. The Aza-SRN2 chain reaction mechanism. Chain initiation with 1,4-benzoquinones and cyanide ion. J. Org. Chem. 64, 6530–6540 (1999).
Giese, B. The stereoselectivity of intermolecular free radical reactions [New Synthetic Methods (78)]. Angew. Chem. Int. Ed. 28, 969–980 (1989).
Acknowledgements
The authors acknowledge the National Science Foundation (CHE-1464744 to P.J.W. and CHE-1464778 to M.C.K.) and the National Institutes of Health (GM-104349 to P.J.W. and GM-087605 to M.C.K.) for financial support. J.A. acknowledges support from Ministerio de Educación, Cultura y Deporte, Subprograma Estatal de Movilidad, Salvador de Madariaga. Computational support was provided by XSEDE on SDSC Gordon (TG-CHEM120052). This work was also financially supported by a SICAM Fellowship by Jiangsu National Synergetic Innovation Center for Advanced Materials. The authors thank S. Montel of UPenn and K. Scheidt of Northwestern University for discussions.
Author information
Authors and Affiliations
Contributions
M.L. and P.J.W. conceived and designed the experiments. M.L., S.B., A.P.-E., A.Y., X.Y., J.A. and G.H. performed the research. O.G. and M.C.K. designed and performed the DFT computational study. M.L. and E.N.-O. performed the EPR study. M.L., O.G., M.C.K. and P.J.W. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 4977 kb)
Rights and permissions
About this article
Cite this article
Li, M., Gutierrez, O., Berritt, S. et al. Transition-metal-free chemo- and regioselective vinylation of azaallyls. Nature Chem 9, 997–1004 (2017). https://doi.org/10.1038/nchem.2760
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2760
This article is cited by
-
Iron-catalysed reductive cross-coupling of glycosyl radicals for the stereoselective synthesis of C-glycosides
Nature Synthesis (2022)
-
Synthesis of highly congested α-amino acids and peptides
Nature Synthesis (2022)
-
Catalytic asymmetric α C(sp3)–H addition of benzylamines to aldehydes
Nature Catalysis (2022)
-
Dearomatization of aromatic asmic isocyanides to complex cyclohexadienes
Nature Communications (2022)
-
α-Amino acid and peptide synthesis using catalytic cross-dehydrogenative coupling
Nature Synthesis (2022)