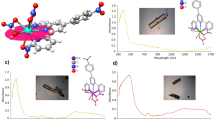
Abstract
Berkelium (Bk) has been predicted to be the only transplutonium element able to exhibit both +III and +IV oxidation states in solution, but evidence of a stable oxidized Bk chelate has so far remained elusive. Here we describe the stabilization of the heaviest 4+ ion of the periodic table, under mild aqueous conditions, using a siderophore derivative. The resulting Bk(IV) complex exhibits luminescence via sensitization through an intramolecular antenna effect. This neutral Bk(IV) coordination compound is not sequestered by the protein siderocalin—a mammalian metal transporter—in contrast to the negatively charged species obtained with neighbouring trivalent actinides americium, curium and californium (Cf). The corresponding Cf(III)–ligand–protein ternary adduct was characterized by X-ray diffraction analysis. Combined with theoretical predictions, these data add significant insight to the field of transplutonium chemistry, and may lead to innovative Bk separation and purification processes.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Accession codes
References
Kosyakov, V. N. Perspective methods for Berkelium–249 preparation and application. J. Nucl. Sci. Technol. 39, 42–44 (2002).
Vértes, A., Nagy, S., Klencsár, Z. & Lovas, R. G. Handbook of Nuclear Chemistry: Instrumentation, Separation Techniques Environmental Issues (Kluwer Academic, 2003).
Hobart, D. E. & Peterson, J. R. in The Chemistry of the Actinide and Transactinide Elements (eds Morss, L. R., Edelstein, N., Fuger, J. & Katz, J. J.) Ch. 10, 1444–1498 (Springer, 2006).
Silver, M. A. et al. Characterization of berkelium(III) dipicolinate and borate compounds in solution and the solid state. Science 353, aaf3762 (2016).
Hungate, F. P. et al. Preliminary data on 253Es and 249Bk metabolism in rats. Health Phys. 22, 653–656 (1972).
Taylor, G. N. et al. Microscopic distribution of californium-249 and berkelium-249 in the soft tissues of beagles. Health Phys. 22, 691–693 (1972).
Runde, W. H. & Mincher, B. J. Higher oxidation states of americium: preparation, characterization and use for separations. Chem. Rev. 111, 5723–5741 (2011).
Czerwinski, K. R. Studies of Fundamental Properties of Rutherfordium (Element 104) Using Organic Complexing Agents. PhD thesis, Univ. California (1992).
Thompson, S. G., Ghiorso, A. & Seaborg, G. T. Element 97. Phys. Rev. 77, 838–839 (1950).
Thompson, S. G., Cunningham, B. B. & Seaborg, G. T. Chemical properties of Berkelium. J. Am. Chem. Soc. 72, 2798–2801 (1950).
Antonio, M. R., Williams, C. W. & Soderholm, L. Berkelium redox speciation. Radiochim. Acta 90, 851–856 (2002).
Cotton, S. Lanthanide and Actinide Chemistry (Wiley, 2006).
Stokely, J. R., Baybarz, R. D. & Peterson, J. R. The formal potential of the Bk(IV)-Bk(III) couple in several media. J. Inorg. Nucl. Chem. 34, 392–393 (1972).
Wadsworth, E., Duke, F. R. & Goetz, C. A. Present status of cerium (IV)–cerium (III) potentials. Anal. Chem. 29, 1824–1825 (1957).
Gutmacher, R. G. et al. The absorption spectra of Bk3+ and Bk4+ in solution. J. Inorg. Nucl. Chem. 29, 2341–2345 (1967).
Baybarz, R. D. & Stokely, J. R. Absorption spectra of Bk(III) and Bk(IV) in several media. J. Inorg. Nucl. Chem. 34, 739–746 (1972).
Gutmacher, R. G., Bodé, D. D., Lougheed, R. W. & Hulet, E. K. The stability of tetravalent berkelium in acid solution and the absorption spectra of Bk(IV) and Bk(III). J. Inorg. Nucl. Chem. 35, 979–994 (1973).
Milyukova, M. S., Malikov, D. A., Kuzovkina, E. V. & Myasoedov, B. F. Extraction of tetravalent berkelium by high molecular weight amines in the presence of heteropolyanions. J. Radioanal. Nucl. Chem. 104, 81–89 (1986).
Payne, G. F. & Peterson, J. R. Possible stabilization of the tetravalent oxidation state of berkelium and californium in acetonitrile with triphenylarsine oxide. Inorganica Chim. Acta 139, 111–112 (1987).
Morris, D. E., Hobart, D. E. & Palmer, P. D. Voltammetric investigation of the berkelium(IV/III) couple in concentrated aqueous carbonate solutions. Radiochim. Acta 49, 125–134 (1990).
Bunin, D. I. et al. Dose-dependent efficacy and safety toxicology of hydroxypyridinonate actinide decorporation agents in rodents: towards a safe and effective human dosing regimen. Radiat. Res. 179, 171–182 (2013).
Choi, T. A. et al. Biodistribution of the multidentate hydroxypyridinonate ligand [14C]-3,4,3-LI(1,2-HOPO), a potent actinide decorporation agent. Drug Dev. Res. 76, 107–122 (2015).
Choi, T. A. et al. In vitro metabolism and stability of the actinide chelating agent 3,4,3-LI(1,2-HOPO). J. Pharm. Sci. 104, 1832–1838 (2015).
Sturzbecher-Hoehne, M., Deblonde, G. J. -P. & Abergel, R. J. Solution thermodynamic evaluation of hydroxypyridinonate chelators 3,4,3-LI(1,2-HOPO) and 5-LIO(Me-3,2-HOPO) for UO2(VI) and Th(IV) decorporation. Radiochim. Acta 101, 359–366 (2013).
Sturzbecher-Hoehne, M., Kullgren, B., Jarvis, E. E., An, D. D. & Abergel, R. J. Highly luminescent and stable hydroxypyridinonate complexes: a step towards new curium decontamination strategies. Chem. Eur. J. 20, 9962–9968 (2014).
Sturzbecher-Hoehne, M., Choi, T. A. & Abergel, R. J. Hydroxypyridinonate complex stability of group (IV) metals and tetravalent f-block elements: the key to the next generation of chelating agents for radiopharmaceuticals. Inorg. Chem. 54, 3462–3468 (2015).
Sturzbecher-Hoehne, M., Yang, P., D'Aléo, A. & Abergel, R. J. Intramolecular sensitization of americium luminescence in solution: shining light on short-lived forbidden 5f transitions. Dalton Trans. 45, 9912–9919, (2016).
Deblonde, G. J.-P., Sturzbecher-Hoehne, M. & Abergel, R. J. Solution thermodynamic stability of complexes formed with the octadentate hydroxypyridinonate ligand 3,4,3-LI(1,2-HOPO): a critical feature for efficient chelation of lanthanide(IV) and actinide(IV) Ions. Inorg. Chem. 52, 8805–8811 (2013).
Shannon, R. Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Crystallogr. A 32, 751–767 (1976).
Baybarz, R. D. Dissociation constants of the transplutonium element chelates of diethylenetriaminepenta-acetic acid (DTPA) and the application of DTPA chelates to solvent extraction separations of transplutonium elements from the lanthanide elements. J. Inorg. Nucl. Chem. 27, 1831–1839 (1965).
Nugent, L. J. et al. Intramolecular energy transfer and sensitized luminescence in actinide (III) β-diketone chelates. J. Phys. Chem. 73, 1540–1549 (1969).
Carnall, W., Beitz, J. & Crosswhite, H. Electronic energy level and intensity correlations in the spectra of the trivalent actinide aquo ions. III. Bk3+. J. Chem. Phys. 80, 2301–2308 (1984).
Barbanel, A. Nephelauxetic effect and hypersensitivity in the optical spectra of actinides. Radiochim. Acta 78, 91–96 (1997).
Liu, G., Carnall, W., Jursich, G. & Williams, C. Analysis of the crystal-field spectra of the actinide tetrafluorides. II. AmF4, CmF4, Cm4+:CeF4, and Bk4+:CeF4 . J. Chem. Phys. 101, 8277–8289 (1994).
Carnall, W. T. A Systematic Analysis of the Spectra of Trivalent Actinide Chlorides in D3h Site Symmetry. (Argonne National Laboratory, 1989).
Jursich, G. M. et al. Laser induced fluorescence of 249Bk4+ in CeF4 . Inorganica Chim. Acta 139, 273–274 (1987).
Nugent, L. J., Baybarz, R. D., Burnett, J. L. & Ryan, J. L. Electron-transfer and fd absorption bands of some lanthanide and actinide complexes and the standard (II-III) oxidation potential for each member of the lanthanide and actinide series. J. Phys. Chem. 77, 1528–1539 (1973).
Allred, B. E. et al. Siderocalin-mediated recognition, sensitization, and cellular uptake of actinides. Proc. Natl Acad. Sci. USA 112, 10342–10347 (2015).
Lundberg, D. & Persson, I. The size of actinoid (III) ions–structural analysis vs. common misinterpretations. Coord. Chem. Rev. 318, 131–134 (2016).
Myasoedov, B. & Lebedev, I. Latest achievements in the analytical chemistry of actinides. J. Radioanal. Nucl. Chem. 147, 5–26 (1991).
Maruyama, T. et al. Rapid chemical separation for Bk. J. Nucl. Radiochem. Sci. 3, 155–158 (2002).
Peppard, D. F., Moline, S. W. & Mason, G. W. Isolation of berkelium by solvent extraction of the tetravalent species. J. Inorg. Nucl. Chem. 4, 344–348 (1957).
Moore, F. L. New method for rapid separation of berkelium (IV) from cerium (IV) by anion exchange. Anal. Chem. 39, 1874–1876 (1967).
Moore, F. L. Liquid–liquid extraction method for the separation of cerium (IV) from berkelium (IV) and other elements. Anal. Chem. 41, 1658–1661 (1969).
Chudinov, E. G. & Pirozhkov, S. V. The separation of berkelium(III) from cerium(III). J. Radioanal. Chem. 10, 41–46 (1972).
Liu, Y.-F. et al. Procedures for a fast separation of berkelium from complex mixtures of reaction products. J. Radioanal. Nucl. Chem. 76, 119–124 (1983).
Chang, P. Y. et al. Analytical methods for the bioavailability evaluation of hydroxypyridinonate actinide decorporation agents in pre-clinical pharmacokinetic studies. J. Chromatogr. Sep. Tech. 4, 196 (2013).
Goetz, D. H. et al. The neutrophil lipocalin NGAL is a bacteriostatic agent that interferes with siderophore-mediated iron acquisition. Mol. Cell 10, 1033–1043 (2002).
Valiev, M. et al. NWChem: a comprehensive and scalable open-source solution for large scale molecular simulations. Comput. Phys. Commun. 181, 1477–1489 (2010).
Becke, A. D. Density-functional exchange-energy approximation with correct asymptotic behavior. Phys. Rev. A 38, 3098–3100 (1988).
Lee, C., Yang, W. & Parr, R. G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 37, 785–789 (1988).
Küchle, W., Dolg, M., Stoll, H. & Preuss, H. Energy-adjusted pseudopotentials for the actinides. Parameter sets and test calculations for thorium and thorium monoxide. J. Chem. Phys. 100, 7535–7542 (1994).
Godbout, N., Salahub, D. R., Andzelm, J. & Wimmer, E. Optimization of Gaussian-type basis sets for local spin density functional calculations. Part I. Boron through neon, optimization technique and validation. Can. J. Chem. 70, 560–571 (1992).
Runge, E. & Gross, E. K. Density-functional theory for time-dependent systems. Phys. Rev. Lett. 52, 997–1000 (1984).
Brabec, J. et al. Efficient algorithms for estimating the absorption spectrum within linear response TDDFT. J. Chem. Theory Comput. 11, 5197–5208 (2015).
Jensen, H. J. Aa. et al. DIRAC12. http://www.diracprogram.org (2012)
Dyall, K. G. Relativistic double-zeta, triple-zeta, and quadruple-zeta basis sets for the actinides Ac–Lr. Theor. Chem. Acc. 117, 491–500 (2007).
Berman, H. M. et al. The Protein Data Bank. Nucleic Acids Res. 28, 235–242 (2000).
Acknowledgements
This work was supported by the US Department of Energy (DoE), Office of Science Early Career Research Program and Office of Science, Office of Basic Energy Sciences, Chemical Sciences, Geosciences, and Biosciences Division at the Lawrence Berkeley National Laboratory under contract DE-AC02-05CH11231 (R.J.A.), by the National Institutes of Health (R01DK073462, R.K.S.), and by the Scientific Discovery through Advanced Computing (SciDAC) program of the US DoE, Office of Science, Office of Advanced Scientific Computing and Office of Basic Energy Sciences (W.A.d.J.). The Radiochemical Engineering and Development Center at Oak Ridge National Laboratory is supported by the US DoE, Isotope Development and Production for Research and Applications Program. The Advanced Light Source (ALS) and Energy Research Scientific Computing Center (NERSC) are supported by the Director, Office of Science, and Office of Basic Energy Sciences, of the US DoE under contract no. DE-AC02-05CH11231. The Innovative and Novel Computational Impact on Theory and Experiment (INCITE) program provided an award of computer time through the Oak Ridge Leadership Computing Facility, a US DoE Office of Science User Facility supported under Contract DE-AC05-00OR22725. We thank M. Allaire, S. Morton, J. Bramble, K. Engle, M. Dupray, and I. Tadesse for assistance in implementing diffraction data collection on radioactive crystals at ALS 5.0.2 beamline.
Author information
Authors and Affiliations
Contributions
G.J.-P.D., M.S.-H., W.A.d.J., R.K.S., and R.J.A. designed the research. G.J.-P.D., M.S.-H., and M.-C.I. collected and analysed optical spectroscopy and mass spectrometry data. R.J.A. and D. D. A crystallized the protein–metal adducts. P.B.R., D.D.A., and C.Y.R. collected and analysed crystallographic data. P.B.R. and R.K.S. solved the structures. J.B. and W.A.d.J. performed theoretical computations. All of the authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 952 kb)
Rights and permissions
About this article
Cite this article
Deblonde, GP., Sturzbecher-Hoehne, M., Rupert, P. et al. Chelation and stabilization of berkelium in oxidation state +IV. Nature Chem 9, 843–849 (2017). https://doi.org/10.1038/nchem.2759
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2759
This article is cited by
-
Polyoxometalates as ligands to synthesize, isolate and characterize compounds of rare isotopes on the microgram scale
Nature Chemistry (2022)
-
Rare radioisotopes at the ready
Nature Chemistry (2022)
-
DGA resin capacity factors for Ac, Am and Th under tetravalent actinide selective complexation
Journal of Radioanalytical and Nuclear Chemistry (2021)
-
Creation of an unexpected plane of enhanced covalency in cerium(III) and berkelium(III) terpyridyl complexes
Nature Communications (2021)
-
Open questions in transplutonium coordination chemistry
Communications Chemistry (2020)