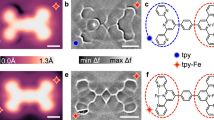
Abstract
Elementary steps in enantioselective heterogeneous catalysis take place on the catalyst surface and the targeted synthesis of a desired enantiomer requires the implantation of chiral information at the surface, which can be achieved—for example—by adsorbing chiral molecules. Studies of the structures of complexes formed between adsorbed prochiral reagents and chiral molecules yield information on the forces exerting stereocontrol, but further insight could be gained by studying the dynamics of their interactions. Here, using time-lapsed scanning tunnelling microscopy and density functional theory, we observe coupling between multiple stereochemical states within individual non-covalently bonded chirality-transfer complexes on a metal surface. We identify two modes of transformation between stereochemical states and find that the prochiral reagent can sample several complexation geometries during the lifetime of a complex, switching between states of opposing prochirality in the process. These results provide insight on the contribution of individual stereochemical states to the overall enantioselectivity of reactions occurring on catalyst surfaces.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Corma, A. Heterogeneous catalysis: understanding for designing, and designing for applications. Angew. Chem. Int. Ed. 55, 6112–6113 (2016).
Ye, R., Hurlburt, T. J., Sabyrov, K., Alayoglu, S. & Somorjai, G. A. Molecular catalysis science: perspective on unifying the fields of catalysis. Proc. Natl Acad. Sci. USA 113, 5159–5166 (2016).
Lauritsen, J. V. & Besenbacher, F. Atom-resolved scanning tunneling microscopy investigations of molecular adsorption on MoS2 and CoMoS hydrodesulfurization catalysts. J. Catal. 328, 49–58 (2015).
Kyriakou, G. et al. Isolated metal atom geometries as a strategy for selective heterogeneous hydrogenation. Science 335, 1209–1212 (2012).
Lucci, F. R. et al. Selective hydrogenation of 1,3-butadiene on platinum–copper alloys at the single-atom limit. Nat. Commun. 6, 8550 (2015).
Orito, Y., Imai, S., Niwa, S. & Nguyen, G.-H. Asymmetric hydrogenation of methyl benzoylformate using platinum-carbon catalysts modified with cinchonidine. J. Synth. Org. Chem. Jpn 37, 173–174 (1979).
Gellman, A. J., Tysoe, W. T. & Zaera, F. Surface chemistry for enantioselective catalysis. Catal. Lett. 145, 220–232 (2015).
Kyriakou, G., Beaumont, S. K. & Lambert, R. M. Aspects of heterogeneous enantioselective catalysis by metals. Langmuir 27, 9687–9695 (2011).
Dementyev, P., Peter, M., Adamovsky, S. & Schauermann, S. Chirally-modified metal surfaces: energetics of interaction with chiral molecules. Phys. Chem. Chem. Phys. 17, 22726–22735 (2015).
Wilson, K. E., Trant, A. G. & Baddeley, C. J. Interaction of the pro-chiral molecule, methylacetoacetate, with (S)-aspartic acid modified Ni(111). J. Phys. Chem. C 116, 1092–1098 (2012).
Meemken, F., Hungerbühler, K. & Baiker, A. Monitoring surface processes during heterogeneous asymmetric hydrogenation of ketones on a chirally modified platinum catalyst by operando spectroscopy. Angew. Chem. Int. Ed. 53, 8640–8644 (2014).
Raval, R. Assembling molecular guidance systems for heterogeneous enantioselective catalysis. Cat. Tech. 5, 12–28 (2001).
Larrea, C. R. & Baddeley, C. J. Fabrication of a high-quality, porous, surface-confined covalent organic framework on a reactive metal surface. Chem. Phys. Chem. 17, 971–975 (2016).
Zaera, F. Regio-, stereo-, and enantioselectivity in hydrocarbon conversion on metal surfaces. Acc. Chem. Res. 42, 1152–1160 (2009).
Fang, Y. et al. Dynamic control over supramolecular handedness by selecting chiral induction pathways at the solution–solid interface. Nat. Chem. 8, 711–717 (2016).
Ernst, K.-H. Stereochemical recognition of helicenes on metal surfaces. Acc. Chem. Res. 49, 1182–1190 (2016).
Forster, M. & Raval, R. Simple rules and the emergence of complexity in surface chirality. Chem. Commun. 52, 14075–14084 (2016).
Yun, Y. & Gellman, A. J. Adsorption-induced auto-amplification of enantiomeric excess on an achiral surface. Nat. Chem. 7, 520–525 (2015).
Yasukawa, T., Suzuki, A., Miyamura, H., Nishino, K. & Kobayashi, S. Chiral metal nanoparticle systems as heterogeneous catalysts beyond homogeneous metal complex catalysts for asymmetric addition of arylboronic acids to α,β-unsaturated carbonyl compounds. J. Am. Chem. Soc. 137, 6616–6623 (2015).
Rodríguez-García, L., Hungerbühler, K., Baiker, A. & Meemken, F. Discrimination of active species in liquid-phase hydrogenation on supported noble metal catalyst: an operando spectroscopic study on the asymmetric hydrogenation of ketopantolactone on Pt/Al2O3 and Pt/C modified by cinchonidine. Catal. Today 283, 66–73 (2017).
Baiker, A. Crucial aspects in the design of chirally modified noble metal catalysts for asymmetric hydrogenation of activated ketones. Chem. Soc. Rev. 44, 7449–7464 (2015).
Demers-Carpentier, V. et al. Direct observation of molecular preorganization for chirality transfer on a catalyst surface. Science 334, 776–780 (2011).
Mahapatra, M. et al. Enhanced hydrogenation activity and diastereomeric interactions of methyl pyruvate co-adsorbed with R-1-(1-naphthyl)ethylamine on Pd(111). Nat. Commun. 7, 12380 (2016).
Demers-Carpentier, V. et al. Stereodirection of an α-ketoester at sub-molecular sites on chirally modified Pt(111): heterogeneous asymmetric catalysis. J. Am. Chem. Soc. 135, 9999–10002 (2013).
Svane, K. et al. Single-chiral-catalytic-surface-sites: STM and DFT study of stereodirecting complexes formed between (R)-1-(1-naphthyl)ethylamine and ketopantolactone on Pt(111). Catal. Sci. Technol. 5, 743–753 (2015).
Orglmeister, E., Bürgi, T., Mallat, T. & Baiker, A. Conformational rigidity: a necessary prerequisite of chiral modifiers used in heterogeneous enantioselective catalysis? J. Catal. 232, 137–142 (2005).
Groves, M. N. et al. Structure determination of chemisorbed chirality transfer complexes: accelerated STM analysis and exchange-correlation functional sensitivity. Surf. Sci. 629, 48–56 (2014).
Goubert, G. et al. Walking-like diffusion of two-footed asymmetric aromatic adsorbates on Pt(111). Surf. Sci. 629, 123–131 (2014).
Dong, Y., Svane, K. L., Lemay, J.-C., Groves, M. N. & McBreen, P. H. STM study of ketopantolactone/(R)-1-(1-naphthyl)ethylamine complexes on Pt(111): comparison of prochiral and enantiomeric ratios and examination of the contribution of CH⋯OC bonding. ACS Catal. 7, 1757–1765 (2017).
Schneider, H. J. Hydrogen bonds with fluorine. Studies in solution, in gas phase and by computations, conflicting conclusions from crystallographic analyses. Chem. Sci. 3, 1381–1394 (2012).
Witte, G., Range, H., Toennies, J. P. & Wöll, C. External vibrations of molecules on surfaces: the case of CO and benzene. J. Electron Spectros. Relat. Phenom. 64–65, 715–723 (1993).
Orglmeister, E., Mallat, T. & Baiker, A. Synthetic modifiers for platinum in the enantioselective hydrogenation of ketopantolactone: a test for the mechanistic models of ketone hydrogenation. Adv. Synth. Catal. 347, 78–86 (2005).
Yasukawa, T., Miyamura, H. & Kobayashi, S. Chiral ligand-modified metal nanoparticles as unique catalysts for asymmetric C–C bond forming reactions. How are active species generated? ACS Catal. 6, 7979–7988 (2016).
Schmidt, E., Vargas, A., Mallat, T. & Baiker, A. Shape-selective enantioselective hydrogenation on Pt nanoparticles. J. Am. Chem. Soc. 131, 12358–12367 (2009).
Meemken, F., Baiker, A., Schenker, S. & Hungerbühler, K. Chiral modification of platinum by co-adsorbed cinchonidine and trifluoroacetic acid: origin of enhanced stereocontrol in the hydrogenation of trifluoroacetophenone. Chem. Eur. J. 20, 1298–1309 (2014).
Beak, P. et al. Dynamic thermodyamic resolution: control of enantioselectivity through diastereomeric equilibration. Acc. Chem. Res. 33, 715–727 (2000).
Blaser, H.-U. Looking back on 35 years of industrial catalysis. Chim. Int. J. Chem. 69, 393–406 (2015).
Mortensen, J. J., Hansen, L. B. & Jacobsen, K. W. Real-space grid implementation of the projector augmented wave method. Phys. Rev. B. 71, 035109 (2005).
Enkovaara, J. et al. Electronic structure calculations with GPAW: a real-space implementation of the projector augmented-wave method. J. Phys. Condens. Matter 22, 253202 (2010).
Klimeš, J., Bowler, D. R. & Michaelides, A. Chemical accuracy for the van der Waals density functional. J. Phys. Condens. Matter. 22, 22201 (2010).
Kolsbjerg, E. L., Groves, M. N. & Hammer, B. An automatic nudged elastic band method. J. Chem. Phys. 145, 094107 (2016).
Rasmussen, A. M. H., Groves, M. N. & Hammer, B. Remote activation of chemical bonds in heterogeneous catalysis. ACS Catal. 4, 1182–1188 (2014).
Acknowledgements
The STM work was carried out within the FQRNT Center on Catalysis and Green Chemistry (CCVC). Y.D. acknowledges an NSERC graduate student scholarship. J.-C.L. and G.G. acknowledge FQRNT scholarships. The work was supported by NSERC Discovery, FQRNT and CFI grants. Financial support from the Danish Council for Independent Research Nature and Universe and the Lundbeck Foundation is also gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
G.G., Y.D. and P.H.McB. designed the STM experiments. G.G., Y.D. and J.-C.L. performed the STM experiments. Y.D. and G.G. performed the statistical analysis of the STM data. M.N.G. and B.H. designed, performed and interpreted the DFT calculations. G.G., Y.D and P.H.McB. co-wrote the manuscript with input from all the authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 1072 kb)
Rights and permissions
About this article
Cite this article
Goubert, G., Dong, Y., Groves, M. et al. Monitoring interconversion between stereochemical states in single chirality-transfer complexes on a platinum surface. Nature Chem 9, 531–536 (2017). https://doi.org/10.1038/nchem.2753
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2753
This article is cited by
-
Giving catalysts a hand
Nature Chemistry (2017)