Abstract
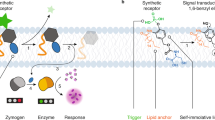
Signal transduction through G-protein-coupled receptors (GPCRs) involves binding to signalling molecules at the cell surface, which leads to global changes in molecular conformation that are communicated through the membrane. Artificial mechanisms for communication involving ligand binding and global conformational switching have been demonstrated so far only in the solution phase. Here, we report a membrane-bound synthetic receptor that responds to binding of a ligand by undergoing a conformational change that is propagated over several nanometres, deep into the phospholipid bilayer. Our design uses a helical foldamer core, with structural features borrowed from a class of membrane-active fungal antibiotics, ligated to a water-compatible, metal-centred binding site and a conformationally responsive fluorophore. Using the fluorophore as a remote reporter of conformational change, we find that binding of specific carboxylate ligands to a Cu(II) cofactor at the binding site perturbs the foldamer's global conformation, mimicking the conformational response of a GPCR to ligand binding.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Mantri, S., Sapra, K. T., Cheley, S., Sharp, T. H. & Bayley, H. An engineered dimeric protein pore that spans adjacent lipid bilayers. Nat. Commun. 4, 1725 (2013).
Villar, G., Graham, A. D. & Bayley, H. A tissue-like printed material. Science 340, 48–52 (2013).
Sakai, N. & Matile, S. Synthetic ion channels. Langmuir 29, 9031–9040 (2013).
Barton, P., Hunter, C. A., Potter, T. J., Webb, S. J. & Williams, N. H. Transmembrane signalling. Angew. Chem. Int. Ed. 41, 3878–3881 (2002).
Bernitzki, K. & Schrader, T. Entirely artificial signal transduction with a primary messenger. Angew. Chem. Int. Ed. 48, 8001–8005 (2009).
Schrader, T., Maue, M. & Ellermann, M. Entirely artificial signal transduction with adrenaline. J. Recept. Signal Transduct. Res. 26, 473–485 (2006).
Huber, T. & Sakmar, T. P. Chemical biology methods for investigating G protein-coupled receptor signaling. Chem. Biol. 21, 1224–1237 (2014).
Manglik, A. & Kobilka, B. The role of protein dynamics in GPCR function: insights from the β2AR and rhodopsin. Curr. Opin. Cell Biol. 27, 136–143 (2014).
Venkatakrishnan, A. J. et al. Molecular signatures of G-protein-coupled receptors. Nature 494, 185–194 (2013).
Nussinov, R., Tsai, C.-J. & Liu, J. Principles of allosteric interactions in cell signaling. J. Am. Chem. Soc. 136, 17692–17701 (2014).
Groves, J. T. & Kuriyan, J. Molecular mechanisms in signal transduction at the membrane. Nat. Struct. Mol. Biol. 17, 659–665 (2010).
Gurevich, V. V. & Gurevich, E. V. GPCR monomers and oligomers: it takes all kinds. Trends Neurosci. 31, 74–81 (2008).
Krauss, R. & Koert, U. Molecular signal transduction by conformational transmission. Synlett 598–608 (2003).
Ousaka, N. & Inai, Y. Transfer of noncovalent chiral information along an optically inactive helical peptide chain: allosteric control of asymmetry of the C-terminal site by external molecule that binds to the N-terminal site. J. Org. Chem. 74, 1429–1439 (2009).
Brown, R. A., Diemer, V., Webb, S. J. & Clayden, J. End-to-end conformational communication through a synthetic purinergic receptor by ligand-induced helicity switching. Nat. Chem. 5, 853–860 (2013).
Brioche, J. et al. Conformational switching of a foldamer in a multicomponent system by pH-filtered selection between competing noncovalent interactions. J. Am. Chem. Soc. 137, 6680–6691 (2015).
Toniolo, C. & Brückner, H. Peptaibiotics (HCA/Wiley-VCH, 2009).
Toniolo, C. & Benedetti, E. The polypeptide 310-helix. Trends Biochem. Sci. 16, 350–353 (1991).
Jones, J. E. et al. Length-dependent formation of transmembrane pores by 310-helical α-aminoisobutyric acid foldamers. J. Am. Chem. Soc. 138, 688–695 (2016).
Hecht, S. & Huc, I. Foldamers: Structure, Properties and Applications (Wiley-VCH, 2007).
Le Bailly, B. A. F. et al. Flaws in foldamers: conformational uniformity and signal decay in achiral helical peptide oligomers. Chem. Sci. 6, 2313–2322 (2015).
Clayden, J., Castellanos, A., Solà, J. & Morris, G. A. Quantifying end-to-end conformational communication of chirality through an achiral peptide chain. Angew. Chem. Int. Ed. 48, 5962–5965 (2009).
Solà, J., Morris, G. A. & Clayden, J. Measuring screw-sense preference in a helical oligomer by comparison of 13C NMR signal separation at slow and fast exchange. J. Am. Chem. Soc. 133, 3712–3715 (2011).
Brown, R. A., Marcelli, T., De Poli, M., Solà, J. & Clayden, J. Induction of unexpected left-handed helicity by an N-terminal L-amino acid in an otherwise achiral peptide chain. Angew. Chem. Int. Ed. 51, 1395–1399 (2012).
De Poli, M. et al. Engineering the structure of an N-terminal β-turn to maximize screw-sense preference in achiral helical peptide chains. J. Org. Chem. 79, 4659–4675 (2014).
Solà, J., Fletcher, S. P., Castellanos, A. & Clayden, J. Nanometer-range communication of stereochemical information by reversible switching of molecular helicity. Angew. Chem. Int. Ed. 49, 6836–6839 (2010).
Byrne, L. et al. Foldamer-mediated remote stereocontrol: >1,60 asymmetric induction. Angew. Chem. Int. Ed. 53, 151–155 (2014).
Ousaka, N., Takeyama, Y., Iida, H. & Yashima, E. Chiral information harvesting in dendritic metallopeptides. Nat. Chem. 3, 856–861 (2011).
De Poli, M. et al. Conformational photoswitching of a synthetic peptide foldamer bound within a phospholipid bilayer. Science 352, 575–580 (2016).
Wei, N., Murthy, N. N., Chen, Q., Zubieta, J. & Karlin, K. D. Copper(I)/dioxygen reactivity of mononuclear complexes with pyridyl and quinolyl tripodal tetradentate ligands: reversible formation of Cu:O2 = 1:1 and 2:1 adducts. Inorg. Chem. 33, 1953–1965 (1994).
Joyce, L. A. et al. A simple method for the determination of enantiomeric excess and identity of chiral carboxylic acids. J. Am. Chem. Soc. 133, 13746–13752 (2011).
Kučerka, N. et al. Lipid bilayer structure determined by the simultaneous analysis of neutron and X-ray scattering data. Biophys. J. 95, 2356–2367 (2008).
Jones, O. T. & Lee, A. G. Interactions of pyrene derivatives with lipid bilayers and with (Ca2+-Mg2+)-ATPase. Biochemistry 24, 2195–2202 (1985).
Strange, P. G. Agonist binding, agonist affinity and agonist efficacy at G protein-coupled receptors. Br. J. Pharmacol. 153, 1353–1363 (2008).
L'Heureux, G. P. & Fragata, M. Micropolarities of lipid bilayers and micelles: 4. Dielectric constant determinations of unilamellar phosphatidylcholine vesicles with the probes pyrene and 16-(1-pyrenyl)hexadecanoic acid. Biophys. Chem. 30, 293–301 (1988).
Schwyzer, R. Molecular mechanism of opioid receptor selection. Biochemistry 25, 6335–6342 (1986).
Frederickson, R. C. & Norris, F. H. Enkephalin-induced depression of single neurons in brain areas with opiate receptors—antagonism by naloxone. Science 194, 440–442 (1976).
Rothman, R. B. & Westfall, T. C. Allosteric modulation by leucine-enkephalin of [3H]naloxone binding in rat brain. Eur. J. Pharmacol. 72, 365–368 (1981).
Acknowledgements
This work was supported by the European Research Council (advanced grant ROCOCO) and the EPRSC (grants EP/N009134/1 and EP/K039547).
Author information
Authors and Affiliations
Contributions
S.J.W. and J.C. conceived the project and wrote the paper. F.G.A.L., B.A.F.L.B., S.J.W. and J.C. devised the experiments and analysed the data. F.G.A.L. and B.A.F.L.B. carried out the experiments.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 7965 kb)
Rights and permissions
About this article
Cite this article
Lister, F., Le Bailly, B., Webb, S. et al. Ligand-modulated conformational switching in a fully synthetic membrane-bound receptor. Nature Chem 9, 420–425 (2017). https://doi.org/10.1038/nchem.2736
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2736
This article is cited by
-
Engineering receptor-mediated transmembrane signaling in artificial and living cells
Communications Materials (2023)
-
Transmembrane signaling by a synthetic receptor in artificial cells
Nature Communications (2023)
-
Regulation of artificial supramolecular transmembrane signal transduction by selenium-containing artificial enzyme receptors
Nano Research (2023)
-
De novo design of discrete, stable 310-helix peptide assemblies
Nature (2022)
-
Chirality transmission in macromolecular domains
Nature Communications (2022)