Abstract
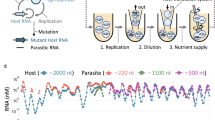
How new species emerge in nature is still incompletely understood and difficult to study directly. Self-replicating molecules provide a simple model that allows us to capture the fundamental processes that occur in species formation. We have been able to monitor in real time and at a molecular level the diversification of self-replicating molecules into two distinct sets that compete for two different building blocks (‘food’) and so capture an important aspect of the process by which species may arise. The results show that the second replicator set is a descendant of the first and that both sets are kinetic products that oppose the thermodynamic preference of the system. The sets occupy related but complementary food niches. As diversification into sets takes place on the timescale of weeks and can be investigated at the molecular level, this work opens up new opportunities for experimentally investigating the process through which species arise both in real time and with enhanced detail.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Schluter, D. Ecology and the origin of species. Trends Ecol. Evol. 16, 372–380 (2001).
Sobel, J. M., Chen, G. F., Watt, L. R. & Schemske, D. W. The biology of speciation. Evolution 64, 295–315 (2010).
Hart, M. W. The species concept as an emergent property of population biology. Evolution 65, 613–616 (2011).
Hey, J. On the failure of modern species concepts. Trends Ecol. Evol. 21, 447–450 (2006).
Orgel, L. E. Molecular replication. Nature 358, 203–209 (1992).
Wintner, E. A. & Rebek, J. Autocatalysis and the generation of self-replicating systems. Acta Chem. Scand. 50, 469–485 (1996).
Lee, D. H., Severin, K. & Ghadiri, M. R. Autocatalytic networks: the transition from molecular self-replication to molecular ecosystems. Curr. Opin. Chem. Biol. 1, 491–496 (1997).
Ghosh, I. & Chmielewski, J. Peptide self-assembly as a model of proteins in the pre-genomic world. Curr. Opin. Chem. Biol. 8, 640–644 (2004).
Paul, N. & Joyce, G. F. Minimal self-replicating systems. Curr. Opin. Chem. Biol. 8, 634–639 (2004).
Dadon, Z., Wagner, N. & Ashkenasy, G. The road to non-enzymatic molecular networks. Angew. Chem. Int. Ed. 47, 6128–6136 (2008).
Vidonne, A. & Philp, D. Making molecules make themselves—the chemistry of artificial replicators. Eur. J. Org. Chem. 5, 593–610 (2009).
Kassianidis, E., Pearson, R. J., Wood, E. A. & Philp, D. Designing instructable networks using synthetic replicators. Discuss. Faraday Soc. 145, 235–254 (2010).
Bissette, A. J. & Fletcher, S. P. Mechanisms of autocatalysis. Angew. Chem. Int. Ed. 52, 12800–12826 (2013).
Szathmáry, E. The evolution of replicators. Phil. Trans. R. Soc. Lond. B 355, 1669–1676 (2000).
Eigen, M. Selforganization of matter and the evolution of biological macromolecules. Naturwissenschaften 58, 465–523 (1971).
Eigen, M. & Schuster, P. The hypercycle. A principle of natural self-organisation. Part A: emergence of the hypercycle. Naturwissenschaften 64, 541–565 (1977).
Domingo, E. & Perales, C. From quasi-species theory to viral quasi-species: how complexity has permeated virology. Math. Model. Nat. Phenom. 7, 105–122 (2012).
Domingo, E., Sheldon, J. & Perales, C. Viral quasispecies evolution. Microbiol. Mol. Biol. Rev. 76, 159–216 (2012).
Eigen, M. Steps toward Life: a Perspective on Evolution 79–87 (Oxford Univ. Press, 1992).
Eigen, M., McCaskill, J. & Schuster, P. Molecular quasi-species. J. Phys. Chem. 92, 6881–6891 (1988).
Nowak, M. A. What is a quasispecies? Trends Ecol. Evol. 7, 118–121 (1992).
Bull, J. J., Meyers, L. A. & Lachmann, M. Quasispecies made simple. PLoS Comput. Biol. 6, e61 (2005).
Ruiz-Mirazo, K., Briones, C. & de la Escosura, A. Prebiotic systems chemistry: new perspectives for the origins of life. Chem. Rev. 114, 285–366 (2014).
Reek, J. N. H. & Otto, S. Dynamic Combinatorial Chemistry (Wiley-VCH, 2010).
Cougnon, F. B. L. & Sanders, J. K. M. Evolution of dynamic combinatorial chemistry. Acc. Chem. Res. 45, 2211–2221 (2012).
Otto, S. Dynamic molecular networks: from synthetic receptors to self-replicators. Acc. Chem. Res. 45, 2200–2210 (2012).
Li, J., Nowak, P. & Otto, S. Dynamic combinatorial libraries: from exploring molecular recognition to systems chemistry. J. Am. Chem. Soc. 135, 9222–9239 (2013).
Lehn, J. M. From supramolecular chemistry towards constitutional dynamic chemistry and adaptive chemistry. Chem. Soc. Rev. 36, 151–160 (2007).
Corbett, P. T. et al. Dynamic combinatorial chemistry. Chem. Rev. 106, 3652–3711 (2006).
Carnall, J. M. A. et al. Mechanosensitive self-replication driven by self-organization. Science 327, 1502–1506 (2010).
Malakoutikhah, M. et al. Uncovering the selection criteria for the emergence of multi-building-block replicators from dynamic combinatorial libraries. J. Am. Chem. Soc. 135, 18406–18417 (2013).
Moulin, E. & Giuseppone, N. Dynamic combinatorial self-replicating systems. Top. Curr. Chem. 322, 87–105 (2012).
Ji, Q., Lirag, R. C. & Miljanić, O. Š. Kinetically controlled phenomena in dynamic combinatorial libraries. Chem. Soc. Rev. 43, 1873–1884 (2014).
Xu, S. & Giuseppone, N. Self-duplicating amplification in a dynamic combinatorial library. J. Am. Chem. Soc. 130, 1826–1827 (2008).
Sadownik, J. W. & Philp, D. A simple synthetic replicator amplifies itself from a dynamic reagent pool. Angew. Chem. Int. Ed. 47, 9965–9970 (2008).
del Amo, V. & Philp, D. Integrating replication-based selection strategies in dynamic covalent systems. Chem. Eur. J. 16, 13304–13318 (2010).
Otto, S., Furlan, R. L. E. & Sanders, J. K. M. Dynamic combinatorial libraries of macrocyclic disulfides in water. J. Am. Chem. Soc. 122, 12063–12064 (2000).
Colomb-Delsuc, M., Mattia, E., Sadownik, J. W. & Otto, S. Exponential replication enabled through a fibre elongation/breakage mechanism. Nature Commun. 6, 7427 (2015).
Pal, A. et al. Controlling the structure and length of self-synthesizing supramolecular polymers through nucleated growth and disassembly. Angew. Chem. Int. Ed. 54, 7852–7856 (2015).
Pascal, R., Pross, A. & Sutherland, J. D. Towards an evolutionary theory of the origin of life based on kinetics and thermodynamics. Open Biol. 3, 130156 (2013).
Acknowledgements
We are grateful to F. Weissing and S. van Doorn for useful discussions and we acknowledge support from the European Research Council, The Netherlands Organization for Scientific Research, European Cooperation in Science and Technology CM1005 and CM1304, and the Dutch Ministry of Education, Culture and Science (Gravitation Program 024.001.035).
Author information
Authors and Affiliations
Contributions
J.W.S. conceived, designed and performed the experiments. E.M. and P.N. performed the UPLC-MS analysis. S.O. supervised the overall project. J.W.S., P.N. and S.O. co-wrote the paper. All the authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 3106 kb)
Rights and permissions
About this article
Cite this article
Sadownik, J., Mattia, E., Nowak, P. et al. Diversification of self-replicating molecules. Nature Chem 8, 264–269 (2016). https://doi.org/10.1038/nchem.2419
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2419
This article is cited by
-
Biomimetic selenocystine based dynamic combinatorial chemistry for thiol-disulfide exchange
Nature Communications (2021)
-
The rise of intelligent matter
Nature (2021)
-
Self-replication of a quantum artificial organism driven by single-photon pulses
Scientific Reports (2021)
-
Emergence of low-symmetry foldamers from single monomers
Nature Chemistry (2020)
-
Selection from a pool of self-assembling lipid replicators
Nature Communications (2020)