Abstract
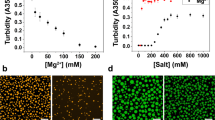
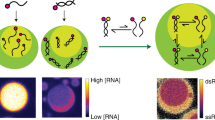
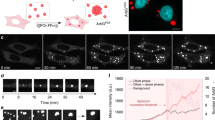
Biological cells are highly organized, with numerous subcellular compartments. Phosphorylation has been hypothesized as a means to control the assembly/disassembly of liquid-like RNA- and protein-rich intracellular bodies, or liquid organelles, that lack delimiting membranes. Here, we demonstrate that charge-mediated phase separation, or complex coacervation, of RNAs with cationic peptides can generate simple model liquid organelles capable of reversibly compartmentalizing biomolecules. Formation and dissolution of these liquid bodies was controlled by changes in peptide phosphorylation state using a kinase/phosphatase enzyme pair. The droplet-generating phase transition responded to modification of even a single serine residue. Electrostatic interactions between the short cationic peptides and the much longer polyanionic RNAs drove phase separation. Coacervates were also formed on silica beads, a primitive model for localization at specific intracellular sites. This work supports phosphoregulation of complex coacervation as a viable mechanism for dynamic intracellular compartmentalization in membraneless organelles.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Brangwynne, C. P. et al. Germline P granules are liquid droplets that localize by controlled dissolution/condensation. Science 324, 1729–1732 (2009).
Brangwynne, C. P., Mitchison, T. J. & Hyman, A. A. Active liquid-like behavior of nucleoli determines their size and shape in Xenopus laevis oocytes. Proc. Natl Acad. Sci. USA 108, 4334–4339 (2011).
Brangwynne, C. P. Soft active aggregates: mechanics, dynamics and self-assembly of liquid-like intracellular protein bodies. Soft Matter 7, 3052–3059 (2011).
Nott, T. J. et al. Phase transition of a disordered nuage protein generates environmentally responsive membraneless organelles. Mol. Cell 57, 936–947 (2015).
Zwicker, D., Decker, M., Jaensch, S., Hyman, A. A. & Jülicher, F. Centrosomes are autocatalytic droplets of pericentriolar material organized by centrioles. Proc. Natl Acad. Sci. USA 111, E2636–E2645 (2014).
Hyman, A. A., Weber, C. A. & Jülicher, F. Liquid–liquid phase separation in biology. Annu. Rev. Cell Dev. Biol. 30, 39–58 (2014).
Mao, Y. S., Zhang, B. & Spector, D. L. Biogenesis and function of nuclear bodies. Trends Genet. 27, 295–306 (2011).
Dundr, M. Nuclear bodies: multifunctional companions of the genome. Curr. Opin. Cell Biol. 24, 415–422 (2012).
Boisvert, F.-M., van Koningsbruggen, S., Navascués, J. & Lamond, A. I. The multifunctional nucleolus. Nature Rev. Mol. Cell Biol. 8, 574–585 (2007).
Hernandez-Verdun, D. Assembly and disassembly of the nucleolus during the cell cycle. Nucleus 2, 189–194 (2011).
Matera, A. G., Terns, R. M. & Terns, M. P. Non-coding RNAs: lessons from the small nuclear and small nucleolar RNAs. Nature Rev. Mol. Cell Biol. 8, 209–220 (2007).
Hearst, S. M. et al. Cajal-body formation correlates with differential coilin phosphorylation in primary and transformed cell lines. J. Cell Sci. 122, 1872–1881 (2009).
Li, P. et al. Phase transitions in the assembly of multivalent signalling proteins. Nature 483, 336–340 (2012).
Endicott, J. A., Noble, M. E. M. & Johnson, L. N. The structural basis for control of eukaryotic protein kinases. Annu. Rev. Biochem. 81, 587–613 (2012).
Ubersax, J. A. & Ferrell, J. E. Mechanisms of specificity in protein phosphorylation. Nature Rev. Mol. Cell Biol. 8, 530–541 (2007).
Wang, J. T. et al. Regulation of RNA granule dynamics by phosphorylation of serine-rich, intrinsically-disordered proteins in C. elegans. eLife 3, e04591 (2014).
Wippich, F. et al. Dual specificity kinase DYRK3 couples stress granule condensation/dissolution to mTORC1 signaling. Cell 152, 791–805 (2013).
Scott, J. D., Dessauer, C. W. & Taskén, K. Creating order from chaos: cellular regulation by kinase anchoring. Annu. Rev. Pharmacol. Toxicol. 53, 187–210 (2013).
Borg, M. et al. Polyelectrostatic interactions of disordered ligands suggest a physical basis for ultrasensitivity. Proc. Natl Acad. Sci. USA 104, 9650–9655 (2007).
Antonov, M., Mazzawi, M. & Dubin, P. L. Entering and exiting the protein–polyelectrolyte coacervate phase via nonmonotonic salt dependence of critical conditions. Biomacromolecules 11, 51–59 (2010).
Priftis, D., Laugel, N. & Tirrell, M. Thermodynamic characterization of polypeptide complex coacervation. Langmuir 28, 15947–15957 (2012).
Evreinova, T. N., Karnaukhov, W. N., Mamontova, T. W. & Ivanizki, G. R. The interaction of biological macromolecules in coacervate systems. J. Colloid Interface Sci. 36, 18–23 (1971).
Arfin, N. & Bohidar, H. B. Condensation, complex coacervation, and overcharging during DNA–gelatin interactions in aqueous solutions. J. Phys. Chem. B 116, 13192–13199 (2012).
Keating, C. D. Aqueous phase separation as a possible route to compartmentalization of biological molecules. Acc. Chem. Res. 45, 2114–2124 (2012).
Aumiller, W. M., Davis, B. W. & Keating, C. D. Phase separation as a possible means of nuclear compartmentalization. Int. Rev. Cell Mol. Biol. 307, 109–149 (2014).
De Kruif, C. G., Weinbreck, F. & de Vries, R. Complex coacervation of proteins and anionic polysaccharides. Curr. Opin. Colloid Interface Sci. 9, 340–349 (2004).
Wang, Q. & Schlenoff, J. B. The polyelectrolyte complex/coacervate continuum. Macromolecules 47, 3108–3116 (2014).
Aumiller, W. M. et al. Coupled enzyme reactions performed in heterogeneous reaction media: experiments and modeling for glucose oxidase and horseradish peroxidase in a PEG/citrate aqueous two-phase system. J. Phys. Chem. B 118, 2506–2517 (2014).
Strulson, C. A., Molden, R. C., Keating, C. D. & Bevilacqua, P. C. RNA catalysis through compartmentalization. Nature Chem. 4, 941–946 (2012).
Koga, S., Williams, D. S., Perriman, A. W. & Mann, S. Peptide–nucleotide microdroplets as a step towards a membrane-free protocell model. Nature Chem. 3, 720–724 (2011).
Black, K. A. et al. Protein encapsulation via polypeptide complex coacervation. ACS Macro Lett. 3, 1088–1091 (2014).
Tang, T.-Y. D. et al. Fatty acid membrane assembly on coacervate microdroplets as a step towards a hybrid protocell model. Nature Chem. 6, 527–533 (2014).
Sokolova, E. et al. Enhanced transcription rates in membrane-free protocells formed by coacervation of cell lysate. Proc. Natl Acad. Sci. USA 110, 11692–11697 (2013).
Semenov, S. N. et al. Rational design of functional and tunable oscillating enzymatic networks. Nature Chem. 7, 160–165 (2015).
Richards, E. G., Flessel, C. P. & Fresco, J. R. Polynucleotides. VI. Molecular properties and conformation of polyribouridylic acid. Biopolymers 1, 431–446 (1963).
Simpkins, H. & Richards, E. G. Spectrophotometric titration studies on poly(uridylic acid). Biopolymers 5, 551–560 (1967).
Kemp, B. E. Phosphorylation of acyl and dansyl derivatives of the peptide Leu-Arg-Arg-Ala-Ser-Leu-Gly by the cAMP-dependent protein kinase. J. Biol. Chem. 255, 2914–2918 (1980).
Kemp, B. E., Graves, D. J., Benjamini, E. & Krebs, E. G. Role of multiple basic residues in determining substrate specificity of cyclic AMP-dependent protein kinase. J. Biol. Chem. 252, 4888–4894 (1977).
Priftis, D. & Tirrell, M. Phase behaviour and complex coacervation of aqueous polypeptide solutions. Soft Matter 8, 9396–9405 (2012).
Spruijt, E., Westphal, A. H., Borst, J. W., Stuart, M. A. C. & van der Gucht, J. Binodal compositions of polyeletrolyte complexes. Macromolecules 43, 6476–6484 (2010).
Zhuo, S., Clemens, J. C., Hakes, D. J., Barford, D. & Dixon, J. E. Expression, purification, crystallization, and biochemical characterization of a recombinant protein phosphatase. J. Biol. Chem. 268, 17754–17761 (1993).
Mitić, N. et al. The catalytic mechanisms of binuclear metallohydrolases. Chem. Rev. 106, 3338–3363 (2006).
Reiter, N. J., White, D. J. & Rusnak, F. Inhibition of bacteriophage lambda protein phosphatase by organic and oxoanion inhibitors. Biochemistry 41, 1051–1059 (2002).
Adams, J. A. Kinetic and catalytic mechanisms of protein kinases. Chem. Rev. 101, 2271–2290 (2001).
Johnson, D. A., Akamine, P., Radzio-Andzelm, E., Madhusudan, M. & Taylor, S. S. Dynamics of cAMP-dependent protein kinase. Chem. Rev. 101, 2243–2270 (2001).
Rosevear, P. R. et al. NMR studies of the backbone protons and secondary structure of pentapeptide and heptapeptide substrates bound to bovine heart protein kinase. Biochemistry 23, 3161–3173 (1984).
Adams, J. A. & Taylor, S. S. Divalent metal ions influence catalysis and active-site accessibility in the cAMP-dependent protein kinase. Protein Sci. 2, 2177–2186 (1993).
Obenauer, J. C., Cantley, L. C. & Yaffe, M. B. Scansite 2.0: proteome-wide prediction of cell signaling interactions using short sequence motifs. Nucleic Acids Res. 31, 3635–3641 (2003).
Harris, D. C. Quantitative Chemical Analysis 6th edn (W.H. Freeman & Co., 2003).
Weber, S. C. & Brangwynne, C. P. Getting RNA and protein in phase. Cell 149, 1188–1191 (2012).
Uversky, V. N., Kuznetsova, I. M., Turoverov, K. K. & Zaslavsky, B. Intrinsically disordered proteins as crucial constituents of cellular aqueous two phase systems and coacervates. FEBS Lett. 589, 15–22 (2015).
Elbaum-Garfinkle, S. et al. The disordered P granule protein LAF-1 drives phase separation into droplets with tunable viscosity and dynamics. Proc. Natl Acad. Sci. USA 112, 7189–7194 (2015).
Till, J. H., Annan, R. S., Carr, S. A. & Miller, W. T. Use of synthetic peptide libraries and phosphopeptide-selective mass spectrometry to probe protein kinase substrate specificity. J. Biol. Chem. 269, 7423–7428 (1994).
Acknowledgements
This work was supported by the National Science Foundation (grant no. MCB-1244180). The authors thank P. Bevilacqua for discussions about RNA, including suggesting polyU RNA. The authors also thank D. Kirby for assisting with image analysis.
Author information
Authors and Affiliations
Contributions
W.M.A. performed the experiments. W.M.A and C.D.K. conceived and designed the experiments, analysed the data and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 2699 kb)
Supplementary information
Supplementary Movie 1 (MOV 530 kb)
Supplementary information
Supplementary Movie 2 (MOV 2363 kb)
Supplementary information
Supplementary Movie 3 (MOV 2437 kb)
Supplementary information
Supplementary Movie 4 (MOV 399 kb)
Supplementary information
Supplementary Movie 5 (MOV 2347 kb)
Supplementary information
Supplementary Movie 6 (MOV 971 kb)
Supplementary information
Supplementary Movie 7 (MOV 1166 kb)
Rights and permissions
About this article
Cite this article
Aumiller, W., Keating, C. Phosphorylation-mediated RNA/peptide complex coacervation as a model for intracellular liquid organelles. Nature Chem 8, 129–137 (2016). https://doi.org/10.1038/nchem.2414
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2414
This article is cited by
-
Crosstalk between protein post-translational modifications and phase separation
Cell Communication and Signaling (2024)
-
Dipeptide coacervates as artificial membraneless organelles for bioorthogonal catalysis
Nature Communications (2024)
-
Multicompartmental coacervate-based protocell by spontaneous droplet evaporation
Nature Communications (2024)
-
Biomolecular condensates formed by designer minimalistic peptides
Nature Communications (2023)
-
Non-enzymatic oligonucleotide ligation in coacervate protocells sustains compartment-content coupling
Nature Communications (2023)