Abstract
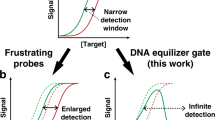
Hybridization of complementary sequences is one of the central tenets of nucleic acid chemistry; however, the unintended binding of closely related sequences limits the accuracy of hybridization-based approaches to analysing nucleic acids. Thermodynamics-guided probe design and empirical optimization of the reaction conditions have been used to enable the discrimination of single-nucleotide variants, but typically these approaches provide only an approximately 25-fold difference in binding affinity. Here we show that simulations of the binding kinetics are both necessary and sufficient to design nucleic acid probe systems with consistently high specificity as they enable the discovery of an optimal combination of thermodynamic parameters. Simulation-guided probe systems designed against 44 sequences of different target single-nucleotide variants showed between a 200- and 3,000-fold (median 890) higher binding affinity than their corresponding wild-type sequences. As a demonstration of the usefulness of this simulation-guided design approach, we developed probes that, in combination with PCR amplification, detect low concentrations of variant alleles (1%) in human genomic DNA.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Kim, S. & Misra, A. SNP genotyping: technologies and biomedical applications. Annu. Rev. Biomed. Eng. 9, 289–320 (2007).
Schwarzenbach, H., Hoon, D. S. B. & Pantel, K. Cell-free nucleic acids as biomarkers in cancer patients. Nature Rev. Cancer 11, 426–437 (2011).
Diehl, F. et al. Detection and quantification of mutations in the plasma of patients with colorectal tumors. Proc. Natl Acad. Sci. 102, 16368–16373 (2005).
Vogelstein, B. et al. Cancer genome landscapes. Science 339, 1546–1558 (2013).
Schmitt, M. et al. Detection of ultra-rare mutations by next-generation sequencing. Proc. Natl Acad. Sci. USA 109, 14508–14513 (2012).
Leamon, J. H., Link, D. R., Egholm, M. & Rothberg, J. M. Overview: methods and applications for droplet compartmentalization of biology. Nature Methods 3, 541–543 (2006).
Compton, J. Nucleic acid sequence-based amplification. Nature 350, 91–92 (1991).
Mori, Y. & Notomi, T. Loop-mediated isothermal amplification (LAMP): a rapid, accurate, and cost-effective diagnostic method for infectious diseases. J. Infect. Chemother. 15, 62–69 (2009).
Van Ness, J., Van Ness, L. K. & Galas, D. J. Isothermal reactions for the amplification of oligonucleotides. Proc. Natl Acad. Sci. USA 100, 4504–4509 (2003).
Lizardi, P. et al. Mutation detection and single-molecule counting using isothermal rolling-circle amplification. Nature Genet. 19, 225–232 (1998).
Schena, M., Shalon, D., Davis, R. W. & Brown, P. O. Quantitative monitoring of gene expression patterns with a complementary DNA microarray. Science 270, 467–470 (1995).
Gunderson, K. L., Steemers, F. J., Lee, G., Mendoza, L. G. & Chee, M. S. A genome-wide scalable SNP genotyping assay using microarray technology. Nature Biotechnol. 37, 549–554 (2005).
Geiss, G. K. et al. Direct multiplexed measurement of gene expression with color-coded probe pairs. Nature Biotechnol. 26, 317–325 (2008).
Dunbar, S. A., Vander Zee, C. A., Oliver, K. G., Karem, K. L. & Jacobson, J. W. Quantitative, multiplexed detection of bacterial pathogens: DNA and protein applications of the Luminex LabMAP system. J. Microbiol. Methods 53, 245–252 (2003).
Pel, J. et al. Nonlinear electrophoretic response yields a unique parameter for separation of biomolecules. Proc. Natl Acad. Sci. USA 106, 14796–14801 (2009).
Morlan, J., Baker, J. & Sinicropi, D. Mutation detection by real-time PCR: a simple, robust and highly selective method. PloS ONE 4, e4584 (2009).
Tyagi, S. & Kramer, F. R. Molecular beacons: probes that fluoresce upon hybridization. Nature Biotechnol. 14, 303–308 (1996).
Tyagi, S., Bratu, D. P., & Kramer, F. R. Multicolor molecular beacons for allele discrimination. Nature Biotechnol. 16, 49–53 (1998).
Li, Q., Luan, G., Guo, Q. & Liang, J. A new class of homogeneous nucleic acid probes based on specific displacement hybridization. Nucleic Acids Res. 30, e5 (2002).
Xiao, Y. et al. Fluorescence detection of single-nucleotide polymorphisms with a single, self-complementary, triple-stem DNA probe, Angew. Chem. Int. Ed. 48, 4354–4358 (2009).
Zhang, D. Y., Chen, S. X. & Yin, P. Thermodynamic optimization of nucleic acid hybridization specificity. Nature Chem. 4, 208–214 (2012).
Petersen, M. & Wengel, J. LNA a versatile tool for therapeutics and genomics. Trends Biotechnol. 21, 74–81, (2003).
He, G., Rapireddy, S., Bahal, R., Sahu, B. & Ly, D. H. Strand invasion of extended, mixed-sequence B-DNA by gamma PNAs. J. Am. Chem. Soc. 131, 12088–12090 (2009).
Kierzek, E., Mathews, D. H., Ciesielska, A., Turner, D. H. & Kierzek, R. Nearest neighbor parameters for Watson–Crick complementary heteroduplexes formed between 2′-O-methyl RNA and RNA oligonucleotides. Nucleic Acids Res. 34, 3609–3614 (2006).
Chun, J. et al. Dual priming oligonucleotide system for the multiplex detection of respiratory viruses and SNP genotyping of CYP2C19 gene. Nucleic Acids Res. 35, e40 (2007).
Andreasen, D. et al. Improved microRNA quantification in total RNA from clinical samples. Methods 50, S6–S9 (2010).
Kamisango, K. et al. Quantitative detection of hepatitis B virus by transcription-mediated amplification and hybridization protection assay. J. Clin. Microbiol. 37, 310–314 (1999).
Itzkovitz, S. & van Oudenaarden, A. Validating transcripts with probes and imaging technology. Nature Methods 8, S12–S19 (2011).
Richter, A. et al. A multisite blinded study for the detection of BRAF mutations in formalin-fixed, paraffin-embedded malignant melanoma. Sci. Rep. 3, 1659 (2013).
Zhang, D. Y. & Winfree, E. Control of DNA strand displacement kinetics using toehold exchange. J. Am. Chem. Soc. 131, 17303–17314.
Reynaldo, L. P., Vologodskii, A. V., Neri, B. P. & Lyamichev, V. I. The kinetics of oligonucleotide replacements. J. Mol. Biol. 297, 511–520 (2000).
SantaLucia, J. & Hicks, D. The thermodynamics of DNA structural motifs. Ann. Rev. Biochem. 33, 415–440 (2004).
Radding, C. M., Beattie, K. L., Holloman, W. K. & Wiegand, R. C. Uptake of homologous single-stranded fragments by superhelical DNA. J. Mol. Biol. 116, 859–839 (1977).
Zhang, D. Y., Turberfield, A. J., Yurke, B. & Winfree, E. Engineering entropy-driven reactions and networks catalyzed by DNA. Science 318, 1121–1125 (2007).
Zhang, D. Y., & Winfree, E. Robustness and modularity properties of a non-covalent DNA catalytic reaction. Nucleic Acids Res. 38, 4182–4197 (2010).
Forbes, S. A. et al. COSMIC: mining complete cancer genomes in the Catalogue of Somatic Mutations in Cancer. Nucleic Acids Res. 39, D945–D950 (2010).
Sugimoto, N. et al. Thermodynamic parameters to predict stability of RNA/DNA hybrid duplexes. Biochemistry 34, 11211–11216 (1995).
Watkins, N. et al. Thermodynamic contributions of single internal rA·dA, rC·dC, rG·dG and rU·dT mismatches in RNA/DNA duplexes. Nucleic Acids Res. 39, 1894–1902 (2011).
Eckert, K. A. & Kunkel, T. A. DNA polymerase fidelity and the polymerase chain reaction. PCR Methods Appl. 1, 17–24 (1991).
Pezza, J. A., Kucera, R. & Sun, L. Polymerase fidelity: what is it, and what does it mean for your PCR? https://www.neb.com/tools-and-resources/feature-articles/polymerase-fidelity-what-is-it-and-what-does-it-mean-for-your-pcr, accessed November 27, 2014.
Stockton, J., Ellis, J. S., Saville, M., Clewley, J. P. & Zambon, M. C. Multiplex PCR for typing and subtyping influenza and respiratory syncytial viruses. J. Clin. Microbiol. 36, 2990–2995 (1998).
de Sanjose, S. et al. Human papillomavirus genotype attribution in invasive cervical cancer: a retrospective cross-sectional worldwide study. Lancet Oncol. 11, 1048–1056 (2010).
Boehme, C. C. et al. Rapid molecular detection of tuberculosis and rifampin resistance. N. Engl. J. Med. 363, 1005–1015 (2010).
Mardis, E. R. A decade's perspective on DNA sequencing technology. Nature 470, 198–203 (2011).
Imperiale, T. F. et al. Multitarget stool DNA testing for colorectal cancer screening. N. Engl. J. Med. 370, 1287–1297 (2014).
Baker, M. Digital PCR hits its stride. Nature Methods 9, 541–544 (2012).
Holland, P. M., Abramson, R. D., Watson, R. & Gelfand, D. H. Detection of specific polymerase chain reaction product by utilizing the 5′–3′ exonuclease activity of Thermus aquaticus DNA polymerase. Proc. Natl Acad. Sci. USA 88, 7276–7280 (1991).
Saiki, R. K. et al. Primer-directed enzymatic amplification of DNA with a thermostable DNA polymerase. Science 239, 487–491 (1988).
Schmittgen, T. D. & Livak, K. J. Analyzing real-time PCR data by the comparative CT method. Nature Protocols 3, 1101–1108 (2008).
Schultz, S., Smith, D. R., Mock, J. J. & Schultz, D. A. Single-target molecule detection with nonbleaching multicolor optical immunolabels. Proc. Natl Acad. Sci. USA 97, 996–1001 (2000).
Acknowledgements
This work was funded by the Rice University Department of Bioengineering start-up fund to D.Y.Z., a John S. Dunn award to D.Y.Z. and a National Institutes of Health Grant (EB015331) to D.Y.Z.
Author information
Authors and Affiliations
Contributions
J.S.W. and D.Y.Z. conceived the project, performed the theoretical analysis, designed the experiments, conducted the experiments, analysed the data and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
Two patents are pending on this work. J.S.W. and D.Y.Z. are equity holders of Searna Technologies, a startup aiming to commercialize the presented technology.
Supplementary information
Supplementary information
Supplementary information (PDF 2641 kb)
Rights and permissions
About this article
Cite this article
Wang, J., Zhang, D. Simulation-guided DNA probe design for consistently ultraspecific hybridization. Nature Chem 7, 545–553 (2015). https://doi.org/10.1038/nchem.2266
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2266
This article is cited by
-
Heteromultivalency enables enhanced detection of nucleic acid mutations
Nature Chemistry (2024)
-
A paper-based assay for the colorimetric detection of SARS-CoV-2 variants at single-nucleotide resolution
Nature Biomedical Engineering (2022)
-
Highly multiplexed rapid DNA detection with single-nucleotide specificity via convective PCR in a portable device
Nature Biomedical Engineering (2021)
-
A deep learning model for predicting next-generation sequencing depth from DNA sequence
Nature Communications (2021)
-
Context-aware seeds for read mapping
Algorithms for Molecular Biology (2020)