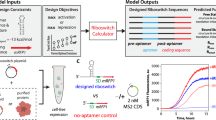
Abstract
Oligonucleotide-based receptors or aptamers can interact with small molecules, but the ability to achieve high-affinity and specificity of these interactions depends strongly on functional groups or epitopes displayed by the binding targets. Some classes of targets are particularly challenging: for example, monosaccharides have scarce functionalities and no aptamers have been reported to recognize, let alone distinguish from each other, glucose and other hexoses. Here we report aptamers that differentiate low-epitope targets such as glucose, fructose or galactose by forming ternary complexes with high-epitope organic receptors for monosaccharides. In a follow-up example, we expand this method to isolate high-affinity oligonucleotides against aromatic amino acids complexed in situ with a nonspecific organometallic receptor. The method is general and enables broad clinical use of aptamers for the detection of small molecules in mix-and-measure assays, as demonstrated by monitoring postprandial waves of phenylalanine in human subjects.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Keefe, A. D., Pai, S. & Ellington, A. D. Aptamers as therapeutics. Nature Rev. Drug Discov. 9, 537–550 (2010).
Cho, E. J., Lee, J-W. & Ellington, A. D. Applications of aptamers as sensors. Annu. Rev. Anal. Chem. 2, 241–264 (2009).
Gold, L. et al. Aptamer-based multiplexed proteomic technology for biomarker discovery. PLoS ONE 5, e15004 (2010).
Kimoto, M., Yamashige, R., Matsunaga, K-I., Yokoyama, S. & Hirao, I. Generation of high-affinity DNA aptamers using an expanded genetic alphabet. Nature Biotechnol. 31, 453–457 (2013).
McKeague, M. & Derosa, M. C. Challenges and opportunities for small molecule aptamer development. J. Nucleic Acids 748913 (2012).
Imaizumi, Y. et al. Efficacy of base-modification on target binding of small molecule DNA aptamers. J. Am. Chem. Soc. 135, 9412–9419 (2013).
Manimala, J. C., Wiskur, S. L., Ellington, A. D. & Anslyn, E. V. Tuning the specificity of a synthetic receptor using a selected nucleic acid receptor. J. Am. Chem. Soc. 126, 16515–16519 (2004).
Klonoff, D. C. The need for separate performance goals for glucose sensors in the hypoglycemic, normoglycemic, and hyperglycemic ranges. Diabetes Care 27, 834–836 (2004).
James, T. D., Samankumara Sandanayake, K. R. A. & Shinkai, S. A glucose-selective molecular fluorescence sensor. Angew. Chem. Int. Ed. 33, 2207–2209 (1994).
Larkin, J. D., Frimat, K. A., Fyles, T. M., Flower, S. E. & James, T. D. Boronic acid based photoinduced electron transfer (PET) fluorescence sensors for saccharides. New J. Chem. 34, 2922–2931 (2010).
Yang, K-A., Pei, R., Stefanovic, D. & Stojanovic, M. N. Optimizing cross-reactivity with evolutionary search for sensors. J. Am. Chem. Soc. 134, 1642–1647 (2012).
Nutiu, R. & Li, Y. In vitro selection of structure-switching signaling aptamers. Angew. Chem. Int. Ed. 44, 1061–1065 (2005).
Rajendran, M. & Ellington, A. D. Selection of fluorescent aptamer beacons that light up in the presence of zinc. Anal. Bioanal. Chem. 390, 1067–1075 (2008).
Blau, N., van Spronsen, F. J. & Levy, H. L. Phenylketonuria. Lancet 376, 1417–1427 (2010).
Hanley, W. B. et al. ‘Hypotyrosinemia’ in phenylketonuria. Mol. Genet. Metab. 69, 286–294 (2000).
Snedden, W., Mellor, C. S. & Martin, J. R. Familial hypertryptophanemia, tryptophanuria and indoleketonuria. Clin. Chim. Acta 131, 247–256 (1983).
Yarus, M., Widmann, J. J. & Knight, R. RNA–amino acid binding: a stereochemical era for the genetic code. J. Mol. Evol. 69, 406–429 (2009).
Illangasekare, M. & Yarus, M. Phenylalanine-binding RNAs and genetic code evolution. J. Mol. Evol. 54, 298–311 (2002).
Manironi, C., Scerch, C., Fruscoloni, P. & Tocchini-Valentini, G. P. Molecular recognition of amino acids by RNA aptamers: the evolution into an L-tyrosine binder of a dopamine-binding RNA motif. RNA 6, 520–527 (2000).
Yang, X. et al. Characterization and application of a DNA aptamer binding to L-tryptophan. Analyst 136, 577–585 (2011).
Buryak, A. & Severin, K. A chemosensor array for the colorimetric identification of 20 natural amino acids. J. Am. Chem. Soc. 127, 3700–3701 (2005).
Rampini, S., Anders, P. W., Curtius, H. C. & Marthaler, T. Detection of heterozygotes for phenylketonuria by column chromatography and discriminatory analysis. Pediatr. Res. 3, 287–297 (1969).
Taylor, S. & Stojanovic, M. N. Is there a future for DNA-based molecular devices in diabetes management? J. Diabetes Sci. Technol. 1, 440–444 (2007).
Pinheiro, A. V., Han, D., Shih, W. M. & Yan, H. Challenges and opportunities for structural DNA nanotechnology. Nature Nanotech. 6, 763–772 (2011).
Acknowledgements
We are grateful to the National Science Foundation (CBET-1033288 and CBET-1026592), National Institutes of Health (RGM104960) and the Juvenile Diabetes Research Foundation (Innovative Program) for funding this research. We thank J. Loeb for help in correcting initial drafts of our manuscript.
Author information
Authors and Affiliations
Contributions
M.N.S. proposed and led the project. K-A.Y. isolated the aptamers listed in this report. S.P. and S.T. performed the synthetic and characterization work on the organic receptors. T.S.W. provided the clinical context and organized self-experiments, with B.K. providing technical support in these. M.B., M.H., P.P. and D.M.K. performed earlier experiments on glucose aptamers that eventually led to the development of the method described here. All co-authors participated in the design of their own experiments and analysis of data. M.N.S., T.S.W., K-A.Y. and S.T. wrote the manuscript, with the exception of synthetic procedures written by S.P.
Corresponding authors
Ethics declarations
Competing interests
The authors declare competing financial interests in the form of patent application(s).
Supplementary information
Supplementary information
Supplementary information (PDF 1396 kb)
Rights and permissions
About this article
Cite this article
Yang, KA., Barbu, M., Halim, M. et al. Recognition and sensing of low-epitope targets via ternary complexes with oligonucleotides and synthetic receptors. Nature Chem 6, 1003–1008 (2014). https://doi.org/10.1038/nchem.2058
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2058
This article is cited by
-
Quantitative Methods for Metabolite Analysis in Metabolic Engineering
Biotechnology and Bioprocess Engineering (2023)
-
Sensing serotonin secreted from human serotonergic neurons using aptamer-modified nanopipettes
Molecular Psychiatry (2021)
-
Graphene/aptamer probes for small molecule detection: from in vitro test to in situ imaging
Microchimica Acta (2020)
-
Cancer-derived exosomes loaded with ultrathin palladium nanosheets for targeted bioorthogonal catalysis
Nature Catalysis (2019)
-
An electrochemical aptamer-based sensor for the rapid and convenient measurement of l-tryptophan
Analytical and Bioanalytical Chemistry (2019)