Abstract
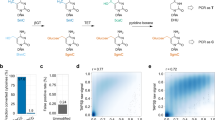
Recently, the cytosine modifications 5-hydroxymethylcytosine (5hmC) and 5-formylcytosine (5fC) were found to exist in the genomic deoxyribonucleic acid (DNA) of a wide range of mammalian cell types. It is now important to understand their role in normal biological function and disease. Here we introduce reduced bisulfite sequencing (redBS-Seq), a quantitative method to decode 5fC in DNA at single-base resolution, based on a selective chemical reduction of 5fC to 5hmC followed by bisulfite treatment. After extensive validation on synthetic and genomic DNA, we combined redBS-Seq and oxidative bisulfite sequencing (oxBS-Seq) to generate the first combined genomic map of 5-methylcytosine, 5hmC and 5fC in mouse embryonic stem cells. Our experiments revealed that in certain genomic locations 5fC is present at comparable levels to 5hmC and 5mC. The combination of these chemical methods can quantify and precisely map these three cytosine derivatives in the genome and will help provide insights into their function.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
06 May 2014
The raw data including sequencing files (fastq format), processed data including counts of modified bases for each library preparation (reduced, oxidative and simple bisulfite) and the estimation of significant sites as reported in the paper, together with some explanation and a summary of the experimental and analytical protocols, are available here: http://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE56572
References
Ndlovu, M. N., Denis, H. & Fuks, F. Exposing the DNA methylome iceberg. Trends Biochem. Sci. 36, 381–387 (2011).
Kriaucionis, S. & Heintz, N. The nuclear DNA base 5-hydroxymethylcytosine is present in Purkinje neurons and the brain. Science 324, 929–930 (2009).
Tahiliani, M. et al. Conversion of 5-methylcytosine to 5-hydroxymethylcytosine in mammalian DNA by MLL partner TET1. Science 324, 930–935 (2009).
Pfaffeneder, T. et al. The discovery of 5-formylcytosine in embryonic stem cell DNA. Angew. Chem. Int. Ed. 50, 7008–7012 (2011).
Ito, S. et al. Tet proteins can convert 5-methylcytosine to 5-formylcytosine and 5-carboxylcytosine. Science 333, 1300–1303 (2011).
Liu, S. et al. Quantitative assessment of Tet-induced oxidation products of 5-methylcytosine in cellular and tissue DNA. Nucleic Acids Res. 41, 6421–6429 (2013).
Hashimoto, H., Hong, S., Bhagwat, A. S., Zhang, X. & Cheng, X. Excision of 5-hydroxymethyluracil and 5-carboxylcytosine by the thymine DNA glycosylase domain: its structural basis and implications for active DNA demethylation. Nucleic Acids Res. 40, 10203–10214 (2012).
Maiti, A. & Drohat, A. C. Thymine DNA glycosylase can rapidly excise 5-formylcytosine and 5-carboxylcytosine: potential implications for active demethylation of CpG sites. J. Biol. Chem. 286, 35334–35338 (2011).
Shen, L. et al. Genome-wide analysis reveals TET- and TDG-dependent 5-methylcytosine oxidation dynamics. Cell 153, 692–706 (2013).
Mellen, M., Ayata, P., Dewell, S., Kriaucionis, S. & Heintz, N. MeCP2 binds to 5hmC enriched within active genes and accessible chromatin in the nervous system. Cell 151, 1417–1430 (2012).
Spruijt, C. G. et al. Dynamic readers for 5-(hydroxy)methylcytosine and its oxidized derivatives. Cell 152, 1146–1159 (2013).
Shen, L. et al. Genome-wide analysis reveals TET- and TDG-dependent 5-methylcytosine oxidation dynamics. Cell 153, 692–706 (2013).
Raiber, E. A. et al. Genome-wide distribution of 5-formylcytosine in embryonic stem cells is associated with transcription and depends on thymine DNA glycosylase. Genome Biol. 13, R69 (2012).
Song, C. X. et al. Genome-wide profiling of 5-formylcytosine reveals its roles in epigenetic priming. Cell 153, 678–691 (2013).
Hayatsu, H., Wataya, Y., Kai, K. & Iida, S. Reaction of sodium bisulfite with uracil, cytosine, and their derivatives. Biochemistry 9, 2858–2865 (1970).
Shapiro, R., Servis, R. E. & Welcher, M. Reactions of uracil and cytosine derivatives with sodium bisulfite. A specific deamination method. J. Am. Chem. Soc. 92, 422–428 (1970).
Frommer, M. et al. A genomic sequencing protocol that yields a positive display of 5-methylcytosine residues in individual DNA strands. Proc. Natl Acad. Sci. USA 89, 1827–1831 (1992).
Huang, Y. et al. The behaviour of 5-hydroxymethylcytosine in bisulfite sequencing. PLoS One 5, e8888 (2010).
Booth, M. J. et al. Quantitative sequencing of 5-methylcytosine and 5-hydroxymethylcytosine at single-base resolution. Science 336, 934–937 (2012).
Booth, M. J. et al. Oxidative bisulfite sequencing of 5-methylcytosine and 5-hydroxymethylcytosine. Nature Protoc. 8, 1841–1851 (2013).
Yu, M. et al. Base-resolution analysis of 5-hydroxymethylcytosine in the mammalian genome. Cell 149, 1368–1380 (2012).
Oakeley, E. J., Schmitt, F. & Jost, J. P. Quantification of 5-methylcytosine in DNA by the chloroacetaldehyde reaction. Biotechniques 27, 744–766 (1999).
Yu, M. et al. Tet-assisted bisulfite sequencing of 5-hydroxymethylcytosine. Nature Protocols 7, 2159–2170 (2012).
Meissner, A. et al. Genome-scale DNA methylation maps of pluripotent and differentiated cells. Nature 454, 766–770 (2008).
Bird, A. P. Use of restriction enzymes to study eukaryotic DNA methylation: II. The symmetry of methylated sites supports semi-conservative copying of the methylation pattern. J. Mol. Biol. 118, 49–60 (1978).
Acknowledgements
We thank the Biotechnology and Biological Sciences Research Council for a studentship to M.J.B. and Cancer Research UK for a studentship to M.B. S.B. is a Senior Investigator of The Wellcome Trust and the Balasubramanian group is core-funded by Cancer Research UK.
Author information
Authors and Affiliations
Contributions
M.J.B. and S.B. co-invented the redBS-Seq method. M.J.B. and S.B. conceived the experiments with contributions from all authors. M.J.B. and M.B. performed experimental work. G.M. and D.B. performed bioinformatics analysis. M.J.B. and S.B. wrote the manuscript with contributions from all the authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 1328 kb)
Rights and permissions
About this article
Cite this article
Booth, M., Marsico, G., Bachman, M. et al. Quantitative sequencing of 5-formylcytosine in DNA at single-base resolution. Nature Chem 6, 435–440 (2014). https://doi.org/10.1038/nchem.1893
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.1893
This article is cited by
-
Bisulfite-free mapping of DNA cytosine modifications: challenges and perspectives
Science China Chemistry (2023)
-
Modular affinity-labeling of the cytosine demethylation base elements in DNA
Scientific Reports (2020)