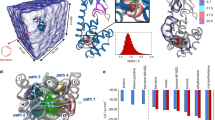
Abstract
Simulations can provide tremendous insight into the atomistic details of biological mechanisms, but micro- to millisecond timescales are historically only accessible on dedicated supercomputers. We demonstrate that cloud computing is a viable alternative that brings long-timescale processes within reach of a broader community. We used Google's Exacycle cloud-computing platform to simulate two milliseconds of dynamics of a major drug target, the G-protein-coupled receptor β2AR. Markov state models aggregate independent simulations into a single statistical model that is validated by previous computational and experimental results. Moreover, our models provide an atomistic description of the activation of a G-protein-coupled receptor and reveal multiple activation pathways. Agonists and inverse agonists interact differentially with these pathways, with profound implications for drug design.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
24 July 2015
In the version of this Article originally published, Figure 4 displayed incorrectly drawn chemical structures for five of the ligands. The correct structures were, however, used in the calculations. The hemiaminal group previously depicted in compounds 2–4 should have been a β-amino alcohol, compound 7 contained an extra benzylic carbon and compound 8 had an extra ring. The corresponding PubChem CID numbers for the correct ligands are as follows. Agonists: 1, 19044758; 2, 44216210; 3, 44209282; 4, 44213610. Antagonists: 5, 15020513; 6, 19823514; 7, 44209768; 8, 44209764. These drawing errors have now been corrected in the online versions of this Article. Additionally, the 'Inverse agonist' label at the top of Fig. 4b has been changed to 'Antagonist' as this was the original designation for this set of the GPCR ligand database used for docking (E. A. Gatica and C. N. Cavasotto, J. Chem. Inf. Model. 52, 1–6; 2012). Some ligands, particularly carazolol used in this study, may have inverse agonist activity. For all calculations, functional groups were protonated according to pH = 7. Stereochemistry is not depicted in the figure because stereoisomer activity for these compounds has not been elucidated. The structures in Figure 4 are each representative of many ligands that define a 3D chemotype and share a similar binding pose in protein conformations with similar progress scores. Stereoisomers were enumerated for up to four chiral centers and docked. The isomer with the highest score, or approximated binding affinity, was selected for a given protein conformation. Different protein conformations score isomers differently, and protein conformations with the same progress score may select different isomers of the same compound. Further experiments on the known agonist and antagonist ligands would be needed in order to determine the activities of stereoisomers, as has been done for albuterol and fenoterol (R. Seifert and S. Dove, Mol. Pharmacol. 75,13–18; 2009).
References
Cherezov, V. et al. High-resolution crystal structure of an engineered human β2-adrenergic G protein-coupled receptor. Science 318, 1258–1265 (2007).
Rasmussen, S. G. F. et al. Structure of a nanobody-stabilized active state of the β2 adrenoceptor. Nature 469, 175–180 (2011).
Rasmussen, S. G. F. et al. Crystal structure of the β2 adrenergic receptor–Gs protein complex. Nature 477, 549–555 (2011).
Dror, R. O. et al. Identification of two distinct inactive conformations of the β2-adrenergic receptor reconciles structural and biochemical observations. Proc. Natl Acad. Sci. USA 106, 4689–4694 (2009).
Dror, R. O. et al. Activation mechanism of the β2-adrenergic receptor. Proc. Natl Acad. Sci. USA 108, 18684–18689 (2011).
Vanni, S., Neri, M., Tavernelli, I. & Rothlisberger, U. Predicting novel binding modes of agonists to β adrenergic receptors using all-atom molecular dynamics simulations. PLoS Comput. Biol. 7, e1001053 (2011).
Ivetac, A. & McCammon, J. A. Mapping the druggable allosteric space of G-protein coupled receptors: a fragment-based molecular dynamics approach. Chem. Biol. Drug Des. 76, 201–217 (2010).
Nygaard, R. et al. The dynamic process of β2-adrenergic receptor activation. Cell 152, 532–542 (2013).
Shaw, D. E. et al. Anton, a special-purpose machine for molecular dynamics simulation. Commun. ACM 51, 91–97 (2008).
Shirts, M. & Pande, V. S. Screen savers of the world unite! Science 290, 1903–1904 (2000).
Hellerstein, J. L., Kohlhoff, K. J. & Konerding, D. E. Science in the cloud: accelerating discovery in the 21st century. IEEE Internet Comput. 16, 64–68 (2012).
Bowman, G. R., Huang, X. & Pande, V. S. Using generalized ensemble simulations and Markov state models to identify conformational states. Methods 49, 197–201 (2009).
Senne, M., Trendelkamp-Schroer, B., Mey, A. S. J. S., Schütte, C. & Noé, F. EMMA: a software package for Markov model building and analysis. J. Chem. Theory Comput. 8, 2223–2238 (2012).
Noé, F. & Fischer, S. Transition networks for modeling the kinetics of conformational change in macromolecules. Curr. Opin Struct. Biol. 18, 154–162 (2008).
Bowman, G. R. & Geissler, P. L. Equilibrium fluctuations of a single folded protein reveal a multitude of potential cryptic allosteric sites. Proc. Natl Acad. Sci. USA 109, 11681–11686 (2012).
Vanden-Eijnden, W. E, E. Transition-path theory and path-finding algorithms for the study of rare events. Annu. Rev. Phys. Chem. 61, 391–420 (2010).
Metzner, P., Schütte, C. & Vanden-Eijnden, E. Transition path theory for Markov jump processes. Mult. Mod. Sim. 7, 1192–1219 (2009).
Noé, F., Schütte, C., Vanden-Eijnden, E., Reich, L. & Weikl, T. R. Constructing the equilibrium ensemble of folding pathways from short off-equilibrium simulations. Proc. Natl Acad. Sci. USA 106, 19011–19016 (2009).
Kofuku, Y. et al. Efficacy of the β2-adrenergic receptor is determined by conformational equilibrium in the transmembrane region. Nature Commun. 3, 1045 (2012).
Strader, C. D. et al. Conserved aspartic acid residues 79 and 113 of the beta-adrenergic receptor have different roles in receptor function. J. Biol. Chem. 263, 10267–10271 (1988).
Liapakis, G. et al. The forgotten serine: a critical role for Ser-2035.42 in ligand binding to and activation of the β2 adrenergic receptor. J. Biol. Chem. 275, 37779–37788 (2000).
Gatica, E. A. & Cavasotto, C. N. Ligand and decoy sets for docking to G protein-coupled receptors. J. Chem. Inf. Model 52, 1–6 (2012).
Jain, A. N. Surflex-Dock 2.1: robust performance from ligand energetic modeling, ring flexibility, and knowledge-based search. J. Comput. Aided Mol. Des. 21, 281–306 (2007).
Spitzer, R. & Jain, A. N. Surflex-Dock: docking benchmarks and real-world application. J Comput. Aided Mol. Des. 26, 687–699 (2012).
Shoichet, B. K. Virtual screening of chemical libraries. Nature 432, 862–865 (2004).
Lane, T. J., Shukla, D., Beauchamp, K. A. & Pande, V. S. To milliseconds and beyond: challenges in the simulation of protein folding. Curr. Opin. Struct. Biol. 23, 58–65 (2013).
Shoichet, B. K. & Kobilka, B. K. Structure-based drug screening for G-protein-coupled receptors. Trends Pharmacol. Sci. 33, 268–272 (2012).
Schames, J. R. et al. Discovery of a novel binding trench in HIV integrase. J. Med. Chem. 47, 1879–1881 (2004).
Lomize, M. A., Lomize, A. L., Pogozheva, I. D. & Mosberg, H. I. OPM: orientations of proteins in membranes database. Bioinformatics 22, 623–625 (2006).
Rosenbaum, D. M. et al. Structure and function of an irreversible agonist–β2 adrenoceptor complex. Nature 469, 236–240 (2011).
Hildebrand, P. W. et al. SuperLooper – a prediction server for the modeling of loops in globular and membrane proteins. Nucleic Acids Res. 37, W571–W574 (2009).
Ballesteros, J. A. et al. Activation of the β2-adrenergic receptor involves disruption of an ionic lock between the cytoplasmic ends of transmembrane segments 3 and 6. J. Biol. Chem. 276, 29171–29177 (2001).
Ghanouni, P. et al. The effect of pH on β2 adrenoceptor function: evidence for protonation-dependent activation. J. Biol. Chem. 275, 3121–3127 (2000).
Hess, B., Kutzner, C., van der Spoel, D. & Lindahl, E. GROMACS 4: algorithms for highly efficient, load-balanced, and scalable molecular simulation. J. Chem. Theory Comput. 4, 435–447 (2008).
Voelz, V. A., Bowman, G. R., Beauchamp, K. A. & Pande, V. S. Molecular simulation of ab initio protein folding for a millisecond folder NTL9(1−39). J. Am. Chem. Soc. 132, 1526–1528 (2010).
Beauchamp, K. A. et al. MSMBuilder2: modeling conformational dynamics on the picosecond to millisecond scale. J. Chem. Theory Comput. 7, 3412–3419 (2011).
Irwin, J. J., Sterling, T., Mysinger, M. M., Bolstad, E. S. & Coleman, R. G. ZINC: a free tool to discover chemistry for biology. J. Chem. Inf. Model. 52, 1757–1768 (2012).
Hawkins, P. C. D., Skillman, A. G., Warren, G. L., Ellingson, B. A. & Stahl, M. T. Conformer generation with OMEGA: algorithm and validation using high quality structures from the Protein Databank and Cambridge Structural Database. J. Chem. Inf. Model. 50, 572–584 (2010).
ROCS, version 3.1.2 (OpenEye Scientific Software, Santa Fe, New Mexico, 2011).
Chambers, C. et al. in Proceedings of the 2010 ACM SIGPLAN Conference on Programming Language Design and Implementation 363–375 (ACM, 2010).
Dean, J. & Ghemawat, S. MapReduce: simplified data processing on large clusters. Commun. ACM 51, 107–113 (2008).
Chang, F. et al. Bigtable: a distributed storage system for structured data. ACM Trans. Comput. Syst. 26, 4:1–4:26 (2008).
Melnik, S. et al. Dremel: interactive analysis of web-scale datasets. Proc. VLDB Endow. 3, 330–339 (2010).
Humphrey, W., Dalke, A. & Schulten, K. VMD: visual molecular dynamics. J. Mol. Graph. 14, 33–38 (1996).
Berezhkovskii, A., Hummer, G. & Szabo, A. Reactive flux and folding pathways in network models of coarse-grained protein dynamics. J. Chem. Phys. 130, 205102 (2009).
Acknowledgements
We are grateful to M. Stumpe for his contributions to setting up the initial molecular systems and P. Kasson and J. Hellerstein for helpful advice and support. This work was funded in part by a 450M CPU core-hour donation by Google Inc. through the Exacycle eScience program, the Simbios NIH National Center on Biocomputing through the NIH Roadmap for Medical Research Grant U54 GM07297 and a Stanford School of Medicine Dean's Fellowship (K.J.K.). We also thank the users of the Folding@home distributed-computing project for donating compute time for some preliminary simulations that ensured a stable production run. Additional computations for docking and chemotype clustering were performed on the Blue Waters supercomputer at the National Center for Supercomputing Applications at the University of Illinois at Urbana-Champaign.
Author information
Authors and Affiliations
Contributions
K.J.K., D.S. and M.L. contributed equally to this work. V.S.P., R.B.A, D.E.K. and D.B. conceived, and V.S.P. and R.B.A. supervised the project. K.J.K. and D.E.K. developed the platform for running MD simulations with Gromacs on Google Exacycle. K.J.K. set up the simulation systems. G.R.B helped with initial analysis. K.J.K. performed simulations on Google Exacycle and processed data on Google's production infrastructure. K.J.K. and G.R.B. performed preliminary simulations on Folding@home. D.S. and M.L. performed additional simulations. D.S., M.L and K.J.K. analysed the data and built MSMs. M.L. performed small-molecule docking calculations. D.S., M.L. and K.J.K. co-wrote the manuscript with inputs from G.R.B, R.B.A and V.S.P. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 4482 kb)
Rights and permissions
About this article
Cite this article
Kohlhoff, K., Shukla, D., Lawrenz, M. et al. Cloud-based simulations on Google Exacycle reveal ligand modulation of GPCR activation pathways. Nature Chem 6, 15–21 (2014). https://doi.org/10.1038/nchem.1821
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.1821
This article is cited by
-
Distinct activation mechanisms regulate subtype selectivity of Cannabinoid receptors
Communications Biology (2023)
-
An overview of the SAMPL8 host–guest binding challenge
Journal of Computer-Aided Molecular Design (2022)
-
Biomolecular modeling thrives in the age of technology
Nature Computational Science (2021)
-
A kinetic ensemble of the Alzheimer’s Aβ peptide
Nature Computational Science (2021)
-
Temperature-dependent kinetic pathways of heterogeneous ice nucleation competing between classical and non-classical nucleation
Nature Communications (2021)