Abstract
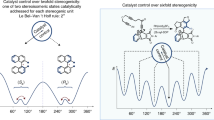
Although asymmetric catalysis is universally dependent on spatial interactions to impart specific chirality on a given substrate, examination of steric effects in these catalytic systems remains empirical. Previous efforts by our group and others have seen correlation between steric parameters developed by Charton and simple substituents in both substrate and ligand; however, more complex substituents were not found to be correlative. Here, we review and compare the steric parameters common in quantitative structure activity relationships (QSAR), a common method for pharmaceutical function optimization, and how they might be applied in asymmetric catalysis, as the two fields are undeniably similar. We re-evaluate steric/enantioselection relationships, which we previously analysed with Charton steric parameters, using the more sophisticated Sterimol parameters developed by Verloop and co-workers in a QSAR context. Use of these Sterimol parameters led to strong correlations in numerous processes where Charton parameters had previously failed. Sterimol parameterization also allows for greater mechanistic insight into the key elements of asymmetric induction within these systems.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Knowles, W. S. Asymmetric hydrogenation. Acc. Chem. Res. 16, 106–112 (1983).
Cheong, P. H.-Y., Legault, C. Y., Um, J. M., Çelebi-Ölçüm, N. & Houk, K. N. Quantum mechanical investigations of organocatalysis: mechanisms, reactivities, and selectivities. Chem. Rev. 111, 5042–5137 (2011).
Walsh, P. J. & Kozlowski, M. C. Fundamentals of Asymmetric Catalysis (University Science Books, 2009).
Houk, K. N. & Cheong, P. H.-Y. Computational prediction of small-molecule catalysts. Nature 455, 309–313 (2008).
Denmark, S. E., Gould, N. D. & Wolf, L. M. A systematic investigation of quaternary ammonium ions as asymmetric phase-transfer catalysts. Application of quantitative structure activity/selectivity relationships. J. Org. Chem. 76, 4337–4357 (2011).
Denmark, S. E., Gould, N. D. & Wolf, L. M. A systematic investigation of quaternary ammonium ions as asymmetric phase-transfer catalysts. Synthesis of catalyst libraries and evaluation of catalyst activity. J. Org. Chem. 76, 4260–4336 (2011).
Maldonado, A. G. & Rothenberg, G. Predictive modeling in homogeneous catalysis: a tutorial. Chem. Soc. Rev. 39, 1891–1902 (2010).
Ianni, J. C., Annamalai, V., Phuan, P.-W., Panda, M. & Kozlowski, M. C. A priori theoretical prediction of selectivity in asymmetric catalysis: design of chiral catalysts by using quantum molecular interaction fields. Angew. Chem. Int. Ed. 45, 5502–5505 (2006).
Kozlowski, M. C. & Panda, M. Computer-aided design of chiral ligands: Part I. Database search methods to identify chiral ligand types for asymmetric reactions. J. Mol. Graphics Modell. 20, 399–409 (2002).
Kozlowski, M. C. & Panda, M. Computer-aided design of chiral ligands. Part 2. Functionality mapping as a method to identify stereocontrol elements for asymmetric reactions. J. Org. Chem. 68, 2061–2076 (2003).
Kozlowski, M. C., Waters, S. P., Skudlarek, J. W. & Evans, C. A. Computer-aided design of chiral ligands. Part III. A novel ligand for asymmetric allylation designed using computational techniques. Org. Lett. 4, 4391–4393 (2002).
Lipkowitz, K. B. & Kozlowski, M. C. Understanding stereoinduction in catalysis via computer: new tools for asymmetric synthesis. Synlett. 2003, 1547–1565 (2003).
Norrby, P.-O., Rasmussen, T., Haller, J., Strassner, T. & Houk, K. N. Rationalizing the stereoselectivity of osmium tetroxide asymmetric dihydroxylations with transition state modeling using quantum mechanics-guided molecular mechanics. J. Am. Chem. Soc. 121, 10186–10192 (1999).
Oslob, J. D., Aakermark, B., Helquist, P. & Norrby, P.-O. Steric influences on the selectivity in palladium-catalyzed allylation. Organometallics 16, 3015–3021 (1997).
Brown, J. M. & Deeth, R. J. Is enantioselectivity predictable in asymmetric catalysis? Angew. Chem. Int. Ed. 48, 4476–4479 (2009).
Knowles, R. R. & Jacobsen, E. N. Attractive noncovalent interactions in asymmetric catalysis: links between enzymes and small molecule catalysts. Proc. Natl Acad. Sci. USA 107, 20678–20685 (2010).
Uyeda, C. & Jacobsen, E. N. Transition-state charge stabilization through multiple non-covalent interactions in the guanidinium-catalyzed enantioselective Claisen rearrangement. J. Am. Chem. Soc. 133, 5062–5075 (2011).
Zuend, S. J. & Jacobsen, E. N. Mechanism of amido-thiourea catalyzed enantioselective imine hydrocyanation: transition state stabilization via multiple non-covalent interactions. J. Am. Chem. Soc. 131, 15358–15374 (2009).
Jonsson, S., Odille, F. G. J., Norrby, P.-O. & Warnmark, K. Modulation of the reactivity, stability and substrate- and enantioselectivity of an epoxidation catalyst by noncovalent dynamic attachment of a receptor functionality—aspects on the mechanism of the Jacobsen–Katsuki epoxidation applied to a supramolecular system. Org. Biomol. Chem. 4, 1927–1948 (2006).
Lipkowitz, K. B., D'Hue, C. A., Sakamoto, T. & Stack, J. N. Stereocartography: a computational mapping technique that can locate regions of maximum stereoinduction around chiral catalysts. J. Am. Chem. Soc. 124, 14255–14267 (2002).
Lipkowitz, K. B., Sakamoto, T. & Stack, J. Using stereocartography for predicting efficacy of stereoinduction by chiral catalysts. Chirality 15, 759–765 (2003).
Jacobsen, E. N., Pfaltz, A. & Yamamoto, H. Comprehensive Asymmetric Catalysis I–III Vol. 1 (Springer, 1999).
Miller, J. J. & Sigman, M. S. Quantitatively correlating the effect of ligand–substituent size in asymmetric catalysis using linear free energy relationships. Angew. Chem. Int. Ed. 47, 771–774 (2008).
Miller, J. J. & Sigman, M. S. Design and synthesis of modular oxazoline ligands for the enantioselective chromium-catalyzed addition of allyl bromide to ketones. J. Am. Chem. Soc. 129, 2752–2753 (2007).
Miller, J. J., Rajaram, S., Pfaffenroth, C. & Sigman, M. S. Synthesis of amine functionalized oxazolines with applications in asymmetric catalysis. Tetrahedron 65, 3110–3119 (2009).
Quintard, A. & Alexakis, A. 1,2-Sulfone rearrangement in organocatalytic reactions. Org. Biomol. Chem. 9, 1407–1418 (2011).
Quintard, A., Alexakis, A. & Mazet, C. Access to high levels of molecular complexity by one-pot iridium/enamine asymmetric catalysis. Angew. Chem. Int. Ed. 50, 2354–2358 (2011).
Mantilli, L., Gérard, D., Torche, S., Besnard, C. & Mazet, C. Improved catalysts for the iridium-catalyzed asymmetric isomerization of primary allylic alcohols based on charton analysis. Chem. Eur. J. 16, 12736–12745 (2010).
Hansch, C. & Leo, A. Exploring QSAR: Fundamentals and Applications in Chemistry and Biology (American Chemical Society, 1995).
Verloop, A. in Drug Design Vol. III (ed. Ariens, E. J.) 133 (Academic Press, 1976).
Winstein, S. & Holness, N. J. Neighboring varbon and hydrogen. XIX. t-Butylcyclohexyl derivatives. Quantitative conformational analysis. J. Am. Chem. Soc. 77, 5562–5578 (1955).
Bott, G., Field, L. D. & Sternhell, S. Steric effects. A study of a rationally designed system. J. Am. Chem. Soc. 102, 5618–5626 (1980).
Adams, R. & Yuan, H. C. The stereochemistry of diphenyls and analogous compounds. Chem. Rev. 12, 261–338 (1933).
Niksch, T., Görls, H. & Weigand, W. The extension of the solid-angle concept to bidentate ligands. Eur. J. Inorg. Chem. 95–105 (2010).
Tolman, C. A. Steric effects of phosphorus ligands in organometallic chemistry and homogeneous catalysis. Chem. Rev. 77, 313–348 (1977).
Fey, N. et al. Computational descriptors for chelating P,P- and P,N-donor ligands 1. Organometallics 27, 1372–1383 (2008).
Jover, J. S. et al. Expansion of the ligand knowledge base for monodentate P-donor ligands (LKB-P)†. Organometallics 29, 6245–6258 (2010).
Taft, R. W. Jr. Polar and steric substituent constants for aliphatic and o-benzoate groups from rates of esterification and hydrolysis of esters. J. Am. Chem. Soc. 74, 3120–3128 (1952).
Taft, R. W. Jr. Linear steric energy relationships. J. Am. Chem. Soc. 75, 4538–4539 (1953).
Hammett, L. P. Some relations between reaction rates and equilibrium constants. Chem. Rev. 17, 125–136 (1935).
Fujita, T., Takayama, C. & Nakajima, M. Nature and composition of Taft–Hancock steric constants. J. Org. Chem. 38, 1623–1630 (1973).
Hancock, C. K., Meyers, E. A. & Yager, B. J. Quantitative separation of hyperconjugation effects from steric substituent constants. J. Am. Chem. Soc. 83, 4211–4213 (1961).
MacPhee, J. A., Panaye, A. & Dubois, J. E. Steric effects. 4. Multiparameter correlation models. Geometrical and proximity site effects for carboxylic acid esterification and related reactions. J. Org. Chem. 45, 1164–1166 (1980).
Sotomatsu, T. & Fujita, T. The steric effect of ortho substituents on the acidic hydrolysis of benzamides. J. Org. Chem. 54, 4443–4448 (1989).
Charton, M. Steric effects. I. Esterification and acid-catalyzed hydrolysis of esters. J. Am. Chem. Soc. 97, 1552–1556 (1975).
Charton, M. Steric effects. II. Base-catalyzed ester hydrolysis. J. Am. Chem. Soc. 97, 3691–3693 (1975).
Charton, M. Steric effects. 7. Additional V constants. J. Org. Chem. 41, 2217–2220 (1976).
Kutter, E. & Hansch, C. Steric parameters in drug design. Monoamine oxidase inhibitors and antihistamines. J. Med. Chem. 12, 647–652 (1969).
Verloop, A. & Tipker, J. in Biological Activity and Chemical Structure (ed. Buisman, J. A.) 63 (Elsevier, 1977).
Verloop, A. & Tipker, J. in QSAR in Drug Dosing and Toxicology (ed. Hadzi. B. & Jerman-Blazic, B.) 97 (Elsevier, 1987).
Verloop, A. in IUPAC Pesticide Chemistry Vol. 1 (ed. Miyamoto, J.) 339 (Pergamon, 1983).
Pauling, L. & Corey, R. B. Atomic coordinates and structure factors for two helical configurations of polypeptide chains. Proc. Natl Acad. Sci. USA 37, 235–240 (1951).
Pauling, L. & Corey, R. B. Configurations of polypeptide chains with favored orientations around single bonds. Proc. Natl Acad. Sci. USA 37, 729–740 (1951).
Jones, J. E. On the determination of molecular fields. I. From the variation of the viscosity of a gas with temperature. Proc. R. Soc. A 106, 441–462 (1924).
Allinger, N. L., Yuh, Y. H. & Lii, J. H. Molecular mechanics. The MM3 force field for hydrocarbons. 1. J. Am. Chem. Soc. 111, 8551–8566 (1989).
Lii, J. H. & Allinger, N. L. Molecular mechanics. The MM3 force field for hydrocarbons. 3. The van der Waals’ potentials and crystal data for aliphatic and aromatic hydrocarbons. J. Am. Chem. Soc. 111, 8576–8582 (1989).
Hammett, L. P. Physical Organic Chemistry (McGraw-Hill, 1940).
Anslyn, E. V. & Dougherty, D. A. Modern Physical Organic Chemistry (University Science Books, 2006).
Jaffé, H. H. A reexamination of the Hammett equation. Chem. Rev. 53, 191–261 (1953).
Johnson, C. D. The Hammett Equation (University Press, 1973).
Exner, O. Advances in Linear Free-Energy Relationships (Plenum, 1972).
Swain, C. G. & Lupton, E. C. Field and resonance components of substituent effects. J. Am. Chem. Soc. 90, 4328–4337 (1968).
Swain, C. G., Unger, S. H., Rosenquist, N. R. & Swain, M. S. Substituent effects on chemical reactivity. Improved evaluation of field and resonance components. J. Am. Chem. Soc. 105, 492–502 (1983).
Hansch, C., Leo, A. & Taft, R. W. A survey of Hammett substituent constants and resonance and field parameters. Chem. Rev. 91, 165–195 (1991).
Sigman, M. S. & Miller, J. J. Examination of the role of Taft-type steric parameters in asymmetric catalysis. J. Org. Chem. 74, 7633–7643 (2009).
Deming, S. N. & Morgan, S. L. Experimental Design: A Chemometric Approach, 2nd edn (Elsevier, 1993).
Gustafson, J. L., Sigman, M. S. & Miller, S. J. Linear free-energy relationship analysis of a catalytic desymmetrization reaction of a diarylmethane-bis(phenol). Org. Lett. 12, 2794–2797 (2010).
Lewis, C. A. et al. A case of remote asymmetric induction in the peptide-catalyzed desymmetrization of a bis(phenol). J. Am. Chem. Soc. 130, 16358–16365 (2008).
Harper, K. C. & Sigman, M. S. Predicting and optimizing asymmetric catalyst performance using the principles of experimental design and steric parameters. Proc. Natl Acad. Sci. USA 108, 2179–2183 (2011).
Harper, K. C. & Sigman, M. S. Three-dimensional correlation of steric and electronic free energy relationships guides asymmetric propargylation. Science 333, 1875–1878 (2011).
Palucki, M., Finney, N. S., Pospisil, P. J., Guler, M. L., Ishida, T. & Jacobsen, E. N. The mechanistic basis for electronic effects on enantioselectivity in the (salen)Mn(III)-catalyzed epoxidation reaction. J. Am. Chem. Soc. 120, 948–954 (1998).
Jensen, K. H., Webb, J. D. & Sigman, M. S. Advancing the mechanistic understanding of an enantioselective palladium-catalyzed alkene difunctionalization reaction. J. Am. Chem. Soc. 132, 17471–17482 (2010).
Acknowledgements
The authors thank Joel Harris for critical discussions on data analysis and Scott Miller for access to data and insightful discussions regarding this work. Thanks also go to Marisa Kozlowski for introducing us to Sterimol parameters. This work was supported by the National Science Foundation (CHE-0749506 and CHE-1110599).
Author information
Authors and Affiliations
Contributions
All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 1665 kb)
Rights and permissions
About this article
Cite this article
Harper, K., Bess, E. & Sigman, M. Multidimensional steric parameters in the analysis of asymmetric catalytic reactions. Nature Chem 4, 366–374 (2012). https://doi.org/10.1038/nchem.1297
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.1297
This article is cited by
-
An artificial protein modulator reprogramming neuronal protein functions
Nature Communications (2024)
-
Ultra-high-throughput mapping of the chemical space of asymmetric catalysis enables accelerated reaction discovery
Nature Communications (2023)
-
Unveiling the impact of the light source and steric factors on [2 + 2] heterocycloaddition reactions
Nature Synthesis (2022)
-
Brønsted acid-enhanced copper-catalyzed atroposelective cycloisomerization to axially chiral arylquinolizones via dearomatization of pyridine
Nature Communications (2022)
-
Automation and computer-assisted planning for chemical synthesis
Nature Reviews Methods Primers (2021)