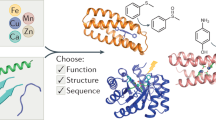
Abstract
Metal ions are an important part of many natural proteins, providing structural, catalytic and electron transfer functions. Reproducing these functions in a designed protein is the ultimate challenge to our understanding of them. Here, we present an artificial metallohydrolase, which has been shown by X-ray crystallography to contain two different metal ions—a Zn(II) ion, which is important for catalytic activity, and a Hg(II) ion, which provides structural stability. This metallohydrolase displays catalytic activity that compares well with several characteristic reactions of natural enzymes. It catalyses p-nitrophenyl acetate (pNPA) hydrolysis with an efficiency only ~100-fold less than that of human carbonic anhydrase (CA)II and at least 550-fold better than comparable synthetic complexes. Similarly, CO2 hydration occurs with an efficiency within ~500-fold of CAII. Although histidine residues in the absence of Zn(II) exhibit pNPA hydrolysis, miniscule apopeptide activity is observed for CO2 hydration. The kinetic and structural analysis of this first de novo designed hydrolytic metalloenzyme reveals necessary design features for future metalloenzymes containing one or more metals.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Holm, R. H., Kennepohl, P. & Solomon, E. I. Structural and functional aspects of metal sites in biology. Chem. Rev. 96, 2239–2314 (1996).
Lu, Y., Yeung, N., Sieracki, N. & Marshall, N. M. Design of functional metalloproteins. Nature 460, 855–862 (2009).
Nanda, V. & Koder, R. Designing artificial enzymes by intuition and computation. Nature Chem. 2, 15–24 (2010).
Kaplan, J. & DeGrado, W. F. De novo design of catalytic proteins. Proc. Natl Acad. Sci. USA 101, 11566–11570 (2004).
Faiella, M. et al. An artificial di-iron oxo-protein with phenol oxidase activity. Nature Chem. Biol. 5, 882–884 (2009).
Dieckmann, G. R. et al. The role of protonation and metal chelation preferences in defining the properties of mercury-binding coiled coils. J. Mol. Biol. 280, 897–912 (1998).
Farrer, B. T., Harris, N. P., Balchus, K. E. & Pecoraro, V. L. Thermodynamic model for the stabilization of trigonal thiolato mercury(II) in designed three-stranded coiled coils. Biochemistry 40, 14696–14705 (2001).
DeGrado, W. F., Summa, C. M., Pavone, V., Nastri, F. & Lombardi, A. De novo design and structural characterization of proteins and metalloproteins. Annu. Rev. Biochem. 68, 779–819 (1999).
Peacock, A. F. A., Stuckey, J. A. & Pecoraro, V. L. Chirally switching metal coordination environments in designed peptides. Angew. Chem. Int. Ed. 48, 7371–7374 (2009).
Handel, T. & DeGrado, W. F. De novo design of a Zn2+-binding protein. J. Am. Chem. Soc. 112, 6710–6711 (1990).
Pessi, A. et al. A designed metal-binding protein with a novel fold. Nature 362, 367–369 (1993).
Kiyokawa, T. et al. Binding of Cu(II) or Zn(II) in a de novo designed triple-stranded α-helical coiled-coil toward a prototype for a metalloenzyme. J. Pept. Res. 63, 347–353 (2004).
Müller, H. N. & Skerra, A. Grafting of a high-affinity Zn(II)-binding site on the β-barrel of retinol-binding protein results in enhanced folding stability and enables simplified purification. Biochemistry 33, 14126–14135 (1994).
Vita, C., Roumestand, C., Toma, F. & Ménez, A. Scorpion toxins as natural scaffolds for protein engineering. Proc. Natl Acad. Sci. USA 92, 6404–6408 (1995).
Farrer, B. T., McClure, C. P., Penner-Hahn, J. E. & Pecoraro, V. L. Arsenic(III)–cysteine interactions stabilize three-helix bundles in aqueous solution. Inorg. Chem. 39, 5422–5423 (2000).
Matzapetakis, M., Ghosh, D., Weng, T.-C., Penner-Hahn, J. E. & Pecoraro, V. L. Peptidic models for the binding of Pb(II), Bi(III) and Cd(II) to mononuclear thiolate binding sites. J. Biol. Inorg. Chem. 11, 876–890 (2006).
Dieckmann, G. R. et al. De novo design of mercury-binding two- and three-helical bundles. J. Am. Chem. Soc. 11, 876–890 (1997).
Pecoraro, V. L., Peacock, A. F. A., Iranzo, O. & Luczkowski, M. Understanding the biological chemistry of mercury using a de novo protein design strategy in Bioinorg. Chem. ACS Sympos. Ser. 183–197 (2009).
Touw, D. S., Nordman, C. E., Stuckey, J. A. & Pecoraro, V. L. Identifying important structural characteristics of arsenic resistance proteins by using designed three-stranded coiled coils. Proc. Natl Acad. Sci. USA 104, 11969–11974 (2007).
Verpoorte, J. A., Mehta, S. & Edsall, J. T. Esterase activities of human carbonic anhydrases B and C. J. Biol. Chem. 242, 4221–4229 (1967).
Gould, S. M. & Tawfik, D. S. Directed evolution of the promiscuous esterase activity of carbonic anhydrase II. Biochemistry 44, 5444–5452 (2005).
Kimura, E., Shiota, T., Koike, T., Shiro, M. & Kodama, M. A Zinc(II) complex of 1,5,9-triazacyclododecane ([12]aneN3) as a model for carbonic anhydrase. J. Am. Chem. Soc. 112, 5805–5811 (1990).
Olmo, C. P., Bohmerle, K. & Vahrenkamp, H. Zinc enzyme modeling with zinc complexes of polar pyrazolylborate ligands. Inorg. Chim. Acta 360, 1510–1516 (2007).
Koerner, T. B. & Brown, R. S. The hydrolysis of an activated ester by a tris(4,5-di-n-propyl-2-imidazolyl)phosphine–Zn2+ complex in neutral micellar medium as a model for carbonic anhydrase. Can. J. Chem. 80, 183–191 (2002).
Bazzicalupi, C. et al. Carboxy and phosphate esters cleavage with mono- and dinuclear zinc(II) macrocyclic complexes in aqueous solution. Crystal structure of [Zn2L1(μ-PP)2(MeOH)2](ClO4)2 (L1=[30]aneN6O4, PP−=diphenyl phosphate). Inorg. Chem. 36, 2784–2790 (1997).
Sprigings, T. G. & Hall, D. C. A simple carbonic anhydrase model which achieves catalytic hydrolysis by the formation of an ‘enzyme-substrate’-like complex. J. Chem. Soc. Perkin Trans. 2, 2063–2067 (2001).
Jairam, R., Potvin, P. G. & Balsky, S. Zn2+ inclusion complexes of endodentate tripodands as carbonic anhydrase-inspired artificial esterases. Part 2. Micellar systems. J. Chem. Soc. Perkin Trans. 2, 363–367 (1999).
Koike, T., Takamura, M. & Kimura, E. Role of zinc(II) in β-lactamase II: a model study with a zinc(II)-macrocyclic tetraamine (1,4,7,10-tetraazacyclododecane, cyclen) complex. J. Am. Chem. Soc. 116, 8443–8449 (1994).
Kimura, E., Hashimoto, H. & Koike, T. Hydrolysis of lipophilic esters catalyzed by a zinc(II) complex of a long alkyl-pendant macrocyclic tetraamine in micellar solution. J. Am. Chem. Soc. 118, 10963–10970 (1996).
Broo, K., Brive, L., Ahlberg, P. & Baltzer, L. Catalysis of hydrolysis and transesterification reactions of p-nitrophenyl esters by a designed helix-loop-helix dimer. J. Am. Chem. Soc. 119, 11362–11372 (1997).
Nilsson, J. & Baltzer, L. Reactive-site design in folded-polypeptide catalysts—the leaving group pKa of reactive esters sets the stage for cooperativity in nucleophilic and general-acid catalysis. Chem. Eur. J. 6, 2214–2220 (2000).
Bolon, D. N. & Mayo, S. L. Enzyme-like proteins by computational design. Proc. Natl Acad. Sci. USA 98, 14274–14279 (2001).
Innocenti, A. et al. Investigations of the esterase, phosphatase, and sulfatase activities of the cytosolic mammalian carbonic anhydrase isoforms I, II, and XIII with 4-nitrophenyl esters as substrates. Bioorg. Med. Chem. Lett. 18, 2267–2271 (2008).
Liang, Z., Xue, Y., Behravan, G., Jonsson, B.-H. & Lindskog, S. Importance of the conserved active-site residues Tyr7, Glu106, Thr199 for the catalytic function of human carbonic anhydrase II. Eur. J. Biochem. 211, 821–827 (1993).
Krebs, J. F., Ippolito, J. A., Christianson, D. W. & Fierke, C. A. Structural and functional importance of a conserved hydrogen bond network in human carbonic anhydrase II. J. Biol. Chem. 268, 27458–27466 (1993).
Woolley, P. Models for metal ion function in carbonic anhydrase. Nature 258, 677–682 (1975).
Huguet, J. & Brown, R. S. Catalytically active models for the active site in carbonic anhydrase. J. Am. Chem. Soc. 102, 7571–7572 (1980).
Brown, R. S., Curtis, N. J. & Huguet, J. Tris(4,5-diisopropylimidazol-2-yl)phosphine:zinc(2+). A catalytically active model for carbonic anhydrase. J. Am. Chem. Soc. 103, 6953–6959 (1981).
Brown, R. S., Salmon, D., Curtis, N. J. & Kusuma, S. Carbonic anhydrase models. 4. [Tris[(4,5-dimethyl-2-imidazolyl)methyl]phosphineoxide]cobalt(2+); a small-molecule mimic of the spectroscopic properties of Co(II) carbonic anhydrase. J. Am. Chem. Soc. 104, 3188–3194 (1982).
Slebocka-Tilk, H., Cocho, J. L., Frakman, Z. & Brown, R. S. Carbonic anhydrase models. 5. Tris(4,5-di-n-propyl-2-imidazolyl)phosphine-zinc(2+) and bis(4,5-diisopropyl-2-imidazolyl)-2-imidazolylphosphine-zinc(2+). Catalysts facilitating HCO3− ↔ CO2 interconversion. J. Am. Chem. Soc. 106, 2421–2431 (1984).
Zhang, X., van Eldik, R., Koike, T. & Kimura, E. Kinetics and mechanism of the hydration of CO2 and dehydration of HCO3− catalyzed by a Zn(II) complex of 1,5,9-triazacyclododecane as a model for carbonic anhydrase. Inorg. Chem. 32, 5749–5755 (1993).
Zhang, X. & van Eldik, R. A functional model for carbonic anhydrase: thermodynamic and kinetic study of a tetraazacyclododecane complex of zinc(II). Inorg. Chem. 34, 5606–5614 (1995).
Nakata, K. et al. Kinetic study of catalytic CO2 hydration by water-soluble model compound of carbonic anhydrase and anion inhibition effect on CO2 hydration. J. Inorg. Biochem. 89, 255–266 (2002).
Jackman, J. E., Merz, K. M. Jr & Fierke, C. A. Disruption of the active site solvent network in carbonic anhydrase II decreases the efficiency of proton transfer. Biochemistry 35, 16421–16428 (1996).
Iranzo, O., Cabello, C. & Pecoraro, V. L. Heterochromia in designed metallopeptides: geometry-selective binding of CdII in a de novo peptide. Angew. Chem. Int. Ed. 46, 6688–6691 (2007).
Peacock, A. F. A., Hemmingsen, L. & Pecoraro, V. L. Using diastereopeptides to control metal ion coordination in proteins. Proc. Natl Acad. Sci. USA 105, 16566–16571 (2008).
Wright, J. G., Tsang, H.-T., Penner-Hahn, J. E. & O'Halloran, T. V. Coordination chemistry of the Hg–MerR metalloregulatory protein: evidence for a novel tridentate Hg–cysteine receptor site. J. Am. Chem. Soc. 112, 2434–2435 (1990).
Gomis-Rüth, F.-X., Kress, L. F. & Bode, W. First structure of a snake venom metalloproteinase: a prototype for matrix metalloproteinases/collagenases. EMBO J. 12, 4151–4157 (1993).
Acknowledgements
V.L.P. acknowledges support from the National Institutes of Health (grant no. R01 ES0 12236), and M.L.Z. from the National Institutes of Health Chemistry–Biology Interface Training Program. Use of the Advanced Photon Source was supported by the US Department of Energy, Office of Science, Office of Basic Energy Sciences (contract no. DE-AC02-06CH11357). Use of the LS-CAT Sector 21 was supported by the Michigan Economic Development Corporation and the Michigan Technology Tri-Corridor (grant no. 085P1000817). J.A.S. is supported by the University of Michigan Center for Structural Biology.
Author information
Authors and Affiliations
Contributions
M.L.Z. carried out the experimental work and completed crystal structure refinement. A.F.A.P. obtained and solved the crystal structure. M.L.Z., A.F.A.P., J.A.S. and V.L.P. contributed to the design of the experiments and analysis of the data. All authors contributed to writing the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 1140 kb)
Rights and permissions
About this article
Cite this article
Zastrow, M., Peacock, A., Stuckey, J. et al. Hydrolytic catalysis and structural stabilization in a designed metalloprotein. Nature Chem 4, 118–123 (2012). https://doi.org/10.1038/nchem.1201
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.1201
This article is cited by
-
Nanozyme as detector and remediator to environmental pollutants: between current situation and future prospective
Environmental Science and Pollution Research (2023)
-
Overcoming universal restrictions on metal selectivity by protein design
Nature (2022)
-
Design and directed evolution of noncanonical β-stereoselective metalloglycosidases
Nature Communications (2022)
-
Nitrite reductase activity within an antiparallel de novo scaffold
JBIC Journal of Biological Inorganic Chemistry (2021)
-
Heteromeric three-stranded coiled coils designed using a Pb(ii)(Cys)3 template mediated strategy
Nature Chemistry (2020)