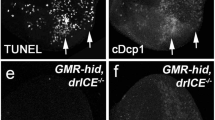
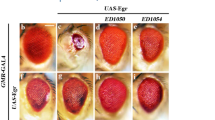
Abstract
Although Jun amino-terminal kinase (JNK) is known to mediate a physiological stress signal that leads to cell death, the exact role of the JNK pathway in the mechanisms underlying intrinsic cell death is largely unknown. Here we show through a genetic screen that a mutant of Drosophila melanogaster tumour-necrosis factor receptor-associated factor 1 (DTRAF1) is a dominant suppressor of Reaper-induced cell death. We show that Reaper modulates the JNK pathway through Drosophila inhibitor-of-apoptosis protein 1 (DIAP1), which negatively regulates DTRAF1 by proteasome-mediated degradation. Reduction of JNK signals rescues the Reaper-induced small eye phenotype, and overexpression of DTRAF1 activates the Drosophila ASK1 (apoptosis signal-regulating kinase 1; a mitogen-activated protein kinase kinase kinase) and JNK pathway, thereby inducing cell death. Overexpresson of DIAP1 facilitates degradation of DTRAF1 in a ubiquitin-dependent manner and simultaneously inhibits activation of JNK. Expression of Reaper leads to a loss of DIAP1 inhibition of DTRAF1-mediated JNK activation in Drosophila cells. Taken together, our results indicate that DIAP1 may modulate cell death by regulating JNK activation through a ubiquitin–proteasome pathway.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
White, K., Grether, M. E., Abrams, J. M., Young, L., Farrell, K. & Steller, H. Science 264, 677–683 (1994).
Grether, M. E., Abrams, J. M., Agapite, J., White, K. & Steller, H. Genes Dev. 9, 1694–1708 (1995).
Chen, P., Nordstrom, W., Gish, B. & Abrams, J. M. Genes Dev. 10, 1773–1782 (1996).
Hay, B. A., Wassarman, D. A. & Rubin, G. M. Cell 83, 1253–1262 (1995).
White, K., Tahaoglu, E. & Steller, H. Science 271, 805–807 (1996).
Pronk, G. J., Ramer, K., Amiri, P. & Williams, L. T. Science 271, 808–810 (1996).
Vucic, D., Kaiser, W. J., Harvey, A. J. & Miller, L. K. Proc. Natl Acad. Sci. USA 94, 10183–10188 (1997).
Vucic, D., Kaiser, W. J. & Miller, L. K. J. Biol. Chem. 273, 33915–33921 (1998).
Wang, S. L., Hawkins, C. J., Yoo, S. J., Muller, H. A. & Hay, B. A. Cell 98, 453–463 (1999).
Goyal, L., McCall, K., Agapite, J., Hartwieg, E. & Steller, H. EMBO J. 19, 589–597 (2000).
Du, C., Fang, M., Li, Y., Li, L. & Wang, X. Cell 102, 33–42 (2000).
Verhagen, A. M. et al. Cell 102, 43–53 (2000).
Suzuki, Y., Imai, Y., Nakayama, H., Takahashi, K., Takio, K. & Takahashi R. Mol. Cell 8, 613–621 (2001).
Wassarman, D. A., Solomon, N. M. & Rubin, G. M. Gene 169, 283–284 (1996).
Liu, H., Su, Y. C., Becker, E., Treisman, J. & Skolnik, E. Y. Curr. Biol. 9, 101–104 (1999).
Bradley, J. R. & Pober, J. S. Oncogene 20, 6482–6491 (2001).
Ichijo, H. et al. Science 275, 90–94 (1997).
Wing, J. P., Schwartz, L. M. & Nambu J. R. Mech. Dev. 102, 193–203 (2001).
Yoo, S. J. et al. Nature Cell Biol. 4, 416–424 (2002).
Hays, R., Wickline, L. & Cagan, R. Nature Cell Biol. 4, 425–431 (2002).
Ryoo, H. D., Bergmann, A., Gonen, H., Ciechanover, A. & Steller, H. Nature Cell Biol. 4, 432–438 (2002).
Holly, C. L., Olson, M. R., Colon-Ramos, D. A. & Kornbluth, S. Nature Cell Biol. 4, 439–444 (2002).
Wing, J. P. et al. Nature Cell Biol. 4, 451–456 (2002).
Kanuka, H. et al. Mol. Cell 4, 757–769 (1999).
Kanuka, H. et al. Proc. Natl Acad. Sci. USA 96, 145–150 (1999).
Sawamoto, K. et al. J. Cell Biol. 146, 361–372 (1999).
Nishitoh, H. et al. Mol. Cell 2, 389–395 (1998).
Geleziunas, R., Xu, W., Takeda, K., Ichijo, H. & Greene, W. C. Nature 410, 834–838 (2001).
Igaki, T. et al. Proc. Natl Acad. Sci. USA 97, 662–667 (2000).
Acknowledgements
We thank T. Hiratou and R. Akai for technical support; H. Steller, G. M. Rubin, B. Hay, M. Nakamura, T. Adachi-Yamada and J. Nambu for fly stocks; the Berkeley Drosophila Genome Project for reagents and information; the Bloomington Stock Center for fly stocks; Y. Uchiyama for encouragement; and B. Nelson for critical reading. This work was supported in part by grants from the Japanese Ministry of Education, Science, Sports, Culture and Technology (K.S., H.O. and M. M.) and by the Core Research for Evolutional Science and Technology (H.O.), the Japan Science and Technology Corporation, and the RIKEN Bioarchitect Research Project. T.I. is a research fellow of the Japan Society for the Promotion of Science. H.K. is a research fellow of the Special Postdoctoral Researchers Program, RIKEN. E.K. is a research fellow of the Junior Research Associate Program, RIKEN.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary figures and table
Figure S1 (PDF 1104 kb)
Figure S2
Supplementary table 1
Rights and permissions
About this article
Cite this article
Kuranaga, E., Kanuka, H., Igaki, T. et al. Reaper-mediated inhibition of DIAP1-induced DTRAF1 degradation results in activation of JNK in Drosophila. Nat Cell Biol 4, 705–710 (2002). https://doi.org/10.1038/ncb842
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb842
This article is cited by
-
Downregulation of respiratory complex I mediates major signalling changes triggered by TOR activation
Scientific Reports (2020)
-
Integrated omics in Drosophila uncover a circadian kinome
Nature Communications (2020)
-
Damage sensing by a Nox-Ask1-MKK3-p38 signaling pathway mediates regeneration in the adult Drosophila midgut
Nature Communications (2019)
-
Rationally-engineered reproductive barriers using CRISPR & CRISPRa: an evaluation of the synthetic species concept in Drosophila melanogaster
Scientific Reports (2018)
-
Drosophila MOF regulates DIAP1 and induces apoptosis in a JNK dependent pathway
Apoptosis (2016)