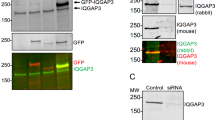
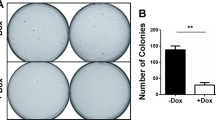
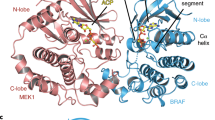
Abstract
Rac is a member of the Ras superfamily of GTPases and functions as a GDP/GTP-regulated switch1. Formation of active Rac-GTP is stimulated by Dbl family guanine nucleotide exchange factors (GEFs), such as Tiam1 (ref. 2). Once activated, Rac stimulates signalling pathways that regulate actin organization, gene expression and cellular proliferation. Rac also functions downstream of the Ras oncoprotein in pathways that stimulate membrane ruffling3, growth transformation4,5, activation of the c-Jun amino-terminal kinase (JNK) mitogen-activated protein kinase6, activation of the NF-κB transcription factor and promotion of cell survival7,8. Although recent studies support phosphatidylinositol 3-OH kinase (PI(3)K)-dependent mechanisms through which Ras might activate Rac (refs 9,10), the precise mechanism remains to be determined. Here we demonstrate that Tiam1, a Rac-specific GEF, preferentially associates with activated GTP-bound Ras through a Ras-binding domain. Furthermore, activated Ras and Tiam1 cooperate to cause synergistic formation of Rac-GTP in a PI(3)K-independent manner. Thus, Tiam1 can function as an effector that directly mediates Ras activation of Rac.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Bar-Sagi, D. & Hall, A. Cell 103, 227–238 (2000).
Zheng, Y. Trends Biochem. Sci. 26, 724–732 (2001).
Ridley, A. J., Paterson, H. F., Johnston,C. L., Diekmann, D. & Hall, A. Cell 70, 401–410 (1992).
Qiu, R.-G., Chen, J., Kirn, D., McCormick, F. & Symons, M. Nature 374, 457–459 (1995).
Khosravi-Far, R., Solski, P. A., Kinch, M. S., Burridge, K. & Der, C. J. Mol. Cell. Biol. 15, 6443–6453 (1995).
Minden, A., Lin, A., Claret, F.-X., Abo, A. & Karin, M. Cell 81, 1147–1157 (1995).
Sulciner, D. J. et al. Mol. Cell. Biol. 16, 7115–7121 (1996).
Joneson, T. & Bar-Sagi, D. Mol. Cell. Biol. 19, 5892–5901 (1999).
Nimnual, A. S., Yatsula, B. A. & Bar-Sagi, D. Science 279, 560–563 (1998).
Scita, G. et al. Nature 401, 290–293 (1999).
Worthylake, D. K., Rossman, K. L. & Sondek, J. Nature 408, 682–688 (2000).
Ponting, C. P. J. Mol. Med. 77, 695–698 (1999).
Bar-Sagi, D. & Feramisco, J. R. Science 233, 1061–1068 (1986).
Rodriguez-Viciana, P. et al. Cell 89, 457–467 (1997).
Zondag, G. C. et al. J. Cell Biol. 149, 775–782 (2000).
Adari, H., Lowy, D. R., Willumsen, B. M., Der, C. J. & McCormick, F. Science 240, 518–521 (1988).
Reuther, G. W. et al. J. Biol Chem. (in the press).
Peterson, S. N. et al. J. Biol. Chem. 271, 29903–29908 (1996).
Fabian, J. R., Vojtek, A. B., Cooper, J. A. & Morrison, D. K. Proc. Natl Acad. Sci. USA 91, 5982–5986 (1994).
Nassar, N. et al. Nature 375, 554–560 (1995).
Han, J. et al. Science 279, 558–560 (1998).
Fleming, I. N., Gray, A. & Downes, C. P. Biochem. J. 351, 173–182 (2000).
Murphy, G. A. et al. J. Biol. Chem. 277, 9966–9975 (2002).
Crompton, A. M. et al. J. Biol. Chem. 275, 25751–25759 (2000).
Snyder, J. T. et al. J. Biol. Chem. 276, 45868–45875 (2001).
Malliri, A. et al. Nature 417, 867–871 (2002).
Chang, E. C. et al. Cell 79, 131–141 (1994).
Westwick, J. K. et al. Mol. Cell. Biol. 17, 1324–1335 (1997).
Altschul, S. F. et al. Nucleic Acids Res. 25, 3389–3402 (1997).
Fischer, D. et al. Proteins 3, S209–S217 (1999).
Acknowledgements
We thank C.I. Behe and M. Pham for technical assistance, and M. Rand for assistance in figure and manuscript preparation. We thank M. Ostrowski, G. Bollag, J. Downward, C. Hauser and B. Mayer for plasmid constructs. This work was supported by grants from the National Institutes of Health to D.P.S. (GM62338), J.S.(GM62299), and C.J.D. (CA69577), as well as a career award from the Pew Charitable Trusts (J.S.). G.W.R. is a Leukemia and Lymphoma Society Special Fellow. D.P.S. is a Year 2000 Scholar of the EJLB Foundation and a recipient of the Burroughs-Wellcome New Investigator Award in the Basic Pharmacological Sciences. Part of this work was supported by the Dutch Cancer Society, the European Community and the Association for International Cancer Research (J.G.C.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lambert, J., Lambert, Q., Reuther, G. et al. Tiam1 mediates Ras activation of Rac by a PI(3)K-independent mechanism. Nat Cell Biol 4, 621–625 (2002). https://doi.org/10.1038/ncb833
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb833
This article is cited by
-
Oncogenic alterations in advanced NSCLC: a molecular super-highway
Biomarker Research (2024)
-
The genetic profile and molecular subtypes of human pseudomyxoma peritonei and appendiceal mucinous neoplasms: a systematic review
Cancer and Metastasis Reviews (2023)
-
Circular dorsal ruffles disturb the growth factor-induced PI3K-AKT pathway in hepatocellular carcinoma Hep3B cells
Cell Communication and Signaling (2022)
-
Drugging KRAS: current perspectives and state-of-art review
Journal of Hematology & Oncology (2022)
-
Multiplexed GTPase and GEF biosensor imaging enables network connectivity analysis
Nature Chemical Biology (2020)