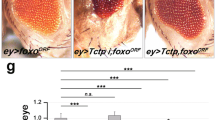
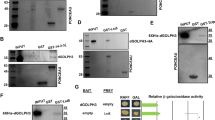
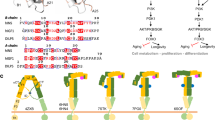
Abstract
Genetic studies in Drosophila melanogaster underscore the importance of the insulin-signalling pathway in controlling cell, organ and animal size1. Effectors of this pathway include Chico (the insulin receptor substrate homologue), dPI(3)K, dPKB, dPTEN, and dS6K2. Mutations in any of these components have a striking effect on cell size and number3,4,5,6,7, with the exception of dS6K8. Mutants in dS6K affect cell size but not cell number, seemingly consistent with arguments that dS6K is a distal effector in the signalling pathway, directly controlled by dTOR, a downstream effector of dPI(3)K and dPKB1,9,10. Unexpectedly, recent studies showed that dS6K activity is unimpaired in chico-deficient larvae, suggesting that dS6K activation may be mediated through the dPI(3)K docking sites of the Drosophila insulin receptor11. Here, we show genetically, pharmacologically and biochemically that dS6K resides on an insulin signalling pathway distinct from that of dPKB, and surprisingly also from that of dPI(3)K. More striking, despite dPKB-dPI(3)K-independence, dS6K activity is dependent on the Drosophila homologue of the phosphoinositide-dependent protein kinase 1, dPDK1, demonstrating that both dPDK1, as well as dTOR, mediated dS6K activation is phosphatidylinositide-3,4,5-trisphosphate (PIP3)–independent.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Edgar, B. A. Nature Cell Biol. 1, E191–E193 (1999).
Thomas, G. Nature Cell Biol. 2, E71–E72 (2000).
Böhni, R. et al. Cell 97, 865–875 (1999).
Leevers, S. J., Weinkove, D., MacDougall, L. K., Hafen, E. & Waterfield, M. D. EMBO J. 15, 6584–6594 (1996).
Stocker, H. & Hafen, E. Curr. Opin. Genet. Dev. 10, 529–535 (2000).
Huang, H. et al. Development 126, 5365–5372 (1999).
Goberdhan, D. C., Paricio, N., Goodman, E. C., Mlodzik, M. & Wilson, C. Genes Dev. 13, 3244–3258 (1999).
Montagne, J. et al. Science 285, 2126–2129 (1999).
Tapon, N., Ito, N., Dickson, B. J., Treisman, J. E. & Hariharan, I. K. Cell 105, 345–355 (2001).
Potter, C. J., Huang, H. & Xu, T. Cell 105, 357–368 (2001).
Oldham, S., Montagne, J., Radimerski, T., Thomas, G. & Hafen, E. Genes Dev. 14, 2689–2694 (2000).
Alessi, D. R. et al. Curr. Biol. 7, 261–269 (1997).
Pullen, N. et al. Science 279, 707–710 (1998).
Alessi, D. R., Kozlowski, M. T., Weng, Q. P., Morrice, N. & Avruch, J. Curr. Biol. 8, 69–81 (1998).
Sekulic, A. et al. Cancer Res. 60, 3504–3513 (2000).
Dennis, P. B., Pullen, N., Pearson, R. B., Kozma, S. C. & Thomas, G. J. Biol. Chem. 273, 14845–14852 (1998).
Zhang, H., Stallock, J. P., Ng, J. C., Reinhard, C. & Neufeld, T. P. Genes Dev. 14, 2712–2724 (2000).
Dufner, A., Andjelkovic, M., Burgering, B. M. T., Hemmings, B. A. & Thomas, G. Mol. Cell. Biol. 19, 4525–4534 (1999).
Rintelen, F., Stocker, H., Thomas, G. & Hafen, E. Proc. Natl Acad. Sci. USA, 98, 15020–15025 (2001).
Hara, K. et al. J. Biol. Chem. 273, 14484–14494 (1998).
Staveley, B. E. et al. Curr. Biol. 8, 599–602 (1998).
Scanga, S. E. et al. Oncogene 19, 3971–3977 (2000).
Haruta, T. et al. Mol. Endocrinol. 14, 783–794 (2000).
Majewski, M. et al. Proc. Natl Acad. Sci. USA 97, 4285–4290 (2000).
Van der Kaay, J., Cullen, P. J. & Downes, C. P. Methods Mol. Biol. 105, 109–125 (1998).
Verdu, J., Buratovich, M. A., Wilder, E. L. & Birnbaum, M. J. Nature Cell Biol. 1, 500–506 (1999).
Miron, M. et al. Nature Cell Biol. 3, 596–601 (2001).
Weinkove, D., Neufeld, T. P., Twardzik, T., Waterfield, M. D. & Leevers, S. J. Curr. Biol. 9, 1019–1029 (1999).
Yano, S., Tokumitsu, H. & Soderling, T. R. Nature 396, 584–587 (1998).
Alessi, D. et al. EMBO J. 23, 6541–6551 (1996).
Conus, N. M., Hemmings, B. A. & Pearson, R. B. J. Biol. Chem. 273, 4776–4782 (1998).
Montagne, J., Radimerski, T. & Thomas, G. Sci. STKE 2001, E36 (2001).
Hidalgo, M. & Rowinsky, E. K. Oncogene 19, 6680–6686 (2000).
Alessi, D. R. et al. Curr. Biol. 7, 776–789 (1997).
Rørth, P. Proc. Natl Acad. Sci. USA 93, 12418–12422 (1996).
Radimerski, T. et al. Biochemistry 39, 5766–5774 (2000).
Andjelkovic, M. et al. J. Biol. Chem. 270, 4066–4075 (1995).
Acknowledgements
We thank S. Leevers for providing fly stocks, B. Hemmings for providing the dPKB antibody, P. Dennis for critical reading of the manuscript, R. Guggenheim and D. Mathys for SEM. This work was supported by grants from the Roche Research Foundation to T.R. and G.T. and from the Swiss Cancer League to E.H. and G.T.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Figure S1
Model of the signalling events leading to the activation of dS6K and dPKB in Drosophila. (PDF 90 kb)
Rights and permissions
About this article
Cite this article
Radimerski, T., Montagne, J., Rintelen, F. et al. dS6K-regulated cell growth is dPKB/dPI(3)K-independent, but requires dPDK1. Nat Cell Biol 4, 251–255 (2002). https://doi.org/10.1038/ncb763
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb763
This article is cited by
-
Ecdysone regulates Drosophila wing disc size via a TORC1 dependent mechanism
Nature Communications (2021)
-
A Drosophila genetic screen for suppressors of S6kinase-dependent growth identifies the F-box subunit Archipelago/FBXW7
Molecular Genetics and Genomics (2019)
-
Inhibition of mammalian target of rapamycin improves neurobehavioral deficit and modulates immune response after intracerebral hemorrhage in rat
Journal of Neuroinflammation (2014)
-
1α,25-Dihydroxyvitamin-D3-3-Bromoacetate Regulates AKT/mTOR Signaling Cascades: A Therapeutic Agent for Psoriasis
Journal of Investigative Dermatology (2013)