Abstract
Microtubules tether centrosomes together during interphase1. How this is accomplished and what benefit it provides to the cell is not known. We have identified a bipolar, minus-end-directed kinesin, Kif25, that suppresses centrosome separation. Kif25 is required to prevent premature centrosome separation during interphase. We show that premature centrosome separation leads to microtubule-dependent nuclear translocation, culminating in eccentric nuclear positioning that disrupts the cortical spindle positioning machinery. The activity of Kif25 during interphase is required to maintain a centred nucleus to ensure the spindle is stably oriented at the onset of mitosis.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
12 May 2017
In the version of this Letter originally published, the authors inadvertently omitted a key reference. The following reference has been added: '37. Chozinski, T. J. et al. Expansion microscopy with conventional antibodies and fluorescent proteins. Nat. Methods 13, 485–488 (2016).' Original reference 37 has been renumbered to reference 38 accordingly. Furthermore, a new sentence citing this reference has been added at the end of the ‘Fixed and live cell imaging’ section in the Methods: 'Super-resolution expansion microscopy was performed as described37.' These changes have been made in the online version of the Letter.
References
Jean, C., Tollon, Y., Raynaud-Messina, B. & Wright, M. The mammalian interphase centrosome: two independent units maintained together by the dynamics of the microtubule cytoskeleton. Eur. J. Cell Biol. 78, 549–560 (1999).
Saunders, W. S. & Hoyt, M. A. Kinesin-related proteins required for structural integrity of the mitotic spindle. Cell 70, 451–458 (1992).
Raaijmakers, J. A. et al. Nuclear envelope-associated dynein drives prophase centrosome separation and enables Eg5-independent bipolar spindle formation. EMBO J. 31, 4179–4190 (2012).
Kapoor, T. M., Mayer, T. U., Coughlin, M. L. & Mitchison, T. J. Probing spindle assembly mechanisms with monastrol, a small molecule inhibitor of the mitotic kinesin, Eg5. J. Cell Biol. 150, 975–988 (2000).
Fry, A. M. et al. C-Nap1, a novel centrosomal coiled-coil protein and candidate substrate of the cell cycle-regulated protein kinase Nek2. J. Cell Biol. 141, 1563–1574 (1998).
Mayor, T., Stierhof, Y. D., Tanaka, K., Fry, A. M. & Nigg, E. A. The centrosomal protein C-Nap1 is required for cell cycle-regulated centrosome cohesion. J. Cell Biol. 151, 837–846 (2000).
Bahe, S., Stierhof, Y. D., Wilkinson, C. J., Leiss, F. & Nigg, E. A. Rootletin forms centriole-associated filaments and functions in centrosome cohesion. J. Cell Biol. 171, 27–33 (2005).
Faragher, A. J. & Fry, A. M. Nek2A kinase stimulates centrosome disjunction and is required for formation of bipolar mitotic spindles. Mol. Biol. Cell 14, 2876–2889 (2003).
Mardin, B. R. et al. Components of the Hippo pathway cooperate with Nek2 kinase to regulate centrosome disjunction. Nat. Cell Biol. 12, 1166–1176 (2010).
Mardin, B. R. et al. EGF-induced centrosome separation promotes mitotic progression and cell survival. Dev. Cell 25, 229–240 (2013).
Endres, N. F., Yoshioka, C., Milligan, R. A. & Vale, R. D. A lever-arm rotation drives motility of the minus-end-directed kinesin Ncd. Nature 439, 875–878 (2006).
Mountain, V. et al. The kinesin-related protein, HSET, opposes the activity of Eg5 and cross-links microtubules in the mammalian mitotic spindle. J. Cell Biol. 147, 351–366 (1999).
Kwon, M. et al. Mechanisms to suppress multipolar divisions in cancer cells with extra centrosomes. Genes Dev. 22, 2189–2203 (2008).
Goshima, G., Nédélec, F. & Vale, R. Mechanisms for focusing mitotic spindle poles by minus end-directed motor proteins. J. Cell Biol. 171, 229–240 (2005).
Endow, S. A. & Komma, D. J. Centrosome and spindle function of the Drosophila Ncd microtubule motor visualized in live embryos using Ncd-GFP fusion proteins. J. Cell Sci. 109, 2429–2442 (1996).
Okamoto, S., Matsushima, M. & Nakamura, Y. Identification, genomic organization, and alternative splicing of KNSL3, a novel human gene encoding a kinesin-like protein. Cytogenet. Cell Genet. 83, 25–29 (1998).
Maliga, Z. et al. A genomic toolkit to investigate kinesin and myosin motor function in cells. Nat. Cell Biol. 15, 325–334 (2013).
Miki, H., Okada, Y. & Hirokawa, N. Analysis of the kinesin superfamily: insights into structure and function. Trends Cell Biol. 15, 467–476 (2005).
Kashina, A. S. et al. A bipolar kinesin. Nature 379, 270–272 (1996).
Smith, E. et al. Differential control of Eg5-dependent centrosome separation by Plk1 and Cdk1. EMBO J. 30, 2233–2245 (2011).
Domnitz, S. B., Wagenbach, M., Decarreau, J. & Wordeman, L. MCAK activity at microtubule tips regulates spindle microtubule length to promote robust kinetochore attachment. J. Cell Biol. 197, 231–237 (2012).
Leonhardt, H. et al. Dynamics of DNA replication factories in living cells. J. Cell Biol. 149, 271–280 (2000).
Kisielewska, J., Lu, P. & Whitaker, M. GFP-PCNA as an S-phase marker in embryos during the first and subsequent cell cycles. Biol. Cell 97, 221–229 (2005).
Tsai, L. H. & Gleeson, J. G. Nucleokinesis in neuronal migration. Neuron 46, 383–388 (2005).
Salpingidou, G., Smertenko, A., Hausmanowa-Petrucewicz, I., Hussey, P. J. & Hutchison, C. J. A novel role for the nuclear membrane protein emerin in association of the centrosome to the outer nuclear membrane. J. Cell Biol. 178, 897–904 (2007).
Minc, N., Burgess, D. & Chang, F. Influence of cell geometry on division-plane positioning. Cell 144, 414–426 (2011).
Grill, S. W. & Hyman, A. A. Spindle positioning by cortical pulling forces. Dev. Cell 8, 461–465 (2005).
Laan, L. et al. Cortical dynein controls microtubule dynamics to generate pulling forces that position microtubule asters. Cell 148, 502–514 (2012).
Burkel, B. M., Von Dassow, G. & Bement, W. M. Versatile fluorescent probes for actin filaments based on the actin-binding domain of utrophin. Cell Motil. Cytoskeleton 64, 822–832 (2007).
Stumpff, J., von Dassow, G., Wagenbach, M., Asbury, C. & Wordeman, L. The kinesin-8 motor Kif18A suppresses kinetochore movements to control mitotic chromosome alignment. Dev. Cell 14, 252–262 (2008).
Kiyomitsu, T. & Cheeseman, I. M. Chromosome- and spindle-pole-derived signals generate an intrinsic code for spindle position and orientation. Nat. Cell Biol. 14, 311–317 (2012).
O’Connell, C. B. & Wang, Y. L. Mammalian spindle orientation and position respond to changes in cell shape in a dynein-dependent fashion. Mol. Biol. Cell 11, 1765–1774 (2000).
Tame, M. A., Raaijmakers, J. A., Afanasyev, P. & Medema, R. H. Chromosome misalignments induce spindle-positioning defects. EMBO Rep. 17, 317–325 (2016).
Peyre, E. et al. A lateral belt of cortical LGN and NuMA guides mitotic spindle movements and planar division in neuroepithelial cells. J. Cell Biol. 193, 141–154 (2011).
Erickson, H. Size and shape of protein molecules at the nanometer level determined by sedimentation, gel filtration, and electron microscopy. Biol. Proced. Online 11, 32–51 (2009).
Carpenter, A. E. et al. CellProfiler: image analysis software for identifying and quantifying cell phenotypes. Genome Biol. 7, R100 (2006).
Chozinski, T. J. et al. Expansion microscopy with conventional antibodies and fluorescent proteins. Nat. Methods 13, 485–488 (2016).
Decarreau, J., Driver, J., Asbury, C. & Wordeman, L. Rapid measurement of mitotic spindle orientation in cultured mammalian cells. Methods Mol. Biol. 1136, 31–40 (2014).
Acknowledgements
This study was supported by National Institutes of Health Grant GM69429 to L.W.; National Institutes of Health Fellowship F32GM105099 and American Cancer Society Fellowship PF1409901CCG to J.D.; NIH grant GM118396 to J.K. and Burroughs Wellcome Career Award to J.C.V.
Author information
Authors and Affiliations
Contributions
J.D. and L.W. are responsible for planning the project and experiments as well as experimental analysis and writing the manuscript. M.W. performed and analysed experiments. E.L. and J.K. performed and analysed experiments using electron microscopy for Kif25 structure. A.R.H. and J.C.V. performed and analysed experiments using super-resolution microscopy.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Initial characterization of Kif25.
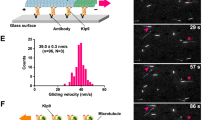
(a) RT-PCR was carried out as described in Methods to show endogenous Kif25 expression in HeLa cells. Kif18A was used as a positive control and showed signal above threshold (Ct) at cycle 15, Kif25 signal increased over threshold at cycle 20 consistent with its expression in our HeLa cells. Inset shows emission of SYBR green/emission of ROX dye, Rn, as a measure of amplification. Negative controls in which the cDNA or the reverse transcriptase were omitted from the reaction showed no significant amplification of signal, the no cDNA reaction shown passed threshold at cycle 36 consistent with no significantly amplified message, error bars represent mean ± S.D. for n = 3 experiments. (b) Cells were treated for 72 hr on 100 cm dishes with either control siRNA or one of two separate Kif25 siRNA constructs. Cell lysates were separated by SDS-Page. Proteins transferred to nitrocellulose membranes were stained with polyclonal anti-Kif25 antibodies (Santa Cruz Biotechnology) to show endogenous Kif25 KD. α-tubulin was used as a loading control. The experiment was performed twice. (c) Hydrodynamic analysis of purified GFP-Kif25 shows it to be a tetrameric motor. Optical scans of gel lanes from gel filtration and velocity sedimentation were quantified with ImageJ. Gaussian peaks were fit to protein gel bands and standard curves were constructed with Prism from published data for standard proteins. Three gel filtration columns, internally controlled, were run on GFP-Kif25 and two dimeric deletion mutants. Gaussian peaks were fit to data of optical scans of Coomassie-stained gel fractions. Quality of fits to the data were assessed by R-squared analysis. For gel filtration chromatograph, the mean R-squared was 0.98 +/− 0.012 (s.d., n = 12). For velocity sedimentation, the mean R-squared was 0.92 +/− 0.122 (s.d., n = 20). [Please provide information on reproducibility].
Supplementary Figure 2 Alignment of Kif25 protein sequences obtained by blast search of human Kif25 sequence.
Alignment was created using CLC Sequence Viewer version 7.0.2 (CLC Bio, Cambridge, MA). Macaca fascicularis Kif25 protein, 586 amino acids (GenBank Accession AB168279), Miki et al. human Kif25, 438 amino acids (Reported in Miki et al., 2005, supplemental application 3), and Okamoto et al., 1998 human Kif25, 384 amino acids (GenBank accession number NP_085118) represent isolated cDNA sequences while all other sequences are predicted based on genome analysis algorithms. Macaque Kif25, Chimp Kif25, 585 amino acids (GenBank accession SM_003820208), Finch Kif25, 587 amino acids (Genbank accession XP_002189451), and Stickleback Kif25, 580 amino acids (UniProt number G3PCB8), all show significant N-terminal extensions relative to the initial human Kif25 sequence. Human Kif25 from Miki et al. contains an additional 54 N-terminal amino acids relative to the original Okamoto et al. human Kif25 sequence suggesting that an N-terminal extension is present on human Kif25 as well. Residues in alignment colored red show <33% homology between sequences, black residues are between 33–66% homologous and blue residues are >66% homologous between sequences. Percent conservation (0–100%) of the sequences at each position is shown as a line plot below the alignment. Very high sequence homology is observed in the putative kinesin motor domain at the C-terminus of each protein.
Supplementary Figure 3 Kif25 MT binding in vitro and in centrosome separation.
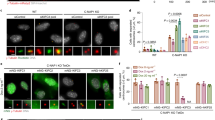
(a) MT cross-linking by EGFP-Kif25 was shown in vitro using Alexa-568 labeled taxol stabilized MTs and 500 nM of conventional kinesin I or EGFP-Kif25, scale bar, 20 μm Shown are representative images from 3 experiments. (b) Asynchronous HeLa cells were fixed and stained for α- and γ-tubulin before imaging interphase cells for centrosome separation. Cumulative frequency plot shows data pooled from 3 separate experiments in each condition (n = 150 control, n = 150 Kif25 KD Noc cells). Control or Kif25 overexpressing cells were treated with 20 μM nocodazole for 2 hr prior to fixation to depolymerize MTs. (c) Schematic of kinase signaling cascade leading to centrosome separation. (d) Quantitative IF to determine the relative levels of phosphor-Aurora A, Phospho-Plk1, Nek2A, and C-Nap1 at the centrosome during interphase. n = 75 cells were analyzed in each condition in cells stained with the indicated antibody and γ-tubulin to mark the centrosome. A circular ROI was drawn around each centrosome and the intensity measured for each experimental protein. A second intensity measurement was taken in the cytoplasm as background intensity for each cell. The displayed intensity represents the background corrected value for protein. Error bars represent mean ± s.e.m., ∗P = 0.03, ∗∗P = 0.0015. N. S; not significant. Statistics were analysed using an unpaired t-test.
Supplementary Figure 4 Overexpression of Kif25 leads to mitotic delay.
(a) Mitotic timing was measured following double thymidine block showing that Kif25 KD does not effect the time from NEB to anaphase (n = 46 cells for Kif25 KD and n = 35 for control cells), while Kif25 overexpression does (P < 0.0001, n = 50 cells for Kif25 OE). (b) Inter-centromere distance was measured in cells transfected with mRFP-CenpB and imaged every 3 s for 5 min and compared to a Nocodazole control representing the resting inter-centromere length in the absence of MTs. Measured distances for control (n = 72 centromere pairs from 7 cells), Kif25 OE (n = 76 centromere pairs from 6 cells, P < 0.0001), Kif25 KD (n = 78 centromere pairs from 5 cells, P < 0.0001), and Nocodazole-treated cells (n = 77 centromere pairs from 6 cells, P < 0.0001). (c) Quantification of MAD1 persistence time, taken as the time elapsed between the spindle reaching 8 μm in length and the disappearance of the last Mad1 foci. Kif25 OE causes Mad1 to persist for much longer at the kinetochore (n = 16 cells, P = 0.001). (d) Cells were fixed 72 hr after transient transfection of mCh-H2B or EGFP-Kif25 to count the DNA content of interphase cells. Data are for n = 3 experiments, counting 100 cells for each independent experiment. ∗P = 0.02. Data are mean ± SEM. Statistics compared using unpaired t-test.
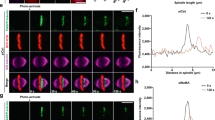
Supplementary Figure 5 Nuclear position dictates spindle orientation defect.
(a) Measurement of cell dimensions was carried out in cells expressing mRFP-utrophin. Cell length (a) was measured from cortex-to-cortex in a plane parallel to the spindle, while width was measured from cortex-to-cortex perpendicular to the spindle in the plane of the metaphase plate (n = 50 cells/condition). Cell height (b) was measured by taking optical slices through fixed cells (n = 50 cells/condition). (c) Histogram of measured spindle angles following Kif18A KD. (d) Positional data for centrosomes and nucleus within the cell just prior to entry into mitosis (data taken from last timepoint prior to cell rounding). Plotted are distances measured in live cells from the centrosome-to-cell center, centrosome-to-nuclear center and nucleus-to-cell center. Nucleus and cell center were determined by using Fiji to measure the center-of-mass of an outline of each structure. n = 40 for control cells and n = 37 for Kif25 KD cells. (e,f) Data from d was parsed to separate cells with parallel and angled mitotic spindles in control cells (n = 18 cells parallel and n = 7 cells angled) (e) or Kif25 KD cells (n = 21 cells parallel and n = 15 cells angled) (f). Kif25 KD cells show a statistically significant increase in distance from nuclear center to cell center in angled spindles compared to parallel spindles (2 rightmost columns in f, P = 0.0024). Error bars represent mean ± s.e.m.
Supplementary information
Supplementary Information
Supplementary Information (PDF 483 kb)
Rights and permissions
About this article
Cite this article
Decarreau, J., Wagenbach, M., Lynch, E. et al. The tetrameric kinesin Kif25 suppresses pre-mitotic centrosome separation to establish proper spindle orientation. Nat Cell Biol 19, 384–390 (2017). https://doi.org/10.1038/ncb3486
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb3486
This article is cited by
-
Kinesin KIFC3 is essential for microtubule stability and cytokinesis in oocyte meiosis
Cell Communication and Signaling (2024)
-
Genetic determinants of micronucleus formation in vivo
Nature (2024)
-
Expansion microscopy: principles and uses in biological research
Nature Methods (2019)
-
KIFC3 directs a centrosome cohesive force
Nature Cell Biology (2019)
-
The balance between KIFC3 and EG5 tetrameric kinesins controls the onset of mitotic spindle assembly
Nature Cell Biology (2019)