Abstract
Reprogrammed glucose metabolism as a result of increased glycolysis and glucose uptake is a hallmark of cancer. Here we show that cancer cells can suppress glucose uptake by non-tumour cells in the premetastatic niche, by secreting vesicles that carry high levels of the miR-122 microRNA. High miR-122 levels in the circulation have been associated with metastasis in breast cancer patients, and we show that cancer-cell-secreted miR-122 facilitates metastasis by increasing nutrient availability in the premetastatic niche. Mechanistically, cancer-cell-derived miR-122 suppresses glucose uptake by niche cells in vitro and in vivo by downregulating the glycolytic enzyme pyruvate kinase. In vivo inhibition of miR-122 restores glucose uptake in distant organs, including brain and lungs, and decreases the incidence of metastasis. These results demonstrate that, by modifying glucose utilization by recipient premetastatic niche cells, cancer-derived extracellular miR-122 is able to reprogram systemic energy metabolism to facilitate disease progression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Vander Heiden, M. G., Cantley, L. C. & Thompson, C. B. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science 324, 1029–1033 (2009).
Zhao, F. Q. & Keating, A. F. Functional properties and genomics of glucose transporters. Curr. Genomics 8, 113–128 (2007).
DeBerardinis, R. J., Lum, J. J., Hatzivassiliou, G. & Thompson, C. B. The biology of cancer: metabolic reprogramming fuels cell growth and proliferation. Cell Metab. 7, 11–20 (2008).
Barthel, A. et al. Regulation of GLUT1 gene transcription by the serine/threonine kinase Akt1. J. Biol. Chem. 274, 20281–20286 (1999).
O’Rourke, J. F., Pugh, C. W., Bartlett, S. M. & Ratcliffe, P. J. Identification of hypoxically inducible mRNAs in HeLa cells using differential-display PCR. Role of hypoxia-inducible factor-1. Eur. J. Biochem. 241, 403–410 (1996).
Semenza, G. L., Roth, P. H., Fang, H. M. & Wang, G. L. Transcriptional regulation of genes encoding glycolytic enzymes by hypoxia-inducible factor 1. J. Biol. Chem. 269, 23757–23763 (1994).
Spoden, G. A. et al. The SUMO-E3 ligase PIAS3 targets pyruvate kinase M2. J. Cell Biochem. 107, 293–302 (2009).
Yang, W. et al. ERK1/2-dependent phosphorylation and nuclear translocation of PKM2 promotes the Warburg effect. Nat. Cell Biol. 14, 1295–1304 (2012).
Gao, X., Wang, H., Yang, J. J., Liu, X. & Liu, Z. R. Pyruvate kinase M2 regulates gene transcription by acting as a protein kinase. Mol. Cell 45, 598–609 (2012).
Yang, W. et al. Nuclear PKM2 regulates β-catenin transactivation upon EGFR activation. Nature 480, 118–122 (2011).
Lee, J., Kim, H. K., Han, Y. M. & Kim, J. Pyruvate kinase isozyme type M2 (PKM2) interacts and cooperates with Oct-4 in regulating transcription. Int. J. Biochem. Cell Biol. 40, 1043–1054 (2008).
Luo, W. et al. Pyruvate kinase M2 is a PHD3-stimulated coactivator for hypoxia-inducible factor 1. Cell 145, 732–744 (2011).
Bartel, D. P. MicroRNAs: target recognition and regulatory functions. Cell 136, 215–233 (2009).
Calin, G. A. & Croce, C. M. MicroRNA signatures in human cancers. Nat. Rev. Cancer 6, 857–866 (2006).
Mitchell, P. S. et al. Circulating microRNAs as stable blood-based markers for cancer detection. Proc. Natl Acad. Sci. USA 105, 10513–10518 (2008).
Taylor, D. D. & Gercel-Taylor, C. MicroRNA signatures of tumor-derived exosomes as diagnostic biomarkers of ovarian cancer. Gynecol. Oncol. 110, 13–21 (2008).
Wu, X. et al. De novo sequencing of circulating miRNAs identifies novel markers predicting clinical outcome of locally advanced breast cancer. J. Transl. Med. 10, 42 (2012).
Zhu, W., Qin, W., Atasoy, U. & Sauter, E. R. Circulating microRNAs in breast cancer and healthy subjects. BMC Res. Notes 2, 89 (2009).
Zen, K. & Zhang, C. Y. Circulating microRNAs: a novel class of biomarkers to diagnose and monitor human cancers. Med. Res. Rev. 32, 326–348 (2012).
Redis, R. S., Calin, S., Yang, Y., You, M. J. & Calin, G. A. Cell-to-cell miRNA transfer: from body homeostasis to therapy. Pharmacol. Ther. 136, 169–174 (2012).
Valadi, H. et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell Biol. 9, 654–659 (2007).
Wang, K., Zhang, S., Weber, J., Baxter, D. & Galas, D. J. Export of microRNAs and microRNA-protective protein by mammalian cells. Nucleic Acids Res. 38, 7248–7259 (2010).
Vickers, K. C. & Remaley, A. T. Lipid-based carriers of microRNAs and intercellular communication. Curr. Opin. Lipidol. 23, 91–97 (2012).
Skog, J. et al. Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nat. Cell Biol. 10, 1470–1476 (2008).
Peinado, H. et al. Melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through MET. Nat. Med. 18, 883–891 (2012).
Zhou, W. et al. Cancer-secreted miR-105 destroys vascular endothelial barriers to promote metastasis. Cancer Cell 25, 501–515 (2014).
Rottiers, V. & Naar, A. M. MicroRNAs in metabolism and metabolic disorders. Nat. Rev. Mol. Cell Biol. 13, 239–250 (2012).
Moore, K. J., Rayner, K. J., Suarez, Y. & Fernandez-Hernando, C. The role of microRNAs in cholesterol efflux and hepatic lipid metabolism. Annu. Rev. Nutr. 31, 49–63 (2011).
Boutz, D. R. et al. Two-tiered approach identifies a network of cancer and liver disease-related genes regulated by miR-122. J. Biol. Chem. 286, 18066–18078 (2011).
Palma, J. et al. MicroRNAs are exported from malignant cells in customized particles. Nucleic Acids Res. 40, 9125–9138 (2012).
Ashby, J. et al. Distribution profiling of circulating MicroRNAs in serum. Anal. Chem. 86, 9343–9349 (2014).
Lloyd, P. G., Hardin, C. D. & Sturek, M. Examining glucose transport in single vascular smooth muscle cells with a fluorescent glucose analog. Physiol. Res. 48, 401–410 (1999).
Loaiza, A., Porras, O. H. & Barros, L. F. Glutamate triggers rapid glucose transport stimulation in astrocytes as evidenced by real-time confocal microscopy. J. Neurosci. 23, 7337–7342 (2003).
Yamada, K. et al. Measurement of glucose uptake and intracellular calcium concentration in single, living pancreatic β-cells. J. Biol. Chem. 275, 22278–22283 (2000).
Itoh, Y., Abe, T., Takaoka, R. & Tanahashi, N. Fluorometric determination of glucose utilization in neurons in vitro and in vivo. J. Cereb. Blood Flow Metab. 24, 993–1003 (2004).
Chuquet, J., Quilichini, P., Nimchinsky, E. A. & Buzsaki, G. Predominant enhancement of glucose uptake in astrocytes versus neurons during activation of the somatosensory cortex. J. Neurosci. 30, 15298–15303 (2010).
Bak, L. K., Schousboe, A., Sonnewald, U. & Waagepetersen, H. S. Glucose is necessary to maintain neurotransmitter homeostasis during synaptic activity in cultured glutamatergic neurons. J. Cereb. Blood Flow Metab. 26, 1285–1297 (2006).
Hu, M. et al. Regulation of in situ to invasive breast carcinoma transition. Cancer Cell 13, 394–406 (2008).
Vickers, K. C., Palmisano, B. T., Shoucri, B. M., Shamburek, R. D. & Remaley, A. T. MicroRNAs are transported in plasma and delivered to recipient cells by high-density lipoproteins. Nat. Cell Biol. 13, 423–433 (2011).
Arroyo, J. D. et al. Argonaute2 complexes carry a population of circulating microRNAs independent of vesicles in human plasma. Proc. Natl Acad. Sci. USA 108, 5003–5008 (2011).
Pavlides, S. et al. The reverse Warburg effect: aerobic glycolysis in cancer associated fibroblasts and the tumor stroma. Cell Cycle 8, 3984–4001 (2009).
Martinez-Outschoorn, U. E., Lisanti, M. P. & Sotgia, F. Catabolic cancer-associated fibroblasts transfer energy and biomass to anabolic cancer cells, fueling tumor growth. Semin. Cancer Biol. 25, 47–60 (2014).
Kaplan, R. N., Psaila, B. & Lyden, D. Bone marrow cells in the ‘pre-metastatic niche’: within bone and beyond. Cancer Metastasis Rev. 25, 521–529 (2006).
Kaplan, R. N. et al. VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature 438, 820–827 (2005).
Peinado, H., Lavotshkin, S. & Lyden, D. The secreted factors responsible for pre-metastatic niche formation: old sayings and new thoughts. Semin. Cancer Biol. 21, 139–146 (2011).
Sethi, N. & Kang, Y. Unravelling the complexity of metastasis - molecular understanding and targeted therapies. Nat. Rev. Cancer 11, 735–748 (2011).
Hood, J. L., San, R. S. & Wickline, S. A. Exosomes released by melanoma cells prepare sentinel lymph nodes for tumor metastasis. Cancer Res. 71, 3792–3801 (2011).
Grange, C. et al. Microvesicles released from human renal cancer stem cells stimulate angiogenesis and formation of lung premetastatic niche. Cancer Res. 71, 5346–5356 (2011).
Erler, J. T. et al. Hypoxia-induced lysyl oxidase is a critical mediator of bone marrow cell recruitment to form the premetastatic niche. Cancer Cell 15, 35–44 (2009).
Hiratsuka, S. et al. Primary tumours modulate innate immune signalling to create pre-metastatic vascular hyperpermeability foci. Nat. Commun. 4, 1853 (2013).
Hiratsuka, S. et al. Endothelial focal adhesion kinase mediates cancer cell homing to discrete regions of the lungs via E-selectin up-regulation. Proc. Natl Acad. Sci. USA 108, 3725–3730 (2011).
Hiratsuka, S., Watanabe, A., Aburatani, H. & Maru, Y. Tumour-mediated upregulation of chemoattractants and recruitment of myeloid cells predetermines lung metastasis. Nat. Cell Biol. 8, 1369–1375 (2006).
Hiratsuka, S. et al. The S100A8-serum amyloid A3-TLR4 paracrine cascade establishes a pre-metastatic phase. Nat. Cell Biol. 10, 1349–1355 (2008).
Bos, R. et al. Biologic correlates of 18fluorodeoxyglucose uptake in human breast cancer measured by positron emission tomography. J. Clin. Oncol. 20, 379–387 (2002).
Kang, S. S. et al. Clinical significance of glucose transporter 1 (GLUT1) expression in human breast carcinoma. Jpn J. Cancer Res. 93, 1123–1128 (2002).
Elmen, J. et al. Antagonism of microRNA-122 in mice by systemically administered LNA-antimiR leads to up-regulation of a large set of predicted target mRNAs in the liver. Nucleic Acids Res. 36, 1153–1162 (2008).
Elmen, J. et al. LNA-mediated microRNA silencing in non-human primates. Nature 452, 896–899 (2008).
Takanaga, H. & Frommer, W. B. Facilitative plasma membrane transporters function during ER transit. FASEB J. 24, 2849–2858 (2010).
Wang, Y. et al. Transforming growth factor-β regulates the sphere-initiating stem cell-like feature in breast cancer through miRNA-181 and ATM. Oncogene 30, 1470–1480 (2011).
Thery, C., Amigorena, S., Raposo, G. & Clayton, A. Isolation and characterization of exosomes from cell culture supernatants and biological fluids. Curr. Protoc. Cell Biol. (2006).
Tsuyada, A. et al. CCL2 mediates cross-talk between cancer cells and stromal fibroblasts that regulates breast cancer stem cells. Cancer Res. 72, 2768–2779 (2012).
Yu, Y. et al. Context-dependent bidirectional regulation of the MutS homolog 2 by transforming growth factor beta contributes to chemoresistance in breast cancer cells. Mol. Cancer Res. 8, 1633–1642 (2010).
Clower, C. V. et al. The alternative splicing repressors hnRNP A1/A2 and PTB influence pyruvate kinase isoform expression and cell metabolism. Proc. Natl Acad. Sci. USA 107, 1894–1899 (2010).
Edwards, R. J. & Watts, D. C. Specific spectrophotometric assay for the M isoenzymes of pyruvate kinase in plasma samples containing mixtures of the muscle (M) and liver (L) isoenzymes. Clin. Chem. 27, 906–909 (1981).
Cano, K. E., Li, Y. J. & Chen, Y. NMR metabolomic profiling reveals new roles of SUMOylation in DNA damage response. J. Proteome Res. 9, 5382–5388 (2010).
Chida, J., Yamane, K., Takei, T. & Kido, H. An efficient extraction method for quantitation of adenosine triphosphate in mammalian tissues and cells. Anal. Chim. Acta 727, 8–12 (2012).
Fong, M. Y. et al. Withaferin a synergizes the therapeutic effect of doxorubicin through ROS-mediated autophagy in ovarian cancer. PLoS ONE 7, e42265 (2012).
Jorgensen, S., Baker, A., Moller, S. & Nielsen, B. S. Robust one-day in situ hybridization protocol for detection of microRNAs in paraffin samples using LNA probes. Methods 52, 375–381 (2010).
Acknowledgements
This work was supported by the United States Army Research and Material Command grant W81-14-1-0029 (M.Y.F.), National Institutes of Health (NIH)/National Cancer Institute (NCI) grants R01CA166020 (S.E.W.) and R01CA163586 (S.E.W.), California Breast Cancer Research Program grant 20IB-0118 (S.E.W.), Breast Cancer Research Foundation-AACR grant 12-60-26-WANG (S.E.W.) and the City of Hope Women’s Cancer Program. Research reported here includes work carried out in Core Facilities supported by the NIH/NCI under grant number P30CA33572. We thank A. Riggs, E. Roberts, L. Malkas, S. Kane, S. Chen, J. Mortimer and P. Sarnow for valuable comments, as well as the Core Facilities at City of Hope for services.
Author information
Authors and Affiliations
Contributions
S.E.W. conceived ideas, and M.Y.F., Y.C. and X.R. contributed to project planning. M.Y.F. and S.E.W. designed and carried out the experiments. W.Zhou, L.L., A.C., S.T.F.O., S.L., A.R.C. and J.R.T. assisted with EV preparation and mouse experiments. G.Somlo and M.P. assisted with patient serum samples. Z.L. assisted with electron microscopy. A.T. assisted with mouse lung fibroblast culture. A.Y.A., M.C. and Y.C. assisted with NMR analysis. J.A. and W.Zhong assisted with AF4 analysis. G.Sun and Y.S. assisted with neuron culture. M.A.R. and M.K. assisted with medium metabolite analysis. X.W. assisted with bioinformatics analysis of miR-122 targets. P.S. assisted with anti-miR-122 and mismatch oligonucleotide synthesis. S.E.W. and M.Y.F. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
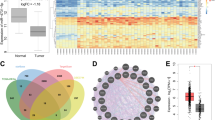
Supplementary Figure 2 Characterization of the 110,000 × g medium pellet.
(a) For each cell line, RNA were extracted from the 110,000 × g medium pellet and concentrated supernatant obtained from equivalent volume of CM and analysed for miR-122 and miR-16 by RT-qPCR. Absolute miRNA levels are calculated based on standard curves (n = 6 extracts). (b) Representative EM images of vesicles in the AF4 fraction eluted at 18–25 min. The measured diameter of vesicles was shown as mean ± s.d. (n = 126 for MCF10A/vec-derived vesicles; n = 222 for MCF10A/miR-122-derived vesicles; n = 41 for MDA-MB-231-derived vesicles). Bars equal 100 nm. (c) RT-qPCR-determined levels of miRNAs in MCF10A/vec- and MCF10A/miR-122-derived protein and vesicle fractions separated by AF4. Absolute miRNA levels are shown (n = 6 extracts). ND: not detected. (d) Absolute miRNA levels in each gradient fraction after sucrose gradient centrifugation of MCF10A/miR-122-derived 110,000 × g medium pellet were determined by RT-qPCR (n = 6 extracts). (e) A representative EM image of MCF10A/miR-122-derived vesicles in sucrose fraction 6 (F6) (n = 362 vesicles). Bar equals 100 nm. ∗P < 0.05 for all panels derived from Kruskal–Wallis test. Data are represented as mean ± s.d. in all panels except (b,e).
Supplementary Figure 3 Examples of peak assignment based on NMR spectra.
For all spectrum snapshots, black represents MCF10A/vec, red represents MCF10A/miR-122, and blue represents library entry. In general, each metabolite produces multiple resonances in different regions of the spectrum. We only analysed those with some of the resonances that can be resolved unambiguously. An example is given by glucose as shown in (a). One of the glucose resonances overlaps with that of O-acetylcholine, lactose, glutathione, homoserine and ethanolamine at around 3.8 ppm, but another glucose resonance is well resolved at around 3.4 ppm. Another example is given by lactate that has peaks in two regions of the spectrum that do not overlap with other resonances (b). The unique patterns of NMR resonances, due to J-coupling, are used for de-convolution of overlapping resonances. An example is given in (c), which shows that one set of peaks of isocitrate overlaps with that of glutathione, but the patterns of the peaks from the two metabolites are different, and at least one peak of the isocitrate resonance can be well resolved for de-convolution to extract the concentrations. Similarly, the pyruvate peak can be well resolved from those of glutamate (d). Examples of assignments of some other resolved peaks are given in (e).
Supplementary Figure 4 Characterization of cell lines with modified miR-122 levels used for in vivo studies.
(a) Cell number counts of MCFDCIS/miR-122 and MCFDCIS/vec cells at indicated time points (n = 6 biological replicates). (b) Medium metabolite analysis after 48 h of culture (n = 6 biological replicates). (c) RT-qPCR analysis showing the relative expression of indicated genes in MCFDCIS/miR-122 and MCFDCIS/vec cells (n = 6 extracts). (d) Western blot analysis in MCFDCIS/miR-122 and MCFDCIS/vec cells. Size of markers (in kDa) are indicated. (e) RT-qPCR-determined levels of intracellular and secreted miR-122 as well as PKM expression in MDA-MB-231 cells with stable knockdown of miR-122 (MDA-MB-231/122KD) compared to the control cells (n = 6 extracts).∗P < 0.05 for all panels derived from Kruskal–Wallis test. Data are represented as mean ± s.d. in all panels except (d). Uncropped, unprocessed images of blots are shown in Supplementary Fig. 5.
Supplementary Figure 5 BLI of the primary tumours established with MDA-MB-231-HM and treated with anti-miR-122 oligos.
(a) BLI images at week 3. (b) Quantification of (a) using Living Image Software (n = 8 mice per group). No significant difference (P > 0.05) between groups based on Kruskal–Wallis test.
Rights and permissions
About this article
Cite this article
Fong, M., Zhou, W., Liu, L. et al. Breast-cancer-secreted miR-122 reprograms glucose metabolism in premetastatic niche to promote metastasis. Nat Cell Biol 17, 183–194 (2015). https://doi.org/10.1038/ncb3094
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb3094
This article is cited by
-
Tumor-derived small extracellular vesicles in cancer invasion and metastasis: molecular mechanisms, and clinical significance
Molecular Cancer (2024)
-
Exosomal circRHCG promotes breast cancer metastasis via facilitating M2 polarization through TFEB ubiquitination and degradation
npj Precision Oncology (2024)
-
Metabolic heterogeneity affects trastuzumab response and survival in HER2-positive advanced gastric cancer
British Journal of Cancer (2024)
-
Tumor Cell-Derived Exosomal miR-191-5p Activates M2-Subtype Macrophages Through SOCS3 to Facilitate Breast Cancer
Molecular Biotechnology (2024)
-
Lean adipose tissue macrophage derived exosome confers immunoregulation to improve wound healing in diabetes
Journal of Nanobiotechnology (2023)