Abstract
The precise relationship of embryonic stem cells (ESCs) to cells in the mouse embryo remains controversial. We present transcriptional and functional data to identify the embryonic counterpart of ESCs. Marker profiling shows that ESCs are distinct from early inner cell mass (ICM) and closely resemble pre-implantation epiblast. A characteristic feature of mouse ESCs is propagation without ERK signalling. Single-cell culture reveals that cell-autonomous capacity to thrive when the ERK pathway is inhibited arises late during blastocyst development and is lost after implantation. The frequency of deriving clonal ESC lines suggests that all E4.5 epiblast cells can become ESCs. We further show that ICM cells from early blastocysts can progress to ERK independence if provided with a specific laminin substrate. These findings suggest that formation of the epiblast coincides with competence for ERK-independent self-renewal in vitro and consequent propagation as ESC lines.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Accession codes
References
Nichols, J. et al. Formation of pluripotent stem cells in the mammalian embryo depends on the POU transcription factor Oct4. Cell 95, 379–391 (1998).
Chambers, I. et al. Functional expression cloning of Nanog, a pluripotency sustaining factor in embryonic stem cells. Cell 113, 643–655 (2003).
Kurimoto, K. et al. An improved single-cell cDNA amplification method for efficient high-density oligonucleotide microarray analysis. Nucleic Acids Res. 34, e42 (2006).
Guo, G. et al. Resolution of cell fate decisions revealed by single-cell gene expression analysis from zygote to blastocyst. Dev. Cell 18, 675–685 (2010).
Avilion, A. A. et al. Multipotent cell lineages in early mouse development depend on SOX2 function. Genes Dev. 17, 126–140 (2003).
Artus, J., Piliszek, A. & Hadjantonakis, A. K. The primitive endoderm lineage of the mouse blastocyst: sequential transcription factor activation and regulation of differentiation by Sox17. Dev. Biol. 350, 393–404 (2011).
Frankenberg, S. et al. Primitive endoderm differentiates via a three-step mechanism involving Nanog and RTK signaling. Dev. Cell 21, 1005–1013 (2011).
Chazaud, C., Yamanaka, Y., Pawson, T. & Rossant, J. Early lineage segregation between epiblast and primitive endoderm in mouse blastocysts through the Grb2-MAPK pathway. Dev. Cell 10, 615–624 (2006).
Morrisey, E. E. et al. GATA6 regulates HNF4 and is required for differentiation of visceral endoderm in the mouse embryo. Genes Dev. 12, 3579–3590 (1998).
Niakan, K. K. et al. Sox17 promotes differentiation in mouse embryonic stem cells by directly regulating extraembryonic gene expression and indirectly antagonizing self-renewal. Genes Dev. 24, 312–326 (2010).
Koutsourakis, M., Langeveld, A., Patient, R., Beddington, R. & Grosveld, F. The transcription factor GATA6 is essential for early extraembryonic development. Development 126, 723–732 (1999).
Evans, M. J. & Kaufman, M. H. Establishment in culture of pluripotential cells from mouse embryos. Nature 292, 154–156 (1981).
Martin, G. R. Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc. Natl Acad. Sci. USA 78, 7634–7638 (1981).
Williams, R. L. et al. Myeloid leukaemia inhibitory factor maintains the developmental potential of embryonic stem cells. Nature 336, 684–687 (1988).
Smith, A. G. et al. Inhibition of pluripotential embryonic stem cell differentiation by purified polypeptides. Nature 336, 688–690 (1988).
Silva, J. & Smith, A. Capturing pluripotency. Cell 132, 532–536 (2008).
Nichols, J. & Smith, A. Naive and primed pluripotent states. Cell Stem Cell 4, 487–492 (2009).
Kunath, T. et al. FGF stimulation of the Erk1/2 signalling cascade triggers transition of pluripotent embryonic stem cells from self-renewal to lineage commitment. Development 134, 2895–2902 (2007).
Burdon, T., Stracey, C., Chambers, I., Nichols, J. & Smith, A. Suppression of SHP-2 and ERK signalling promotes self-renewal of mouse embryonic stem cells. Dev. Biol. 210, 30–43 (1999).
Ying, Q. L. et al. The ground state of embryonic stem cell self-renewal. Nature 453, 519–523 (2008).
Wray, J. et al. Inhibition of glycogen synthase kinase-3 alleviates Tcf3 repression of the pluripotency network and increases embryonic stem cell resistance to differentiation. Nat. Cell Biol. 13, 838–845 (2011).
Brons, I. G. et al. Derivation of pluripotent epiblast stem cells from mammalian embryos. Nature 448, 191–195 (2007).
Tesar, P. J. et al. New cell lines from mouse epiblast share defining features with human embryonic stem cells. Nature 448, 196–199 (2007).
Guo, G. et al. Klf4 reverts developmentally programmed restriction of ground state pluripotency. Development 136, 1063–1069 (2009).
Silva, J. et al. Promotion of reprogramming to ground state pluripotency by signal inhibition. PLoS Biol. 6, e253 (2008).
Theunissen, T. W. et al. Nanog overcomes reprogramming barriers and induces pluripotency in minimal conditions. Curr. Biol. 21, 65–71 (2011).
Nichols, J. et al. Validated germline-competent embryonic stem cell lines from nonobese diabetic mice. Nat. Med. 15, 814–818 (2009).
Hanna, J. et al. Metastable pluripotent states in NOD-mouse-derived ESCs. Cell Stem Cell 4, 513–524 (2009).
Buehr, M. et al. Capture of authentic embryonic stem cells from rat blastocysts. Cell 135, 1287–1298 (2008).
Li, P. et al. Germline competent embryonic stem cells derived from rat blastocysts. Cell 135, 1299–1310 (2008).
Tesar, P. J. Derivation of germ-line-competent embryonic stem cell lines from preblastocyst mouse embryos. Proc. Natl Acad. Sci. USA 102, 8239–8244 (2005).
Delhaise, F., Bralion, V., Schuurbiers, N. & Dessy, F. Establishment of an embryonic stem cell line from 8-cell stage mouse embryos. Eur. J. Morphol. 34, 237–243 (1996).
Chung, Y. et al. Embryonic and extraembryonic stem cell lines derived from single mouse blastomeres. Nature 439, 216–219 (2006).
Wakayama, S. et al. Efficient establishment of mouse embryonic stem cell lines from single blastomeres and polar bodies. Stem Cells 25, 986–993 (2007).
Bao, S. et al. Epigenetic reversion of post-implantation epiblast to pluripotent embryonic stem cells. Nature 461, 1292–1295 (2009).
Macfarlan, T. S. et al. Embryonic stem cell potency fluctuates with endogenous retrovirus activity. Nature 487, 57–63 (2012).
Tang, F. et al. RNA-Seq analysis to capture the transcriptome landscape of a single-cell. Nat. Protoc. 5, 516–535 (2010).
Tang, F. et al. Tracing the derivation of embryonic stem cells from the inner cell mass by single-cell RNA-Seq analysis. Cell Stem Cell 6, 468–478 (2010).
Solter, D. & Knowles, B. B. Immunosurgery of mouse blastocyst. Proc. Natl Acad. Sci. USA 72, 5099–5102 (1975).
Plusa, B., Piliszek, A., Frankenberg, S., Artus, J. & Hadjantonakis, A. K. Distinct sequential cell behaviours direct primitive endoderm formation in the mouse blastocyst. Development 135, 3081–3091 (2008).
Hamilton, T. G., Klinghoffer, R. A., Corrin, P. D. & Soriano, P. Evolutionary divergence of platelet-derived growth factor alpha receptor signaling mechanisms. Mol. Cell Biol. 23, 4013–4025 (2003).
He, B. et al. Cloning and characterization of a functional promoter of the human SOCS-3 gene. Biochem. Biophys. Res. Commun. 301, 386–391 (2003).
Bourillot, P. Y. et al. Novel STAT3 target genes exert distinct roles in the inhibition of mesoderm and endoderm differentiation in cooperation with Nanog. Stem Cells 27, 1760–1771 (2009).
Coucouvanis, E. & Martin, G. R. BMP signaling plays a role in visceral endoderm differentiation and cavitation in the early mouse embryo. Development 126, 535–546 (1999).
Guzman-Ayala, M. et al. Graded Smad2/3 activation is converted directly into levels of target gene expression in embryonic stem cells. PLoS ONE 4, e4268 (2009).
Marks, H. et al. The transcriptional and epigenomic foundations of ground state pluripotency. Cell 149, 590–604 (2012).
Wray, J., Kalkan, T. & Smith, A. G. The ground state of pluripotency. Biochem. Soc. Trans. 38, 1027–1032 (2010).
Lorthongpanich, C., Doris, T. P., Limviphuvadh, V., Knowles, B. B. & Solter, D. Developmental fate and lineage commitment of singled mouse blastomeres. Development 139, 3722–3731 (2012).
Nichols, J., Silva, J., Roode, M. & Smith, A. Suppression of Erk signalling promotes ground state pluripotency in the mouse embryo. Development 136, 3215–3222 (2009).
Yamanaka, Y., Lanner, F. & Rossant, J. FGF signal-dependent segregation of primitive endoderm and epiblast in the mouse blastocyst. Development 137, 715–724 (2010).
Roode, M. et al. Human hypoblast formation is not dependent on FGF signalling. Dev. Biol. 361, 358–363 (2012).
Grabarek, J. B. et al. Differential plasticity of epiblast and primitive endoderm precursors within the ICM of the early mouse embryo. Development 139, 129–139 (2012).
Gardner, R. L. & Rossant, J. Investigation of the fate of 4–5 day post-coitum mouse inner cell mass cells by blastocyst injection. J. Embryol. Exp. Morphol. 52, 141–152 (1979).
Hall, J. et al. Oct4 and LIF/Stat3 additively induce Kruppel factors to sustain embryonic stem cell self-renewal. Cell Stem Cell 5, 597–609 (2009).
Martello, G., Bertone, P. & Smith, A. Identification of the missing pluripotency factor downstream of leukaemia inhibitory factor. EMBO J. 2561–2574 (2013).
Martello, G. et al. Esrrb is a pivotal target of the Gsk3/Tcf3 axis regulating embryonic stem cell self-renewal. Cell Stem Cell 11, 491–504 (2012).
Nichols, J. & Smith, A. Pluripotency in the embryo and in culture. Cold Spring Harb. Perspect. Biol. 4, a008128 (2012).
Pelton, T. A., Sharma, S., Schulz, T. C., Rathjen, J. & Rathjen, P. D. Transient pluripotent cell populations during primitive ectoderm formation: correlation of in vivo and in vitro pluripotent cell development. J. Cell Sci. 115, 329–339 (2002).
Chambers, I. et al. Nanog safeguards pluripotency and mediates germline development. Nature 450, 1230–1234 (2007).
Hayashi, K., Lopes, S. M., Tang, F. & Surani, M. A. Dynamic equilibrium and heterogeneity of mouse pluripotent stem cells with distinct functional and epigenetic states. Cell Stem Cell 3, 391–401 (2008).
Toyooka, Y., Shimosato, D., Murakami, K., Takahashi, K. & Niwa, H. Identification and characterization of subpopulations in undifferentiated ES cell culture. Development 135, 909–918 (2008).
Smith, A. Nanog heterogeneity: tilting at windmills? Cell Stem Cell 13, 6–7 (2013).
Brook, F. A. & Gardner, R. L. The origin and efficient derivation of embryonic stem cells in the mouse. Proc. Natl Acad. Sci. USA 94, 5709–5712 (1997).
Nichols, J. & Smith, A. The origin and identity of embryonic stem cells. Development 138, 3–8 (2011).
Zwaka, T. P. & Thomson, J. A. A germ cell origin of embryonic stem cells? Development 132, 227–233 (2005).
Chu, L. F., Surani, M. A., Jaenisch, R. & Zwaka, T. P. Blimp1 expression predicts embryonic stem cell development in vitro. Curr. Biol. 21, 1759–1765 (2011).
Ohinata, Y. et al. Blimp1 is a critical determinant of the germ cell lineage in mice. Nature 436, 207–213 (2005).
Vincent, S. D. et al. The zinc finger transcriptional repressor Blimp1/Prdm1 is dispensable for early axis formation but is required for specification of primordial germ cells in the mouse. Development 132, 1315–1325 (2005).
Domogatskaya, A., Rodin, S., Boutaud, A. & Tryggvason, K. Laminin-511 but not −332, −111, or −411 enables mouse embryonic stem cell self-renewal in vitro. Stem Cells 26, 2800–2809 (2008).
Smyth, N. et al. Absence of basement membranes after targeting the LAMC1 gene results in embryonic lethality due to failure of endoderm differentiation. J. Cell Biol. 144, 151–160 (1999).
Miner, J. H., Li, C., Mudd, J. L., Go, G. & Sutherland, A. E. Compositional and structural requirements for laminin and basement membranes during mouse embryo implantation and gastrulation. Development 131, 2247–2256 (2004).
Keely, P. J. Mechanisms by which the extracellular matrix and integrin signaling act to regulate the switch between tumor suppression and tumor promotion. J. Mammary Gland Biol. Neoplasia 16, 205–219 (2011).
Fassler, R. & Meyer, M. Consequences of lack of beta 1 integrin gene expression in mice. Genes Dev. 9, 1896–1908 (1995).
Bi, L., Okabe, I., Bernard, D. J. & Nussbaum, R. L. Early embryonic lethality in mice deficient in the p110beta catalytic subunit of PI 3-kinase. Mamm. Genome 13, 169–172 (2002).
Hooper, M., Hardy, K., Handyside, A., Hunter, S. & Monk, M. HPRT-deficient (Lesch-Nyhan) mouse embryos derived from germline colonization by cultured cells. Nature 326, 292–295 (1987).
Haapa-Paananen, S. et al. HES6 gene is selectively overexpressed in glioma and represents an important transcriptional regulator of glioma proliferation. Oncogene 31, 1299–1310 (2012).
Tang, F. et al. mRNA-Seq whole-transcriptome analysis of a single-cell. Nat. Methods 6, 377–382 (2009).
Dvinge, H. & Bertone, P. HTqPCR: high-throughput analysis and visualization of quantitative real-time PCR data in R. Bioinformatics 25, 3325–3326 (2009).
Mamo, S., Gal, A. B., Bodo, S. & Dinnyes, A. Quantitative evaluation and selection of reference genes in mouse oocytes and embryos cultured in vivo and in vitro. BMC Dev. Biol. 7, 14 (2007).
Aird, D. et al. Analyzing and minimizing PCR amplification bias in Illumina sequencing libraries. Genome Biol. 12, R18 (2011).
Wu, T. D. & Nacu, S. Fast and SNP-tolerant detection of complex variants and splicing in short reads. Bioinformatics 26, 873–881 (2010).
Flicek, P. et al. Ensembl 2013. Nucleic Acids Res. 41, D48–D55 (2013).
Acknowledgements
We thank C-É Dumeau and W. Mansfield for chimaera production, P. Humphreys for assistance with imaging, S. Jameson and staff for animal husbandry, the EMBL Genomics Core Facility for sequencing, and G. Martello for helpful discussion on the manuscript. This work was financially supported by the Wellcome Trust, Medical Research Council, BBSRC, the Louis Jeantet Foundation, EMBL and the University of Cambridge. A.S. is a Medical Research Council Professor.
Author information
Authors and Affiliations
Contributions
T.B. and J.N. performed embryology and wet laboratory experiments; R.L. and P.B. were responsible for bioinformatics and data analysis. A.S. and J.N. conceived the project and all authors contributed to preparation of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
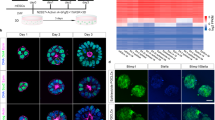
Supplementary Figure 1 Increasing the amount of starting material improves the fidelity of whole-transcriptome preamplification.
(A) To evaluate the accuracy of whole-transcriptome preamplification37 from different amounts of starting material, total RNA from 2i and 2i-LIF ESC cultures was diluted to 200pg (equivalent to 10–20 cells) and 20pg (equivalent to 1–2 cells) and subsequently preamplified using 18, 20 and 22 preamplification cycles (2 technical replicates each). qPCR of conventional cDNA indicates that ESC respond to LIF by upregulation of Socs3 and Klf4, consistent with previous observations, while levels of other pluripotency factors did not change43. On average, preamplified samples corresponding to 10–20 cells recapitulated the expression pattern of the unamplified control more faithfully than preamplified samples equivalent to 1–2 cells. Error bars are s.d. between biological replicates. (B) Correlation of diluted and subsequently preamplified RNA of ESC in 2i-LIF compared to conventional cDNA of the same RNA. (C) Heatmaps of RNA levels of Notch and p38 signalling genes normalised to mean expression in embryonic samples E1.5–5.5. (D) Hierarchical clustering of embryonic samples from E1.5–5.5.
Supplementary Figure 2 ESC cultured in 2i and 2i-LIFcorrelate with the E4.5 epiblast based on 96 gene qRT-PCR arrays.
Correlation analysis between embryonic samples and ESC samples obtained from 2i and 2i-LIF cultures on gelatin. For the ESC samples, two independent biological replicates, each containing 20 randomly picked cells of the 4 ESC lines indicated in the graph, for both 2i and 2i-LIF, were processed and profiled by qRT-PCR arrays.
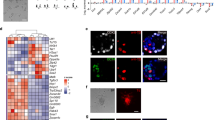
Supplementary Figure 3 Early preimplantation stages progress in development during ESC derivation.
(A) Images of typical primary ESC colonies derived in 2i-LIF on gelatin from an E2.5 morula and the ICM of an E3.5 blastocyst after 6 and 5 days, respectively. (B) ESC derivation efficiencies from various developmental stages assayed by primary ES colony formation, as depicted in A. The numbers of embryos yielding an ESC colony per embryo are indicated. (C) Blastocoel formation of a morula 24 h after plating in 2i-LIF on gelatin. (D) PrE-like epithelial formation in an outgrowth from an E2.5 morula 4 days after plating in 2i-LIF on gelatin. We suspect that partial PrE specification is mediated by compensatory effects of LIF in 2i. (E) Surviving cells as judged by morphology at day 7 in percentage of wells per embryo after single ESC derivation in 2i on gelatin. The graph shows the mean and s.e.m. for the percentage of surviving cell-containing wells per embryo resulting from the analysis of n = ‘e’ embryos (with at least 3 wells analysed per embryo for each time point of embryonic development shown on the x axis, ‘w’ represents the total number of wells analysed per time point). (F) Images of surviving cells 7 days after single cell ESC derivation from E3.5 embryos in 2i-LIF on gelatin. (G,H) Single cell ESC derivation efficiency in serum-LIF (G) and under EpiSC conditions (ActA, bFGF) (H); Efficiency is displayed as the percentage of ESC-colony positive wells per embryo (ESC-col. pos. wells (%)/embryo) after 7 days. The total efficiency is further subdivided into ESC colonies arising from truly individual cells (ESC-col (single cell)) and ESC arising from small groups, usually between 2 and 5 cells (ESC-col (small group of cells)). The graphs show the mean and s.e.m. for the percentage of ESC colony-containing wells per embryo resulting from the analysis of n = ‘e’ embryos (with at least 3 wells analysed per embryo for each time point of embryonic development shown on the x axis, ‘w’ represents the total number of wells analysed per time point). Embryos were obtained from at least 3 individual litters on at least 2 different days.
Supplementary Figure 4 TE marker profiling of vacuolated cells.
Relative RNA levels of trophectoderm specific genes for the samples indicated. Error bars are s.d. between biological replicates.
Supplementary Figure 5 Correlation to previously published data and testing extracellular matrix components with stringent early ICM cells.
(A) Correlation of RNA levels as determined by RNA-seq of the E3.5-ICM samples analysed in this study. (B, C) Correlation of RNA levels as determined by RNA-seq of the E3.5-ICM samples 1 (B) and 2 (C) of the present study to the average of 9 individually processed and sequenced E3.5 ICM cells in Tang et al.38. (D) Average RNA levels at E3.5 for the same extracellular matrix genes shown in Fig5.B, as determined in Tang et al.by single-cell RNA-seq. (E) Single cell ESC derivation efficiency of stringent early ICM cells with a 24 h maturation step for the experimental conditions indicated on Fn-Lam511. The graph shows the mean and s.e.m. for the percentage of ESC-colony containing wells per embryo resulting from the analysis of n = ‘e’ embryos (with at least 4 wells analysed per embryo for each experimental condition shown on the x axis, ‘w’ represents the total number of wells analysed per experimental condition).
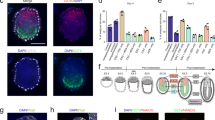
Supplementary Figure 6 Characterisation of Fn-Lam511 ES cells.
(A) Alkaline phosphatase staining of ESC colonies and PrE-like cells at day 7 after single ESC derivation in 2i-LIF on Fn-Lam511. (B, C) Immunofluorescence staining of ESC colonies and PrE-like cells at days 3 and 6 (B) and at day 7 (C) from single cell ESC derivation in 2i-LIF on Fn-Lam511. (D) Phase contrast images of the first 5 clonal ESC lines at passage 4 originally derived on either Fn-Lam511 (FL), Fibronectin (F) and Laminin511 (L). After manually picking and dissociating primary single cell ESC colonies at day 7, ESC lines were expanded in 2i-LIF on gelatin. Two of the FL lines were randomly selected (FL4 and FL11) and successfully tested for germline transmission. (E) PrE-like cells obtained after single cell ESC derivation in 2i-LIF on Fn-Lam511 of embryos cultured in the presence of FGF2 for 3 days from the 8-cell stage.
Supplementary Figure 7 Expression dynamics of Esrrb and Tcfcp2l1 and correlation analysis of primary ESC colonies to embryonic samples.
(A) Direct comparison of RNA levels in embryonic samples E3.5-ICM, E4.0-ICM and E4.5-EPI (epiblast), single cell ESC derivation samples day 2–6 and established ESC in 2i-LIF. Expression levels are shown relative to the preimplantation epiblast (E4.5-EPI) on a logarithmic scale for Esrrb and Tcfcp2l1. Error bars are s.d. between biological replicates. (B) Correlation analysis between embryonic samples, established ESC samples and primary ESC colony day 2, 4 and 6 samples. Samples were processed as described in Fig. 7c.
Supplementary information
Supplementary Information
Supplementary Information (PDF 1674 kb)
Supplementary Table 1
Supplementary Information (XLSX 18 kb)
Supplementary Table 2
Supplementary Information (XLSX 80 kb)
Supplementary Table 3
Supplementary Information (XLSX 19 kb)
Time-lapse imaging of single cell ESC derivation from E4.5 Pdgfra::GFP ICM.
Live imaging of a single cell isolated from an E4.5 ICM of a Pdgfra::GFP embryo during formation of an ESC colony. Green fluorescence identifies cells of the PrE lineage. In this example, a GFP-negative (epiblast) cell is filmed. (AVI 5306 kb)
Time-lapse imaging of single cell ESC derivation from E4.5 Rex1GFP ICM.
Live imaging of a single cell isolated from an E4.5 ICM of a Rex1GFP embryo during formation of an ESC colony. Green fluorescence marks cells of the epiblast lineage. The chosen cell and its progeny exhibit GFP expression throughout the movie. (AVI 16628 kb)
Rights and permissions
About this article
Cite this article
Boroviak, T., Loos, R., Bertone, P. et al. The ability of inner-cell-mass cells to self-renew as embryonic stem cells is acquired following epiblast specification. Nat Cell Biol 16, 513–525 (2014). https://doi.org/10.1038/ncb2965
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2965
This article is cited by
-
PRAMEL7 and CUL2 decrease NuRD stability to establish ground-state pluripotency
EMBO Reports (2024)
-
The BAF chromatin remodeler synergizes with RNA polymerase II and transcription factors to evict nucleosomes
Nature Genetics (2024)
-
Inhibition of TGF-β pathway improved the pluripotency of porcine pluripotent stem cells
In Vitro Cellular & Developmental Biology - Animal (2023)
-
Metabolic and cell cycle shift induced by the deletion of Dnm1l attenuates the dissolution of pluripotency in mouse embryonic stem cells
Cellular and Molecular Life Sciences (2023)
-
A hexa-species transcriptome atlas of mammalian embryogenesis delineates metabolic regulation across three different implantation modes
Nature Communications (2022)