Abstract
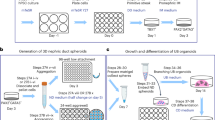
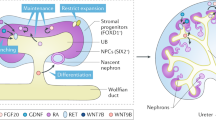
Diseases affecting the kidney constitute a major health issue worldwide. Their incidence and poor prognosis affirm the urgent need for the development of new therapeutic strategies. Recently, differentiation of pluripotent cells to somatic lineages has emerged as a promising approach for disease modelling and cell transplantation. Unfortunately, differentiation of pluripotent cells into renal lineages has demonstrated limited success. Here we report on the differentiation of human pluripotent cells into ureteric-bud-committed renal progenitor-like cells. The generated cells demonstrated rapid and specific expression of renal progenitor markers on 4-day exposure to defined media conditions. Further maturation into ureteric bud structures was accomplished on establishment of a three-dimensional culture system in which differentiated human cells assembled and integrated alongside murine cells for the formation of chimeric ureteric buds. Altogether, our results provide a new platform for the study of kidney diseases and lineage commitment, and open new avenues for the future application of regenerative strategies in the clinic.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Robinton, D. A. & Daley, G. Q. The promise of induced pluripotent stem cells in research and therapy. Nature 481, 295–305 (2012).
Spence, J. R. et al. Directed differentiation of human pluripotent stem cells into intestinal tissue in vitro. Nature 470, 105–109 (2011).
Lian, X. et al. Directed cardiomyocyte differentiation from human pluripotent stem cells by modulating Wnt/beta-catenin signaling under fully defined conditions. Nat. Protoc. 8, 162–175 (2013).
Osakada, F., Ikeda, H., Sasai, Y. & Takahashi, M. Stepwise differentiation of pluripotent stem cells into retinal cells. Nat. Protoc. 4, 811–824 (2009).
Wong, A. P. et al. Directed differentiation of human pluripotent stem cells into mature airway epithelia expressing functional CFTR protein. Nat. Biotechnol. 30, 876–882 (2012).
Eiraku, M. et al. Self-organizing optic-cup morphogenesis in three-dimensional culture. Nature 472, 51–56 (2011).
Dressler, G. R. The cellular basis of kidney development. Annu. Rev. Cell Dev. Biol. 22, 509–529 (2006).
Dressler, G. R. Advances in early kidney specification, development and patterning. Development 136, 3863–3874 (2009).
Costantini, F. & Kopan, R. Patterning a complex organ: branching morphogenesis and nephron segmentation in kidney development. Dev. Cell 18, 698–712 (2010).
Little, M. H. & McMahon, A. P. Mammalian kidney development: principles, progress, and projections. Cold Spring Harb Perspect Biol. 4, a008300 (2012).
Bouchard, M., Souabni, A., Mandler, M., Neubuser, A. & Busslinger, M. Nephric lineage specification by Pax2 and Pax8. Genes Dev. 16, 2958–2970 (2002).
Carroll, T. J. & Vize, P. D. Synergism between Pax-8 and lim-1 in embryonic kidney development. Dev. Biol. 214, 46–59 (1999).
Grote, D., Souabni, A., Busslinger, M. & Bouchard, M. Pax 2/8-regulated Gata 3 expression is necessary for morphogenesis and guidance of the nephric duct in the developing kidney. Development 133, 53–61 (2006).
Boyle, S. et al. Fate mapping using Cited1-CreERT2 mice demonstrates that the cap mesenchyme contains self-renewing progenitor cells and gives rise exclusively to nephronic epithelia. Dev. Biol. 313, 234–245 (2008).
Kobayashi, A. et al. Six2 defines and regulates a multipotent self-renewing nephron progenitor population throughout mammalian kidney development. Cell Stem Cell 3, 169–181 (2008).
Batchelder, C. A., Lee, C. C., Matsell, D. G., Yoder, M. C. & Tarantal, A. F. Renal ontogeny in the rhesus monkey (Macaca mulatta) and directed differentiation of human embryonic stem cells towards kidney precursors. Differentiation 78, 45–56 (2009).
Bruce, S. J. et al. In vitro differentiation of murine embryonic stem cells toward a renal lineage. Differentiation 75, 337–349 (2007).
Kim, D. & Dressler, G. R. Nephrogenic factors promote differentiation of mouse embryonic stem cells into renal epithelia. J. Am. Soc. Nephrol. 16, 3527–3534 (2005).
Mae, S. et al. Monitoring and robust induction of nephrogenic intermediate mesoderm from human pluripotent stem cells. Nature Commun. 4, 1367 (2013).
Narayanan, K. et al. Human embryonic stem cells differentiate into functional renal proximal tubular-like cells. Kidney Int. 83, 593–603 (2013).
Song, B. et al. The directed differentiation of human iPS cells into kidney podocytes. PLoS ONE 7, e46453 (2012).
Vigneau, C. et al. Mouse embryonic stem cell-derived embryoid bodies generate progenitors that integrate long term into renal proximal tubules in vivo. J. Am. Soc. Nephrol. 18, 1709–1720 (2007).
Hendry, C. E. et al. Direct transcriptional reprogramming of adult cells to embryonic nephron progenitors. J. Am. Soc. Nephrol. 24, 1424–1434 (2013).
Okita, K. et al. A more efficient method to generate integration-free human iPS cells. Nature Methods 8, 409–412 (2011).
Hendry, C., Rumballe, B., Moritz, K. & Little, M. H. Defining and redefining the nephron progenitor population. Pediatr. Nephrol. 26, 1395–1406 (2011).
Kuure, S. et al. Crosstalk between Jagged1 and GDNF/Ret/GFRalpha1 signalling regulates ureteric budding and branching. Mech. Dev. 122, 765–780 (2005).
Shakya, R., Watanabe, T. & Costantini, F. The role of GDNF/Ret signaling in ureteric bud cell fate and branching morphogenesis. Dev. Cell 8, 65–74 (2005).
Davies, J. A., Unbekandt, M., Ineson, J., Lusis, M. & Little, M. H. Dissociation of embryonic kidney followed by re-aggregation as a method for chimeric analysis. Methods Mol. Biol. 886, 135–146 (2012).
Moretti, A. et al. Patient-specific induced pluripotent stem-cell models for long-QT syndrome. New Engl. J. Med. 363, 1397–1409 (2010).
Itzhaki, I. et al. Modelling the long QT syndrome with induced pluripotent stem cells. Nature 471, 225–229 (2011).
Nagao, S., Kugita, M., Yoshihara, D. & Yamaguchi, T. Animal models for human polycystic kidney disease. Exp. Anim. 61, 477–488 (2012).
Nagao, S. et al. Polycystic kidney disease in Han:SPRD Cy rats is associated with elevated expression and mislocalization of SamCystin. Am. J. Physiol. Renal. Physiol. 299, F1078–F1086 (2010).
Ren, X. et al. Differentiation of murine embryonic stem cells toward renal lineages by conditioned medium from ureteric bud cells in vitro. Acta Biochim. Biophys. Sin. 42, 464–471 (2010).
Acknowledgements
We thank M. Schwarz for administrative support. We thank J. Kasuboski from Waitt Advanced Biophotonics Core at Salk for help with imaging processing. We thank B. C. Lu for his suggestions regarding kidney dissection and organ culture. Y.X. and K.S. were partially supported by the California Institute for Regenerative Medicine. I.S-M. was partially supported by a Nomis Foundation postdoctoral fellowship. Work in the laboratory of J.C.I.B. was supported by grants from Fundacion Cellex, the G. Harold and L. Y. Mathers Charitable Foundation, The Leona M. and Harry B. Helmsley Charitable Trust, IPSEN Foundation, Fundació La Marató de TV3 (121330), CIBER BBN and ISCIII-TERCEL-MINECO.
Author information
Authors and Affiliations
Contributions
Y.X., I.S-M., E.N. and J.C.I.B. designed all experiments. Y.X., I.S-M., E.N. and J.C.I.B. wrote the manuscript. Y.X., I.S-M., T.G. and E.N. performed and analysed all experiments. D.O., I.D., C.R.E. and Y.X. performed in vivo experiments. N.M. was responsible for cell culture and maintenance of the patient samples related to this work. K.S. and M-Z.W. provided reagents. J.M.C. contributed to the overall design of the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 iPS cells derived by non-integrative approaches demonstrated the typical hallmarks of pluripotency.
a) Immunofluorescence analysis demonstrating expression of pluripotency markers. b) Karyotype of iPSCs (Fibro-Epi). (c) mRNA fold change of pluripotency markers and different lineage markers during EB differentiation of iPSC/ESCs. (d) Copy numbers of episomal vectors in iPSCs (Fibro-Epi), human ESCs and fibroblasts infected with episomal vectors. Data are represented as mean +/− SD. *p<0.05, (c,d) n = 6 (3 dishes-containing cells per experiment x 2 independent experiments). Scale bars: 100 μm (a).
Supplementary Figure 2 Differentiation of hPSCs into intermediate mesodermlike cells does not induce other lineages.
a,b) mRNA fold change of endodermal and ectodermal-related genes during the course of differentiation of human iPSCs (a) and ESCs (b). Data are represented as mean +/− SD. *p<0.05, n = 9 (3 dishes-containing cells per experiment×3 independent experiments).
Supplementary Figure 3 Differentiation of hPSCs into intermediate mesoderm UB progenitor-like cells.
a) Immunofluorescence analysis demonstrating specificity of the Human Nuclear antigen antibody used in the presence of pure murine cultures. b,c) Immunofluorescence analysis demonstrating specificity of the in vitro differentiation protocol of human PSCs. Non-renal epithelial cells of human origin failed to induce any chimeric UB-like structure. n = >3. Scale bars: 50 μm (a,b,c).
Supplementary information
Supplementary Information
Supplementary Information (PDF 731 kb)
Supplementary Table 1
Supplementary Information (XLSX 47 kb)
The video displays one UB arm comprised with only human H1(GFP)-derived UB progenitor-like cells that is surrounded by Six2+ mouse cap metanephric mesenchyme cells.
GFP: human cells; Red: CK8; Magenta: Six2. (MOV 14113 kb)
The video displays one UB arm comprised with only human H1-derived UB progenitor-like cells that is surrounded by Six2+ mouse cap metanephric mesenchyme cells.
Green: HuNu; Red: CK8; Magenta: Six2. (MOV 13954 kb)
Rights and permissions
About this article
Cite this article
Xia, Y., Nivet, E., Sancho-Martinez, I. et al. Directed differentiation of human pluripotent cells to ureteric bud kidney progenitor-like cells. Nat Cell Biol 15, 1507–1515 (2013). https://doi.org/10.1038/ncb2872
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2872
This article is cited by
-
Cells sorted off hiPSC-derived kidney organoids coupled with immortalized cells reliably model the proximal tubule
Communications Biology (2023)
-
Engraftment of Kidney Organoids In Vivo
Current Transplantation Reports (2023)
-
Urological cancer organoids, patients' avatars for precision medicine: past, present and future
Cell & Bioscience (2022)
-
Human organoid models to study SARS-CoV-2 infection
Nature Methods (2022)
-
Translating Organoids into Artificial Kidneys
Current Transplantation Reports (2022)