Abstract
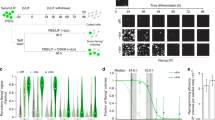
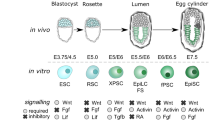
Oct4 is considered a master transcription factor for pluripotent cell self-renewal, but its biology remains poorly understood. Here, we investigated the role of Oct4 using the process of induced pluripotency. We found that a defined embryonic stem cell (ESC) level of Oct4 is required for pluripotency entry. However, once pluripotency is established, the Oct4 level can be decreased up to sevenfold without loss of self-renewal. Unexpectedly, cells constitutively expressing Oct4 at an ESC level robustly differentiated into all embryonic lineages and germline. In contrast, cells with low Oct4 levels were deficient in differentiation, exhibiting expression of naive pluripotency genes in the absence of pluripotency culture requisites. The restoration of Oct4 expression to an ESC level rescued the ability of these to restrict naive pluripotent gene expression and to differentiate. In conclusion, a defined Oct4 level controls the establishment of naive pluripotency as well as commitment to all embryonic lineages.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Accession codes
Change history
07 May 2013
In the version of this Article originally published online, there were errors in Fig. 1k,l. This has been corrected in the HTML and PDF versions.
References
Nichols, J. & Smith, A. Naive and primed pluripotent states. Cell Stem. Cell 4, 487–492 (2009).
Rosner, M. H. et al. A POU-domain transcription factor in early stem cells and germ cells of the mammalian embryo. Nature 345, 686–692 (1990).
Scholer, H. R., Dressler, G. R., Balling, R., Rohdewohld, H. & Gruss, P. Oct-4: a germline-specific transcription factor mapping to the mouse t-complex. EMBO J. 9, 2185–2195 (1990).
Okamoto, K. et al. A novel octamer binding transcription factor is differentially expressed in mouse embryonic cells. Cell 60, 461–472 (1990).
Yeom, Y. I. et al. Germline regulatory element of Oct-4 specific for the totipotent cycle of embryonal cells. Development 122, 881–894 (1996).
Nichols, J. et al. Formation of pluripotent stem cells in the mammalian embryo depends on the POU transcription factor Oct4. Cell 95, 379–391 (1998).
Niwa, H., Miyazaki, J. & Smith, A. G. Quantitative expression of Oct-3/4 defines differentiation, dedifferentiation or self-renewal of ES cells. Nat. Genet 24, 372–376 (2000).
Theunissen, T. W. et al. Nanog overcomes reprogramming barriers and induces pluripotency in minimal conditions. Curr. Biol. 21, 65–71 (2011).
Kim, J. B. et al. Direct reprogramming of human neural stem cells by OCT4. Nature 461, 649–643 (2009).
Yuan, X. et al. Combined chemical treatment enables Oct4-induced reprogramming from mouse embryonic fibroblasts. Stem Cells 29, 549–553 (2011).
Li, Y. et al. Generation of iPSCs from mouse fibroblasts with a single gene, Oct4, and small molecules. Cell Res. 21, 196–204 (2011).
Kim, J. B. et al. Oct4-induced pluripotency in adult neural stem cells. Cell 136, 411–419 (2009).
Chambers, I. et al. Functional expression cloning of Nanog, a pluripotency sustaining factor in embryonic stem cells. Cell 113, 643–655 (2003).
Ying, Q. L., Griffiths, D., Li, M. & Smith, A. Conversion of embryonic stem cells into neuroectodermal precursors in adherent monoculture. Nat. Biotechnol. 21, 183–186 (2003).
Wilkinson, D. G., Bhatt, S. & Herrmann, B. G. Expression pattern of the mouse T gene and its role in mesoderm formation. Nature 343, 657–659 (1990).
Avilion, A. A. et al. Multipotent cell lineages in early mouse development depend on SOX2 function. Genes Dev. 17, 126–140 (2003).
Monaghan, A. P., Kaestner, K. H., Grau, E. & Schutz, G. Postimplantation expression patterns indicate a role for the mouse forkhead/HNF- 3α,β and γ genes in determination of the definitive endoderm, chordamesoderm and neuroectoderm. Development 119, 567–578 (1993).
Kanai-Azuma, M. et al. Depletion of definitive gut endoderm in Sox17-null mutant mice. Development 129, 2367–2379 (2002).
Downs, K. M. Systematic localization of Oct-3/4 to the gastrulating mouse conceptus suggests manifold roles in mammalian development. Dev. Dyn. 237, 464–475 (2008).
Chambers, I. et al. Nanog safeguards pluripotency and mediates germline development. Nature 450, 1230–1234 (2007).
Hayashi, K., Lopes, S. M., Tang, F. & Surani, M. A. Dynamic equilibrium and heterogeneity of mouse pluripotent stem cells with distinct functional and epigenetic states. Cell Stem. Cell 3, 391–401 (2008).
Toyooka, Y., Shimosato, D., Murakami, K., Takahashi, K. & Niwa, H. Identification and characterization of subpopulations in undifferentiated ES cell culture. Development 135, 909–918 (2008).
Kobayashi, T. et al. The cyclic gene Hes1 contributes to diverse differentiation responses of embryonic stem cells. Genes Dev. 23, 1870–1875 (2009).
Canham, M. A., Sharov, A. A., Ko, M. S. & Brickman, J. M. Functional heterogeneity of embryonic stem cells revealed through translational amplification of an early endodermal transcript. PLoS Biol. 8, e1000379 (2010).
Festuccia, N. et al. Esrrb is a direct Nanog target gene that can substitute for Nanog function in pluripotent cells. Cell Stem. Cell 11, 477–490 (2012).
Hall, J. et al. Oct4 and LIF/Stat3 additively induce Kruppel factors to sustain embryonic stem cell self-renewal. Cell Stem. Cell 5, 597–609 (2009).
Niwa, H., Ogawa, K., Shimosato, D. & Adachi, K. A parallel circuit of LIF signalling pathways maintains pluripotency of mouse ES cells. Nature 460, 118–122 (2009).
Van Oosten, A. L., Costa, Y., Smith, A. & Silva, J. C. JAK/STAT3 signalling is sufficient and dominant over antagonistic cues for the establishment of naive pluripotency. Nat. Commun. 3, 817 (2012).
Niwa, H., Masui, S., Chambers, I., Smith, A. G. & Miyazaki, J. Phenotypic complementation establishes requirements for specific POU domain and generic transactivation function of Oct-3/4 in embryonic stem cells. Mol. Cell Biol. 22, 1526–1536 (2002).
Vigano, M. A. & Staudt, L. M. Transcriptional activation by Oct-3: evidence for a specific role of the POU-specific domain in mediating functional interaction with Oct-1. Nucleic Acids Res. 24, 2112–2118 (1996).
Silva, J. et al. Promotion of reprogramming to ground state pluripotency by signal inhibition. PLoS Biol. 6, e253 (2008).
Ying, Q. L. et al. The ground state of embryonic stem cell self-renewal. Nature 453, 519–523 (2008).
Thomson, M. et al. Pluripotency factors in embryonic stem cells regulate differentiation into germ layers. Cell 145, 875–889 (2011).
Teo, A. K. et al. Pluripotency factors regulate definitive endoderm specification through eomesodermin. Genes Dev. 25, 238–250 (2011).
Du, P., Kibbe, W. A. & Lin, S. M. Lumi: a pipeline for processing Illumina microarray. Bioinformatics 24, 1547–1548 (2008).
Lin, S. M., Du, P., Huber, W. & Kibbe, W. A. Model-based variance-stabilizing transformation for Illumina microarray data. Nucleic Acids Res. 36, e11 (2008).
Smyth, G. K. Linear models and empirical Bayes methods for assessing differential expression in microarray experiments. Stat. Appl. Genet Mol. Biol. 3, Article 3 (2004).
Acknowledgements
We thank W. Mansfield and C-E. Dumeau for blastocyst injections and morula aggregations, R. Walker for flow cytometry, and M. McLeish and H. Skelton for histological processing of teratomas. We are grateful to H. Niwa for providing mice with different Oct4 genotypes and A. Smith and J. Betschinger for providing plasmids. We are also grateful to Y. Costa and P. Shliaha for technical assistance and H. Stuart for critical reading of the manuscript. The study was supported by Wellcome Trust Fellowship WT086692MA. J.C.R.S. is a Wellcome Trust Career Development Fellow. A.R. is a recipient of the Darwin Trust of Edinburgh Postgraduate Scholarship.
Author information
Authors and Affiliations
Contributions
A.R. performed and designed the experiments, analysed the data and wrote the manuscript. R.L.S. and G.L.B.C. performed experiments. T.W.T., L.F.C., A.R. and J.S. designed the study. J.N. analysed data. J.S. supervised the study, designed the experiments, analysed the data, and wrote and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 1736 kb)
Supplementary Table 1
Supplementary Information (XLSX 13 kb)
PB-Oct4 iPSCs−/− differentiate into beating heart cells.
Embryoid body outgrowths of PB-Oct4 iPSCs−/− contain beating heart cells 3 days after plating on gelatine-coated dishes. (AVI 4310 kb)
Rights and permissions
About this article
Cite this article
Radzisheuskaya, A., Le Bin Chia, G., dos Santos, R. et al. A defined Oct4 level governs cell state transitions of pluripotency entry and differentiation into all embryonic lineages. Nat Cell Biol 15, 579–590 (2013). https://doi.org/10.1038/ncb2742
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2742
This article is cited by
-
Epigenetic OCT4 regulatory network: stochastic analysis of cellular reprogramming
npj Systems Biology and Applications (2024)
-
RSL24D1 sustains steady-state ribosome biogenesis and pluripotency translational programs in embryonic stem cells
Nature Communications (2023)
-
Molecular versatility during pluripotency progression
Nature Communications (2023)
-
CasTuner is a degron and CRISPR/Cas-based toolkit for analog tuning of endogenous gene expression
Nature Communications (2023)
-
A critical review on therapeutic approaches of CRISPR-Cas9 in diabetes mellitus
Naunyn-Schmiedeberg's Archives of Pharmacology (2023)