Abstract
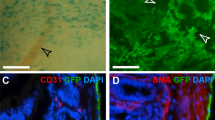
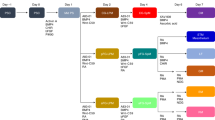
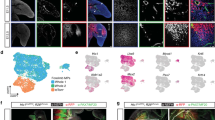
Fibroblasts and smooth muscle cells (FSMCs) are principal cell types of connective and adventitial tissues that participate in the development, physiology and pathology of internal organs, with incompletely defined cellular origins. Here, we identify and prospectively isolate from the mesothelium a mouse cell lineage that is committed to FSMCs. The mesothelium is an epithelial monolayer covering the vertebrate thoracic and abdominal cavities and internal organs. Time-lapse imaging and transplantation experiments reveal robust generation of FSMCs from the mesothelium. By targeting mesothelin (MSLN), a surface marker expressed on mesothelial cells, we identify and isolate precursors capable of clonally generating FSMCs. Using a genetic lineage tracing approach, we show that embryonic and adult mesothelium represents a common lineage to trunk FSMCs, and trunk vasculature, with minimal contributions from neural crest, or circulating cells. The isolation of FSMC precursors enables the examination of multiple aspects of smooth muscle and fibroblast biology as well as the prospective isolation of these precursors for potential regenerative medicine purposes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Hay, E. D. Development of the vertebrate cornea. Int. Rev. Cytol. 63, 263–322 (1979).
Bochaton-Piallat, M. L. & Gabbiani, G. Smooth muscle cell: a key cell for plaque vulnerability regulation? Circ. Res. 98, 448–449 (2006).
Kennedy, L. J. & Weissman, I. L. Dual origin of intimal cells in cardiac-allograft arteriosclerosis. N. Engl. J. Med. 285, 884–887 (1971).
De Wever, O., Demetter, P., Mareel, M. & Bracke, M. Stromal myofibroblasts are drivers of invasive cancer growth. Int. J. Cancer 123, 2229–2238 (2008).
Desmoulière, A., Guyot, C. & Gabbiani, G. The stroma reaction myofibroblast: a key player in the control of tumor cell behavior. Int. J. Dev. Biol. 48, 509–517 (2004).
Albini, A. & Sporn, M. B. The tumour microenvironment as a target for chemoprevention. Nat. Rev. Cancer 7, 139–147 (2007).
Gittenberger-de Groot, A. C., Vrancken Peeters, M. P., Mentink, M. M., Gourdie, R. G. & Poelmann, R. E. Epicardium-derived cells contribute a novel population to the myocardial wall and the atrioventricular cushions. Circ. Res. 82, 1043–1052 (1998).
Prockop, D. J. Marrow stromal cells as stem cells for nonhematopoietic tissues. Science 276, 71–74 (1997).
Zhang, N. et al. Blood-borne stem cells differentiate into vascular and cardiac lineages during normal development. Stem Cells Dev. 15, 17–28 (2006).
Hashimoto, N., Jin, H., Liu, T., Chensue, S. W. & Phan, S. H. Bone marrow- derived progenitor cells in pulmonary fibrosis. J. Clin. Invest. 113, 243–52 (2004).
Wagers, A. J., Sherwood, R. I., Christensen, J. L. & Weissman, I. L. Little evidence for developmental plasticity of adult hematopoietic stem cells. Science 297, 2256–2259 (2002).
Jiang, X., Rowitch, D. H., Soriano, P., McMahon, A. P. & Sucov, H. M. Fate of the mammalian cardiac neural crest. Science 297, 2256–2259 (2002).
Sorrell, J. M. & Caplan, A. I. Fibroblasts-a diverse population at the center of it all. Int. Rev. Cell Mol. Biol. 276, 161–214 (2009).
Rinn, J. L., Bondre, C., Gladstone, H. B., Brown, P. O. & Chang, H. Y. Anatomic demarcation by positional variation in fibroblast gene expression programs. PLoS Genet. 2, e119 (2006).
Mutsaers, S. E. & Wilkosz, S. Structure and function of mesothelial cells. Cancer Treat. Res. 134, 1–19 (2007).
Yung, S. & Chan, T. M. Intrinsic cells: mesothelial cells—central players in regulating inflammation and resolution. Perit. Dial. Int. 29, S21–S27 (2009).
Herrick, S. E. & Mutsaers, S. E. Mesothelial progenitor cells and their potential in tissue engineering. Int. J. Biochem. Cell Biol. 36, 621–642 (2004).
Elmadbouh, I., Michel, J. B. & Chachques, J. C. Mesothelial cell transplantation in myocardial infarction. Int. J. Artif. Organs 30, 541–549 (2007).
Mutsaers, S. E. & Di Paolo, N. Future directions in mesothelial transplantation research. Int. J. Artif. Organs 30, 557–561 (2007).
Hardy, R. R. B-1 B cell development. J. Immunol. 177, 2749–2754 (2006).
Chang, K. & Pastan, I. Molecular cloning of mesothelin, a differentiation antigen present on mesothelium, mesotheliomas, and ovarian cancers. Proc. Natl Acad. Sci. USA 93, 136–140 (1996).
Sahoo, D. et al. MiDReG: a method of mining developmentally regulated genes using Boolean implications. Proc. Natl Acad. Sci. USA 107, 5732–5737 (2010).
Olivera-Martinez, I., Missier, S., Fraboulet, S., Thélu, J. & Dhouailly, D. Differential regulation of the chick dorsal thoracic dermal progenitors from the medial dermomyotome. Development 129, 4763–4772 (2002).
Atit, R. et al. β-catenin activation is necessary and sufficient to specify the dorsal dermal fate in the mouse. Dev. Biol. 296, 164–176 (2006).
Tran, T. H. et al. Role of canonical Wnt signaling/ß-catenin via Dermo1 in cranial dermal cell development. Development 137, 3973–3984 (2010).
Ueno, H. & Weissman, I. L. Clonal analysis of mouse development reveals a polyclonal origin for yolk sac blood islands. Dev. Cell 11, 519–533 (2006).
Kirsch, D. G. et al. p53 controls radiation-induced gastrointestinal syndrome in mice independent of apoptosis. Science 327, 593–596 (2010).
Jiang, X., Rowitch, D. H., Soriano, P., McMahon, A. P. & Sucov, H. M. Fate of the mammalian cardiac neural crest. Development 127, 1607–1616 (2000).
Muzumdar, M. D., Tasic, B., Miyamichi, K., Li, L. & Luo, L. A global double fluorescent Cre reporter mouse. Genesis 45, 593–605 (2007).
Yamauchi, Y. et al. A novel transgenic technique that allows specific marking of the neural crest cell lineage in mice. Dev. Biol. 212, 191–203 (1999).
Cheung, M. et al. The transcriptional control of trunk neural crest induction, survival, and delamination. Dev. Cell 8, 179–192 (2005).
Choudhary, B. et al. Cardiovascular malformations with normal smooth muscle differentiation in neural crest-specific type II TGFβ receptor (Tgfbr2) mutant mice. Dev. Biol. 289, 420–429 (2006).
Wright, D. H., Wagers, A. J., Gulati, A. P., Johnson, F. L. & Weissman, I. L. Physiological migration of hematopoietic stem and progenitor cells. Science 294, 1933–1936 (2001).
Mikawa, T. & Gourdie, R. G. Pericardial mesoderm generates a population of smooth muscle cells migrating into the heart along with ingrowth of the pericardial organ. Dev. Biol. 174, 221–232 (1996).
Männer, J. Does the subepicardial mesenchyme contribute myocardioblasts to the myocardium of the chick embryo heart? A quail-chick chimera study tracing the fate of the epicardial primordium. Anat. Rec. 255, 212–226 (1999).
Wessels, A. & Pérez-Pomares, J. M. The epicardium and epicardially derived cells (EPDCs) as cardiac stem cells. Anat. Rec. A Discov. Mol. Cell Evol. Biol. 276, 43–57 (2004).
Smart, N. et al. De novo cardiomyocytes from within the activated adult heart after injury. Nature 474, 640–644 (2011).
Cai, C. L. et al. A myocardial lineage derives from Tbx18 epicardial cells. Nature 454, 104–108 (2008).
Zhou, B. et al. Epicardial progenitors contribute to the cardiomyocyte lineage in the developing heart. Nature 454, 109–113 (2008).
Chong, J. J. et al. Adult cardiac-resident MSC-like stem cells with a proepicardial origin. Cell Stem Cell 9, 527–540 (2011).
Zeng, B., Ren, X. F., Cao, F., Zhou, X. Y. & Zhang, J. Developmental patterns and characteristics of epicardial cell markers Tbx18 and Wt1 in murine embryonic heart. J. Biomed. Sci. 18, 67 (2011).
Christoffels, V. M. et al. Tbx18 and the fate of epicardial progenitors. Nature 458, E8–E9 (2009).
Hosen, N. et al. The Wilms’ tumor gene WT1-GFP knock-in mouse reveals the dynamic regulation of WT1 expression in normal and leukemic hematopoiesis. Leukemia 21, 1783–1791 (2007).
Wilm, B., Ipenberg, A., Hastie, N. D., Burch, J. B. & Bader, D. M. The serosal mesothelium is a major source of smooth muscle cells of the gut vasculature. Development 132, 5317–5328 (2005).
Que, J. et al. Mesothelium contributes to vascular smooth muscle and mesenchyme during lung development. Proc. Natl Acad. Sci. USA 105, 16626–16630 (2008).
Sahoo, D., Dill, D. L., Gentles, A. J., Tibshirani, R. & Plevritis, S. K. Boolean implication networks derived from large scale, whole genome microarray datasets. Gen. Biol. 9, R157 (2008).
Acknowledgements
We thank A. Mosley for assisting with generating the transgenic M s l nCLN mice, and R. Kopito, M. Brandeis and K. Bersuker for usage and their assistance with the time-lapse video. We thank T. Violante for her assistance with sectioning, D. Hunter for his assistance with histochemical staining and G. Paz and D. Montoro for their assistance with figure preparation. The CreERT2IRES–lacZ–PGK–neo cassette was a gift from R. J. Gilbertson (St. Jude Children’s Research Hospital, USA). This work was supported in part by a grant from the California Institute of Regenerative Medicine (RC1 00354 to I.L.W.), the Smith Family Trust (to I.L.W.) and from the National Institutes of Health (RO1 DK064640 to P.X.X.). Y.R. is supported by the Human Frontier Science Program (HFSP) Long Term Fellowship, and the Machiah Foundation Fellowship.
Author information
Authors and Affiliations
Contributions
Y.R. designed and performed the experiments, with suggestions from I.L.W. Y.R. imaged and analysed the data from all experiments. T.M. created the MSLN–CreER construct, with help from Y.R. D.S. contributed the Boolean analysis. P-X.X. contributed the tetrachimaera mouse models. J.R.B. contributed the PoCre transgenic mice model. Y.R. and I.L.W. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 1595 kb)
Supplementary Information
Supplementary Information (AVI 2376 kb)
Supplementary Information
Supplementary Information (AVI 2209 kb)
Rights and permissions
About this article
Cite this article
Rinkevich, Y., Mori, T., Sahoo, D. et al. Identification and prospective isolation of a mesothelial precursor lineage giving rise to smooth muscle cells and fibroblasts for mammalian internal organs, and their vasculature. Nat Cell Biol 14, 1251–1260 (2012). https://doi.org/10.1038/ncb2610
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2610
This article is cited by
-
Fibroblast diversity and plasticity in the tumor microenvironment: roles in immunity and relevant therapies
Cell Communication and Signaling (2023)
-
Cancer-associated fibroblasts: tumor defenders in radiation therapy
Cell Death & Disease (2023)
-
The small and large intestine contain related mesenchymal subsets that derive from embryonic Gli1+ precursors
Nature Communications (2023)
-
A topographic atlas defines developmental origins of cell heterogeneity in the human embryonic lung
Nature Cell Biology (2023)
-
Multi-disciplinary Insights from the First European Forum on Visceral Myopathy 2022 Meeting
Digestive Diseases and Sciences (2023)