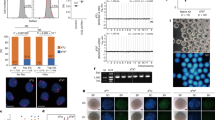
Abstract
The idea that females of most mammalian species have lost the capacity for oocyte production at birth1,2,3,4,5 has been challenged recently by the finding that juvenile and adult mouse ovaries possess mitotically active germ cells6. However, the existence of female germline stem cells (FGSCs) in postnatal mammalian ovaries still remains a controversial issue among reproductive biologists and stem cell researchers6,7,8,9,10. We have now established a neonatal mouse FGSC line, with normal karyotype and high telomerase activity, by immunomagnetic isolation and culture for more than 15 months. FGSCs from adult mice were isolated and cultured for more than 6 months. These FGSCs were infected with GFP virus and transplanted into ovaries of infertile mice. Transplanted cells underwent oogenesis and the mice produced offspring that had the GFP transgene. These findings contribute to basic research into oogenesis and stem cell self-renewal and open up new possibilities for use of FGSCs in biotechnology and medicine.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Zuckerman, S. The number of oocytes in the mature ovary. Recent Prog. Horm. Res. 6, 63–108 (1951).
Borum, K. Oogenesis in the mouse. A study of meiotic prophase. Exp. Cell Res. 24, 495–507 (1961).
Peters, H. Migration of gonocytes into the mammalian gonad and their differentiation. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 259, 91–101 (1970).
McLaren, A. Meiosis and differentiation of mouse germ cells. Symp. Soc. Exp. Biol. 38, 7–23 (1984).
Anderson, L. D. & Hirshfield, A. N. An overview of follicular development in the ovary: from embryo to the fertilized ovum in vitro. Md. Med. J. 41, 614–620 (1992).
Johnson, J., Canning, J., Kaneko, T., Pru, J. K. & Tilly, J. L. Germline stem cells and follicular renewal in the postnatal mammalian ovary. Nature 428, 145–150 (2004).
Eggan, k., Jurga, S., Gosden, R., Min, I. M. & Wagers, A. J. Ovulated oocytes in adult mice derive from non-circulating germ cells. Nature 441, 1109–1114 (2006).
Telfer, E. E. et al. On regenerating the ovary and generating controversy. Cell 122, 821–822 (2005).
Honda, A. et al. Isolation, characterization, and in vitro and in vivo differentiation of putative thecal stem cells. Proc. Natl Acad. Sci. USA 101, 16489–16494 (2007).
Gosden, R. G. Germline stem cells in the postnatal ovary: is the ovary more like a testis? Hum. Reprod. Update 10, 193–195 (2004).
Johnson J. J. et al. Oocyte generation in adult mammalian ovaries by putative germ cells in bone marrow and peripheral blood. Cell 122, 303–315 (2005).
Pepling, M. E., Wilhelm, J. E., O'Hara, A. L., Gephardt, G. W. & Spradling, A. C. Mouse oocytes within germ cell cysts and primordial follicles contain a Balbiani body. Proc. Natl Acad. Sci. U S A. 104, 187–192 (2007).
Fujiwara, Y. et al. Isolation of a DEAD-family protein gene that encodes a murine homology of Drosophila vasa and its specific expression in germ cell lineage. Proc. Natl Acad. Sci. USA 91, 12258–12262 (1994).
Noce, T., Okamoto-Ito, S. & Tsunekawa, N. Vasa homolog genes in mammalian germ cell development. Cell Struct. Funct. 26, 131–136 (2001).
Feng, L. et al. Generation and in vitro differentiation of a spermatogonial cell line. Science 297, 392–395 (2002).
Ruggiu, M. et al. The mouse Dazla gene encodes a cytoplasmic protein essential for gametogenesis. Nature 389, 73–77 (1997).
Scholer, H. R., Ruppert, S., Suzuki, N., Chowdhury, K. & Gruss, P. New type of POU domain in germ line-specific protein Oct-4. Nature 344, 435–439 (1990).
Ohinata, Y. et al. Blimp1 is a critical determinant of the germ cell lineage in mice. Nature 436, 207–213 (2005).
Tanaka, S. S., Yamaguchi, Y. L., Tsoi, B., Lickert, H. & Tam, P. P. IFITM/Mil/fragilis family proteins IFITM1 and IFITM3 play distinct roles in mouse primordial germ cell homing and repulsion. Dev. Cell. 9, 745–756 (2005).
Lange, U. C., Saitou, M., Western, P. S., Barton, S. C. & Surani, M. A. The Fragilis interferon-inducible gene family of transmembrane proteins is associated with germ cell specification in mice. BMC Dev. Bio. 3, 1 (2003).
Bortvin, A., Goodheart, M., Liao, M. & Page, D. C. Dppa3 / Pgc7 / stella is a maternal factor and is not required for germ cell specification in mice. BMC Dev. Bio. 4, 2 (2004).
Hu, B. & Colletti, L. M. Stem cell factor and c-kit are involved in hepatic recovery after acetaminophen-induced liver injury in mice. Am J. Physiol Gastrointest. Liver Physiol. 295, 45–53 (2008).
Joshi, S., Davies, H., Sims, L. P., Levy, S. E. & Dean J. Ovarian gene expression in the absence of FIGLA, an oocyte-specific transcription factor. BMC Dev. Biol. 7, 67 (2007).
Masui, S. et al. Pluripotency governed by Sox2 via regulation of Oct3/4 expression in mouse embryonic stem cells. Nature Cell Biol. 9, 625–635 (2007).
Xu, J., Sylvester, R., Tighe, A P., Chen, S. & Gudas, L. J. Transcriptional activation of the suppressor of cytokine signaling-3 (SOCS-3) gene via STAT3 is increased in F9 REX1 (ZFP-42) knockout teratocarcinoma stem cells relative to wild-type cells. J. Mol. Biol. 377, 28–46 (2008).
Chambers, I. et al. Functional expression cloning of Nanog, a pluripotency sustaining factor in embryonic stem cells. Cell 113, 643–655 (2003).
Yuan, L. et al. Female germ cell aneuploidy and embryo death in mice lacking the meiosis-specific protein SCP3. Science 296, 1115–1118 (2002).
Ravindranath, N., Dalal, R., Solomon, B., Djakiew, D. & Dym, M. Loss of telomerase activity during male germ cell differentiation. Endocrinology 138, 4026–4029 (1997).
Shiromizu, K., Thorgeirsson, S. S. & Mattison, D. R. Effect of cyclophosphamide on oocyte and follicle number in Sprague-Dawley rats, C57BL/6N and DBA/2N mice. Pediatr. Pharmacol. 4, 213–221 (1984).
Yang, Z. & Wu, J. Mouse dynein axonemal intermediate chain 2: cloning and expression. DNA Cell Biol. 27, 479–488 (2008).
Nagano, M., Avarbock, M. R., Leonida, E. B., Brinster, C. J. & Brinster, R. L. –Culture of mouse spermatogonial stem cells. Tissue Cell 30, 389–397 (1998).
Wu, J., Jester, W. F. Jr, Laslett. A. L., Meinhardt, A. & Orth, J. M. Expression of a novel factor, short-type PB-cadherin, in Sertoli cells and spermatogenic stem cells of the neonatal rat testis. J. Endocrinol. 176, 381–391 (2003).
Wu, J., Jester, W. F. Jr & Orth, J. M. Short-type PB-cadherin promotes survival of gonocytes and activates JAK–STAT signaling. Dev. Biol. 284, 437–450 (2005).
Wu, J. et al. Short-type PB-cadherin promotes self-renewal of spermatogonial stem cells via multiple signaling pathways. Cell Signal. 20, 1052–1060 (2008).
Xiong, Z., Laird, P. W. COBRA: a sensitive and quantitative DNA methylation assay. Nucleic Acids Res. 25, 2532–2534 (1997).
Acknowledgements
The authors would like to thank Il-Hoan Oh for providing the MSCV–PGK–GFP vector used in this study. This work was supported by Key Program of National Natural Scientific Foundation of China (No. 30630012), and sponsored by Shanghai Pujiang Program, China (No. 06PJ14058) and Shanghai Leading Academic Discipline Project (No. B205).
Author information
Authors and Affiliations
Contributions
K.Z. and L.S. isolated and cultured FGSCs, and created the FGSC line; K.Z., Z.Yu, Y.Z. and R.H. characterized the nFGSC line and long-term cultured aFGSCs; Z.Yu, Z.Ya and Q.Y. examined the physiological function of FGSCs and the presence of GFP transgenes; H.L. and K.S. carried out COBRA; L.Z. and J.X. performed identification of FGSCs in ovaries; K.Z. participated in data analysis; J.W. planned and supervised the project, carried out FGSC transplantation, analysed data and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 1160 kb)
Rights and permissions
About this article
Cite this article
Zou, K., Yuan, Z., Yang, Z. et al. Production of offspring from a germline stem cell line derived from neonatal ovaries. Nat Cell Biol 11, 631–636 (2009). https://doi.org/10.1038/ncb1869
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1869
This article is cited by
-
Exploring the Protective Effect of Total Flavonoids from Semen Cuscutae on Ovarian Germline Stem Cells Based on Notch Signaling Pathway
Stem Cell Reviews and Reports (2024)
-
STING Signaling Contributes to Celastrol-induced Pyroptosis of Female Germline Stem Cells by Upregulating NLRP3
Stem Cell Reviews and Reports (2024)
-
Metformin promotes female germline stem cell proliferation by upregulating Gata-binding protein 2 with histone β-hydroxybutyrylation
Stem Cell Research & Therapy (2023)
-
Chito-oligosaccharides and macrophages have synergistic effects on improving ovarian stem cells function by regulating inflammatory factors
Journal of Ovarian Research (2023)
-
A rapid and stable spontaneous reprogramming system of Spermatogonial stem cells to Pluripotent State
Cell & Bioscience (2023)