Abstract
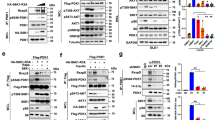
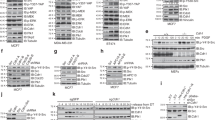
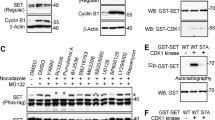
Deregulated Skp2 function promotes cell transformation, and this is consistent with observations of Skp2 overexpression in many human cancers. However, the mechanisms underlying elevated Skp2 expression are still unknown. Here we show that the serine/threonine protein kinase Akt1, but not Akt2, directly controls Skp2 stability by a mechanism that involves degradation by the APC–Cdh1 ubiquitin ligase complex. We show further that Akt1 phosphorylates Skp2 at Ser 72, which is required to disrupt the interaction between Cdh1 and Skp2. In addition, we show that Ser 72 is localized within a putative nuclear localization sequence and that phosphorylation of Ser 72 by Akt leads to cytoplasmic translocation of Skp2. This finding expands our knowledge of how specific signalling kinase cascades influence proteolysis governed by APC–Cdh1 complexes, and provides evidence that elevated Akt activity and cytoplasmic Skp2 expression may be causative for cancer progression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Change history
18 March 2009
In the version of this article initially published online, Fig 6e had no separating lines between Akt1 and Akt2 or Cdh1 and Cdh1/Akt1. This error has now been corrected for the HTML and PDF versions of the article.
References
Cardozo, T. & Pagano, M. The SCF ubiquitin ligase: insights into a molecular machine. Nature Rev. Mol. Cell Biol. 5, 739–751 (2004).
Gstaiger, M. et al. Skp2 is oncogenic and overexpressed in human cancers. Proc. Natl Acad. Sci. USA 98, 5043–5048 (2001).
Signoretti, S. et al. Oncogenic role of the ubiquitin ligase subunit Skp2 in human breast cancer. J. Clin. Invest. 110, 633–641 (2002).
Bashir, T., Dorrello, N. V., Amador, V., Guardavaccaro, D. & Pagano, M. Control of the SCF(Skp2-Cks1) ubiquitin ligase by the APC/C(Cdh1) ubiquitin ligase. Nature 428, 190–193 (2004).
Wei, W. et al. Degradation of the SCF component Skp2 in cell-cycle phase G1 by the anaphase-promoting complex. Nature 428, 194–198 (2004).
van Duijn, P. W. & Trapman, J. PI3K/Akt signaling regulates p27kip1 expression via Skp2 in PC3 and DU145 prostate cancer cells, but is not a major factor in p27kip1 regulation in LNCaP and PC346 cells. Prostate 66, 749–760 (2006).
Andreu, E. J. et al. BCR-ABL induces the expression of Skp2 through the PI3K pathway to promote p27Kip1 degradation and proliferation of chronic myelogenous leukemia cells. Cancer Res. 65, 3264–3272 (2005).
Mamillapalli, R. et al. PTEN regulates the ubiquitin-dependent degradation of the CDK inhibitor p27KIP1 through the ubiquitin E3 ligase SCFSKP2. Curr. Biol. 11, 263–267 (2001).
Woodgett, J. R. Recent advances in the protein kinase B signaling pathway. Curr. Opin. Cell Biol. 17, 150–157 (2005).
Parsons, R. Human cancer, PTEN and the PI-3 kinase pathway. Semin. Cell Dev. Biol. 15, 171–176 (2004).
Datta, S. R. et al. Akt phosphorylation of BAD couples survival signals to the cell-intrinsic death machinery. Cell 91, 231–241 (1997).
Zhao, X. et al. Multiple elements regulate nuclear/cytoplasmic shuttling of FOXO1: characterization of phosphorylation- and 14-3-3-dependent and -independent mechanisms. Biochem. J. 378, 839–849 (2004).
Brunet, A. et al. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 96, 857–868 (1999).
Zhou, B. P. et al. Cytoplasmic localization of p21Cip1/WAF1 by Akt-induced phosphorylation in HER-2/neu-overexpressing cells. Nature Cell Biol. 3, 245–252 (2001).
Viglietto, G. et al. Cytoplasmic relocalization and inhibition of the cyclin-dependent kinase inhibitor p27Kip1 by PKB/Akt-mediated phosphorylation in breast cancer. Nature Med. 8, 1136–1144 (2002).
Liang, J. et al. PKB/Akt phosphorylates p27, impairs nuclear import of p27 and opposes p27-mediated G1 arrest. Nature Med. 8, 1153–1160 (2002).
Shin, I. et al. PKB/Akt mediates cell-cycle progression by phosphorylation of p27Kip1 at threonine 157 and modulation of its cellular localization. Nature Med. 8, 1145–1152 (2002).
Whiteman, E. L., Cho, H. & Birnbaum, M. J. Role of Akt/protein kinase B in metabolism. Trends Endocrinol. Metab. 13, 444–451 (2002).
Heron-Milhavet, L. et al. Only Akt1 is required for proliferation, while Akt2 promotes cell cycle exit through p21 binding. Mol. Cell. Biol. 26, 8267–8280 (2006).
Irie, H. Y. et al. Distinct roles of Akt1 and Akt2 in regulating cell migration and epithelial–mesenchymal transition. J. Cell Biol. 171, 1023–1034 (2005).
Drobnjak, M. et al. Altered expression of p27 and Skp2 proteins in prostate cancer of African-American patients. Clin. Cancer Res. 9, 2613–2619 (2003).
Radke, S., Pirkmaier, A. & Germain, D. Differential expression of the F-box proteins Skp2 and Skp2B in breast cancer. Oncogene 24, 3448–3458 (2005).
Shapira, M., Kakiashvili, E., Rosenberg, T. & Hershko, D. D. The mTOR inhibitor rapamycin down-regulates the expression of the ubiquitin ligase subunit Skp2 in breast cancer cells. Breast Cancer Res. 8, R46 (2006).
Reichert, M., Saur, D., Hamacher, R., Schmid, R. M. & Schneider, G. Phosphoinositide-3-kinase signaling controls S-phase kinase-associated protein 2 transcription via E2F1 in pancreatic ductal adenocarcinoma cells. Cancer Res. 67, 4149–4156 (2007).
Barre, B. & Perkins, N. D. A cell cycle regulatory network controlling NF-κB subunit activity and function. EMBO J. 26, 4841–4855 (2007).
Zhou, B. P. et al. HER-2/neu induces p53 ubiquitination via Akt-mediated MDM2 phosphorylation. Nature Cell Biol. 3, 973–982 (2001).
Zhang, H. et al. Phosphoprotein analysis using antibodies broadly reactive against phosphorylated motifs. J. Biol. Chem. 277, 39379–39387 (2002).
Hong, F. et al. mTOR-raptor binds and activates SGK1 to regulate p27 phosphorylation. Mol. Cell 30, 701–711 (2008).
Yam, C. H., Ng, R. W., Siu, W. Y., Lau, A. W. & Poon, R. Y. Regulation of cyclin A-Cdk2 by SCF component Skp1 and F-box protein Skp2. Mol. Cell. Biol. 19, 635–645 (1999).
Tran, H., Brunet, A., Griffith, E. C. & Greenberg, M. E. The many forks in FOXO's road. Sci. STKE 2003, RE5 (2003).
Carrano, A. C., Eytan, E., Hershko, A. & Pagano, M. SKP2 is required for ubiquitin-mediated degradation of the CDK inhibitor p27. Nature Cell Biol. 1, 193–199 (1999).
Rodier, G., Coulombe, P., Tanguay, P. L., Boutonnet, C. & Meloche, S. Phosphorylation of Skp2 regulated by CDK2 and Cdc14B protects it from degradation by APCCdh1 in G1 phase. EMBO J. 27, 679–691 (2008).
Ji, P. et al. Skp2 contains a novel cyclin A binding domain that directly protects cyclin A from inhibition by p27Kip1. J. Biol. Chem. 281, 24058–24069 (2006).
Vogt, P. K., Jiang, H. & Aoki, M. Triple layer control: phosphorylation, acetylation and ubiquitination of FOXO proteins. Cell Cycle 4, 908–913 (2005).
Chiang, C. W. et al. Protein phosphatase 2A dephosphorylation of phosphoserine 112 plays the gatekeeper role for BAD-mediated apoptosis. Mol. Cell. Biol. 23, 6350–6362 (2003).
Wang, B. et al. Isolation of high-affinity peptide antagonists of 14-3-3 proteins by phage display. Biochemistry 38, 12499–12504 (1999).
Poon, I. K. & Jans, D. A. Regulation of nuclear transport: central role in development and transformation? Traffic 6, 173–186 (2005).
Fabbro, M. & Henderson, B. R. Regulation of tumor suppressors by nuclear-cytoplasmic shuttling. Exp. Cell Res. 282, 59–69 (2003).
Fujita, E. et al. Akt phosphorylation site found in human caspase-9 is absent in mouse caspase-9. Biochem. Biophys. Res. Commun. 264, 550–555 (1999).
Harper, J. W., Burton, J. L. & Solomon, M. J. The anaphase-promoting complex: it's not just for mitosis any more. Genes Dev. 16, 2179–2206 (2002).
Mailand, N. & Diffley, J. F. CDKs promote DNA replication origin licensing in human cells by protecting Cdc6 from APC/C-dependent proteolysis. Cell 122, 915–926 (2005).
Hennekes, H., Peter, M., Weber, K. & Nigg, E. A. Phosphorylation on protein kinase C sites inhibits nuclear import of lamin B2. J. Cell Biol. 120, 1293–1304 (1993).
Zhang, F., White, R. L. & Neufeld, K. L. Cell density and phosphorylation control the subcellular localization of adenomatous polyposis coli protein. Mol. Cell. Biol. 21, 8143–8156 (2001).
Dowen, S. E., Scott, A., Mukherjee, G. & Stanley, M. A. Overexpression of Skp2 in carcinoma of the cervix does not correlate inversely with p27 expression. Int. J. Cancer 105, 326–330 (2003).
Yoeli-Lerner, M. et al. Akt blocks breast cancer cell motility and invasion through the transcription factor NFAT. Mol. Cell 20, 539–550 (2005).
Hamada, K. et al. The PTEN/PI3K pathway governs normal vascular development and tumor angiogenesis. Genes Dev. 19, 2054–2065 (2005).
Zhang, G. J. et al. Bioluminescent imaging of Cdk2 inhibition in vivo. Nature Med. 10, 643–648 (2004).
Wei, W., Jin, J., Schlisio, S., Harper, J. W. & Kaelin, W. G. Jr. The v-Jun point mutation allows c-Jun to escape GSK3-dependent recognition and destruction by the Fbw7 ubiquitin ligase. Cancer Cell 8, 25–33 (2005).
Acknowledgements
We thank William Kaelin Jr, Lewis Cantley, Roya Khosravi-Far and Susan Glueck for critical reading of the manuscript; James DeCaprio, Christoph Geisen, Ronald DePinho, Laura Benjamin, Suzanne Conzen, John Blenis and Peter Jackson for providing reagents; Ross Tomaino for his kind assistance on the mass spectrum analysis; Isaac Robinovitz for his technical support on the fluorescence microscopy; Pier Paolo Pandolfi for sharing unpublished data; and members of the Wei and Toker laboratories for useful discussions. W.W. is a Leukemia and Lymphoma Society Special Fellow, Kimmel Scholar and V Scholar. This work was supported in part by the Harvard Medical School Milton Fund (W.W.) and the Emerald Foundation, and by grants from the National Institutes of Health (W.G.K., CA076120; A.T., CA122099) and the Susan G. Komen Breast Cancer Foundation (R.Y.C., 0706963).
Author information
Authors and Affiliations
Contributions
D.G. and H.I. performed most of the experiments with the assistance of A.Tseng. R.Y.C. performed the shAkt1 and shAkt2 experiment to examine its effects on Skp2 Ser 72 phosphorylation. W.W. and A.T. designed the experiments. W.W. supervised the study. W.W. wrote the paper with the assistance of A.T. All authors commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 4968 kb)
Rights and permissions
About this article
Cite this article
Gao, D., Inuzuka, H., Tseng, A. et al. Phosphorylation by Akt1 promotes cytoplasmic localization of Skp2 and impairs APCCdh1-mediated Skp2 destruction. Nat Cell Biol 11, 397–408 (2009). https://doi.org/10.1038/ncb1847
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1847
This article is cited by
-
FOXO transcription factors as mediators of stress adaptation
Nature Reviews Molecular Cell Biology (2024)
-
Akt: a key transducer in cancer
Journal of Biomedical Science (2022)
-
ACK1 upregulated the proliferation of head and neck squamous cell carcinoma cells by promoting p27 phosphorylation and degradation
Journal of Cell Communication and Signaling (2022)
-
Prolonged cetuximab treatment promotes p27Kip1-mediated G1 arrest and autophagy in head and neck squamous cell carcinoma
Scientific Reports (2021)
-
Regulation of SKP2 protein stability by heat shock protein 90 chaperone machinery
Signal Transduction and Targeted Therapy (2021)