Abstract
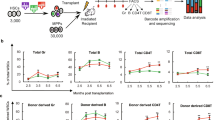
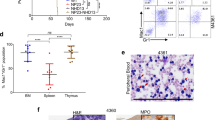
Recent studies have suggested that regeneration of non-haematopoietic cell lineages can occur through heterotypic cell fusion1,2,3 with haematopoietic cells of the myeloid lineage2,3,4,5,6. Here we show that lymphocytes also form heterotypic-fusion hybrids with cardiomyocytes, skeletal muscle, hepatocytes and Purkinje neurons. However, through lineage fate-mapping we demonstrate that such in vivo fusion of lymphoid and myeloid blood cells does not occur to an appreciable extent in steady-state adult tissues or during normal development. Rather, fusion of blood cells with different non-haematopoietic cell types is induced by organ-specific injuries or whole-body irradiation1,2,3,4,5,6,7,8,9,10, which has been used in previous studies to condition recipients of bone marrow transplants. Our findings demonstrate that blood cells of the lymphoid and myeloid lineages contribute to various non-haematopoietic tissues by forming rare fusion hybrids, but almost exclusively in response to injuries or inflammation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Nygren, J. M., Jovinge, S., Breitbach, M., Sawen, P., Röll, W., Hescheler, J., Taneera, J., Fleischmann, B. K. & Jacobsen, S. E. Bone marrow-derived hematopoietic cells generate cardiomyocytes at a low frequency through cell fusion, but not transdifferentiation. Nature Med. 10, 494–501 (2004).
Alvarez-Dolado, M., Pardal, R., Garcia-Verdugo, J. M., Fike, J. R., Lee, H. O., Pfeffer, K., Lois, C., Morrison, S. J. & Alvarez-Buylla, A. Fusion of bone-marrow-derived cells with Purkinje neurons, cardiomyocytes and hepatocytes. Nature 425, 968–973 (2003).
Camargo, F. D., Green, R., Capetenaki, Y., Jackson, K. A. & Goodell, M. A. Single hematopoietic stem cells generate skeletal muscle through myeloid intermediates. Nature Med. 9, 1520–1527 (2003).
Weimann, J. M., Johansson, C. B., Trejo, A. & Blau, H. M. Stable reprogrammed heterokaryons form spontaneously in Purkinje neurons after bone marrow transplant. Nature Cell Biol. 5, 959–966 (2003).
Corbel, S. Y., Lee, A., Yi, L., Duenas, J., Brazelton, T. R., Blau, H. M. & Rossi, F. M. Contribution of hematopoietic stem cells to skeletal muscle. Nature Med. 9, 1528–1532 (2003).
Camargo, F. D., Finegold, M. & Goodell, M. A. Hematopoietic myelomonocytic cells are the major source of hepatocyte fusion partners. J. Clin. Invest. 113, 1266–1270 (2004).
Willenbring, H., Bailey, A. S., Foster, M., Akkari, Y., Dorrell, C., Olson, S., Finegold, M., Fleming, W. H. & Grompe, M. Myelomonocytic cells are sufficient for therapeutic cell fusion in liver. Nature Med. 10, 744–748 (2004).
Vassilopoulos, G., Wang, P. R. & Russell, D. W. 2003. Transplanted bone marrow regenerates liver by cell fusion. Nature 422: 901–4.
Wang, X., Willenbring, H., Akkari, Y., Torimaru, Y., Foster, M., Al-Dhalimy, M., Lagasse, E., Finegold, M., Olson, S. & Grompe, M. Cell fusion is the principal source of bone-marrow-derived hepatocytes. Nature 422, 897–901 (2003).
Doyonnas, R., LaBarge, M. A., Sacco, A., Charlton, C. & Blau, H. M. Hematopoietic contribution to skeletal muscle regeneration by myelomonocytic precursors. Proc. Natl Acad. Sci USA 101, 13507–13512 (2004).
Pomerantz, J. & Blau, H. M. Nuclear reprogramming: a key to stem cell function in regenerative medicine. Nature Cell Biol. 6, 810–816 (2004).
Mombaerts, P., Iacomini, J., Johnson, R. S., Herrup, K., Tonegawa, S. & Papaioannou, V. E. RAG-1-deficient mice have no mature B and T lymphocytes. Cell 68, 869–877 (1992).
Okabe, M., Ikawa, M., Kominami, K., Nakanishi, T. & Nishimune, Y. 'Green mice' as a source of ubiquitous green cells. FEBS Lett 407, 313–319 (1997).
Priller, J., Persons, D. A., Klett, F. F., Kempermann, G., Kreutzberg, G. W. & Dirnagl, U. Neogenesis of cerebellar Purkinje neurons from gene-marked bone marrow cells in vivo. J. Cell Biol. 155, 733–738 (2001).
Priller, J., Flugel, A., Wehner, T., Boentert, M., Haas, C. A., Prinz, M., Fernandez-Klett, F., Prass, K., Bechmann, I., de Boer, B. A., Frotscher, M., Kreutzberg, G. W., Persons, D. A. & Dirnagl, U. Targeting gene-modified hematopoietic cells to the central nervous system: use of green fluorescent protein uncovers microglial engraftment. Nature Med. 7, 1356–1361 (2001).
Cao, X., Shores, E. W., Hu-Li, J., Anver, M. R., Kelsall, B. L., Russell, S. M., Drago, J., Noguchi, M., Grinberg, A., Bloom, E. T. & et al. Defective lymphoid development in mice lacking expression of the common cytokine receptor gamma chain. Immunity 2, 223–38 (1995).
Lagasse, E., Connors, H., Al-Dhalimy, M., Reitsma, M., Dohse, M., Osborne, L., Wang, X., Finegold, M., Weissman, I. L. & Grompe, M. Purified hematopoietic stem cells can differentiate into hepatocytes in vivo. Nature Med. 6, 1229–1234 (2000).
Palermo, A. T., Labarge, M. A., Doyonnas, R., Pomerantz, J. & Blau, H. M. Bone marrow contribution to skeletal muscle: a physiological response to stress. Dev. Biol. 279, 336–344 (2005).
LaBarge, M. A. & Blau, H. M. Biological progression from adult bone marrow to mononucleate muscle stem cell to multinucleate muscle fiber in response to injury. Cell 111, 589–601 (2002).
Reith, A. D., Rottapel, R., Giddens, E., Brady, C., Forrester, L. & Bernstein, A. W mutant mice with mild or severe developmental defects contain distinct point mutations in the kinase domain of the c-kit receptor. Genes Dev. 4, 390–400 (1990).
Irintchev, A., Zweyer, M., Cooper, R. N., Butler-Browne, G. S. & Wernig, A. Contractile properties, structure and fiber phenotype of intact and regenerating slow-twitch muscles of mice treated with cyclosporin A. Cell Tissue Res. 308, 143–156 (2002).
Maetzler, W., Nitsch, C., Bendfeldt, K., Racay, P., Vollenweider, F. & Schwaller, B. Ectopic parvalbumin expression in mouse forebrain neurons increases excitotoxic injury provoked by ibotenic acid injection into the striatum. Exp. Neurol. 186, 78–88 (2004).
Ogle, B. M., Cascalho, M. & Platt, J. L. Biological implications of cell fusion. Nature Rev. Mol. Cell Biol. (2005).
Zambrowicz, B. P., Imamoto, A., Fiering, S., Herzenberg, L. A., Kerr, W. G. & Soriano, P. Disruption of overlapping transcripts in the ROSA β geo 26 gene trap strain leads to widespread expression of β-galactosidase in mouse embryos and hematopoietic cells. Proc.Natl Acad. Sci. USA 94, 3789–3794 (1997).
Hallahan, D., Kuchibhotla, J. & Wyble, C. Cell adhesion molecules mediate radiation-induced leukocyte adhesion to the vascular endothelium. Cancer Res. 56, 5150–5155 (1996).
Stadtfeld, M. & Graf, T. Assessing the role of hematopoietic plasticity for endothelial and hepatocyte development by non-invasive lineage tracing. Development 132, 203–213 (2005).
Stadtfeld, M., Ye, M. & Graf, T. Identification of interventricular septum precursor cells in the mouse embryo. Dev. Biol. (2007).
Kawada, H., Fujita, J., Kinjo, K., Matsuzaki, Y., Tsuma, M., Miyatake, H., Muguruma, Y., Tsuboi, K., Itabashi, Y., Ikeda, Y., Ogawa, S., Okano, H., Hotta, T., Ando, K. & Fukuda, K. Nonhematopoietic mesenchymal stem cells can be mobilized and differentiate into cardiomyocytes after myocardial infarction. Blood 104, 3581–3587 (2004).
Jackson, K. A., Snyder, D. S. & Goodell, M. A. Skeletal muscle fiber-specific green autofluorescence: potential for stem cell engraftment artifacts. Stem Cells 22, 180–187 (2004).
Jackson, K. A., Majka, S. M., Wang, H., Pocius, J., Hartley, C. J., Majesky, M. W., Entman, M. L., Michael, L. H., Hirschi, K. K. & Goodell, M. A. Regeneration of ischemic cardiac muscle and vascular endothelium by adult stem cells. J. Clin. Invest. 107, 1395–1402 (2001).
Acknowledgements
We thank L. Wittman, U. Jarl, A. Josefsson and E. Cordero for reliable technical assistance. This work was supported by the Swedish Childhood Cancer Foundation, the Swedish Heart Lung Foundation, The Juvenile Diabetes Research Foundation, the Swedish Diabetes Foundation, the Swedish Research Council, the Deutsche Forschungsgemeinschaft (FL 276/4–2/3) and the Scientific Exchange Program, North Rhine Westphalia-Sweden. The Lund Stem Cell Center is supported by a Center of Excellence grant in life sciences from the Swedish Foundation for Strategic Research.
Author information
Authors and Affiliations
Contributions
J. M. N., K. L., L. T., C. N. and S. E. W. J. designed, performed and analysed bone marrow transplantation models; J. M. N., M. B., B. F. K. and S. E. W. J. designed, performed and analysed muscle injury models. J. M. N., S. S., D. K., A. B. and S. E. W. J. designed, performed and analysed brain injury models; J. M. N., M. B., W. R., B. K. J., S. J. and S. E. W. J. designed, performed and analysed heart injury models. J. M. N., K. L., S. J. and S. E. W. J. designed, performed and analysed irradiation injury models; J. M. N., M. B., S. S., C. G., P. S., B. K. F., S. J. and S. E. W. J. designed, performed and analysed fused cell characterization; J. M. N., K. L., S. S., D. K., A. B., S. J. and S. E. W. J. performed and analysed fetal and neonatal transplantation models; J. M. N. and S. E. W. J. wrote the manuscript and K. L., M. B., S. S. L. T., W. R., C. G., P. S., D. K., A. B., C. N., B. K. F. and S. J. reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1, S2, S3, S4, S5, S6 and Supplemental Methods (PDF 1495 kb)
Rights and permissions
About this article
Cite this article
Nygren, J., Liuba, K., Breitbach, M. et al. Myeloid and lymphoid contribution to non-haematopoietic lineages through irradiation-induced heterotypic cell fusion. Nat Cell Biol 10, 584–592 (2008). https://doi.org/10.1038/ncb1721
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1721
This article is cited by
-
Cell Therapy Strategies on Duchenne Muscular Dystrophy: A Systematic Review of Clinical Applications
Stem Cell Reviews and Reports (2024)
-
The impact and outcomes of cancer-macrophage fusion
BMC Cancer (2023)
-
Cell fusion upregulates PD-L1 expression for evasion from immunosurveillance
Cancer Gene Therapy (2023)
-
Direct cell-to-cell transfer in stressed tumor microenvironment aggravates tumorigenic or metastatic potential in pancreatic cancer
npj Genomic Medicine (2022)
-
Mesenchymal stem cell-mediated transfer of mitochondria: mechanisms and functional impact
Cellular and Molecular Life Sciences (2022)