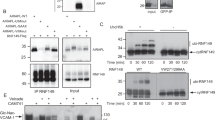
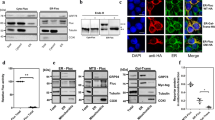
Abstract
Terminally misfolded or unassembled proteins in the early secretory pathway are degraded by a ubiquitin- and proteasome-dependent process known as ER-associated degradation (ERAD). How substrates of this pathway are recognized within the ER and delivered to the cytoplasmic ubiquitin-conjugating machinery is unknown. We report here that OS-9 and XTP3-B/Erlectin are ER-resident glycoproteins that bind to ERAD substrates and, through the SEL1L adaptor, to the ER-membrane-embedded ubiquitin ligase Hrd1. Both proteins contain conserved mannose 6-phosphate receptor homology (MRH) domains, which are required for interaction with SEL1L, but not with substrate. OS-9 associates with the ER chaperone GRP94 which, together with Hrd1 and SEL1L, is required for the degradation of an ERAD substrate, mutant α1-antitrypsin. These data suggest that XTP3-B and OS-9 are components of distinct, partially redundant, quality control surveillance pathways that coordinate protein folding with membrane dislocation and ubiquitin conjugation in mammalian cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Hammond, C. & Helenius, A. Quality control in the secretory pathway. Curr. Opin. Cell Biol. 7, 523–529 (1995).
Lippincott-Schwartz, J., Bonifacino, J. S., Yuan, L. C. & Klausner, R. D. Degradation from the endoplasmic reticulum: disposing of newly synthesized proteins. Cell 54, 209–220 (1988).
McCracken, A. A. & Brodsky, J. L. Assembly of ER-associated protein degradation in vitro: dependence on cytosol, calnexin, and ATP. J. Cell Biol. 132, 291–298 (1996).
Meusser, B., Hirsch, C., Jarosch, E. & Sommer, T. ERAD: the long road to destruction. Nature Cell Biol. 7, 766–772 (2005).
Yu, H. & Kopito, R. R. The role of multiubiquitination in dislocation and degradation of the α subunit of the T cell antigen receptor. J. Biol. Chem. 274, 36852–36858 (1999).
Shamu, C. E., Flierman, D., Ploegh, H. L., Rapoport, T. A. & Chau, V. Polyubiquitination is required for US11-dependent movement of MHC class I heavy chain from endoplasmic reticulum into cytosol. Mol. Biol. Cell 12, 2546–2555. (2001).
Jarosch, E. et al. Protein dislocation from the ER requires polyubiquitination and the AAA-ATPase Cdc48. Nature Cell Biol. 4, 134–139 (2002).
Carvalho, P., Goder, V. & Rapoport, T. A. Distinct ubiquitin-ligase complexes define convergent pathways for the degradation of ER proteins. Cell 126, 361–373 (2006).
Denic, V., Quan, E. M. & Weissman, J. S. A luminal surveillance complex that selects misfolded glycoproteins for ER-associated degradation. Cell 126, 349–359 (2006).
Schuberth, C. & Buchberger, A. Membrane-bound Ubx2 recruits Cdc48 to ubiquitin ligases and their substrates to ensure efficient ER-associated protein degradation. Nature Cell Biol. 7, 999–1006 (2005).
Neuber, O., Jarosch, E., Volkwein, C., Walter, J. & Sommer, T. Ubx2 links the Cdc48 complex to ER-associated protein degradation. Nature Cell Biol. 7, 993–998 (2005).
Lilley, B. N. & Ploegh, H. L. Multiprotein complexes that link dislocation, ubiquitination, and extraction of misfolded proteins from the endoplasmic reticulum membrane. Proc. Natl Acad. Sci. USA 102, 14296–14301 (2005).
Schulze, A. et al. The ubiquitin-domain protein HERP forms a complex with components of the endoplasmic reticulum associated degradation pathway. J. Mol. Biol. 354, 1021–1027 (2005).
Hebert, D. N., Garman, S. C. & Molinari, M. The glycan code of the endoplasmic reticulum: asparagine-linked carbohydrates as protein maturation and quality-control tags. Trends Cell Biol. 15, 364–370 (2005).
Molinari, M., Calanca, V., Galli, C., Lucca, P. & Paganetti, P. Role of EDEM in the release of misfolded glycoproteins from the calnexin cycle. Science. 299, 1397–1400 (2003).
Oda, Y., Hosokawa, N., Wada, I. & Nagata, K. EDEM as an acceptor of terminally misfolded glycoproteins released from calnexin. Science. 299, 1394–1397 (2003).
Bhamidipati, A., Denic, V., Quan, E. M. & Weissman, J. S. Exploration of the topological requirements of ERAD identifies Yos9p as a lectin sensor of misfolded glycoproteins in the ER lumen. Mol. Cell 19, 741–751 (2005).
Kim, W., Spear, E. D. & Ng, D. T. Yos9p detects and targets misfolded glycoproteins for ER-associated degradation. Mol. Cell 19, 753–764 (2005).
Szathmary, R., Bielmann, R., Nita-Lazar, M., Burda, P. & Jakob, C. A. Yos9 protein is essential for degradation of misfolded glycoproteins and may function as lectin in ERAD. Mol. Cell 19, 765–775 (2005).
Buschhorn, B. A., Kostova, Z., Medicherla, B. & Wolf, D. H. A genome-wide screen identifies Yos9p as essential for ER-associated degradation of glycoproteins. FEBS Lett. 577, 422–426 (2004).
Gauss, R., Jarosch, E., Sommer, T. & Hirsch, C. A complex of Yos9p and the HRD ligase integrates endoplasmic reticulum quality control into the degradation machinery. Nature Cell Biol. 8, 849–854 (2006).
Cruciat, C. M., Hassler, C. & Niehrs, C. The MRH protein Erlectin is a member of the endoplasmic reticulum synexpression group and functions in N-glycan recognition. J. Biol. Chem. 281, 12986–12993 (2006).
Su, Y. A., Hutter, C. M., Trent, J. M. & Meltzer, P. S. Complete sequence analysis of a gene (OS-9) ubiquitously expressed in human tissues and amplified in sarcomas. Mol. Carcinog. 15, 270–275 (1996).
Kimura, Y., Nakazawa, M. & Yamada, M. Cloning and characterization of three isoforms of OS-9 cDNA and expression of the OS-9 gene in various human tumor cell lines. J. Biochem. (Tokyo) 123, 876–882 (1998).
Nakayama, T., Yaoi, T., Kuwajima, G., Yoshie, O. & Sakata, T. Ca2+-dependent interaction of N-copine, a member of the two C2 domain protein family, with OS-9, the product of a gene frequently amplified in osteosarcoma. FEBS Lett. 453, 77–80 (1999).
Litovchick, L., Friedmann, E. & Shaltiel, S. A selective interaction between OS-9 and the carboxyl-terminal tail of meprin beta. J. Biol. Chem. 277, 34413–34423 (2002).
Baek, J. H. et al. OS-9 interacts with hypoxia-inducible factor 1α and prolyl hydroxylases to promote oxygen-dependent degradation of HIF-1α. Mol. Cell 17, 503–512 (2005).
Mueller, B., Lilley, B. N. & Ploegh, H. L. SEL1L, the homologue of yeast Hrd3p, is involved in protein dislocation from the mammalian ER. J. Cell Biol. 175, 261–270 (2006).
Grant, B. & Greenwald, I. The Caenorhabditis elegans sel-1 gene, a negative regulator of lin-12 and glp-1, encodes a predicted extracellular protein. Genetics 143, 237–247 (1996).
Hirao, K. et al. EDEM3, a soluble EDEM homolog, enhances glycoprotein ERAD and mannose trimming. J. Biol. Chem. 281, 9650–9658 (2006).
Oda, Y. et al. Derlin-2 and Derlin-3 are regulated by the mammalian unfolded protein response and are required for ER-associated degradation. J. Cell Biol. 172, 383–393 (2006).
de Virgilio, M., Weninger, H. & Ivessa, N. E. Ubiquitination is required for the retro-translocation of a short-lived luminal endoplasmic reticulum glycoprotein to the cytosol for degradation by the proteasome. J. Biol. Chem. 273, 9734–9743 (1998).
Yu, H., Kaung, G., Kobayashi, S. & Kopito, R. R. Cytosolic degradation of T-cell receptor alpha chains by the proteasome. J. Biol. Chem. 272, 20800–20804 (1997).
Huppa, J. B. & Ploegh, H. L. The α chain of the T cell antigen receptor is degraded in the cytosol. Immunity 7, 113–122 (1997).
Johnson, S. M. et al. Native state kinetic stabilization as a strategy to ameliorate protein misfolding diseases: a focus on the transthyretin amyloidoses. Acc. Chem. Res. 38, 911–921 (2005).
Sekijima, Y. et al. The biological and chemical basis for tissue-selective amyloid disease. Cell 121, 73–85 (2005).
Young, J. C., Moarefi, I. & Hartl, F. U. Hsp90: a specialized but essential protein-folding tool. J. Cell Biol. 154, 267–273 (2001).
Yang, Y. et al. Heat shock protein gp96 is a master chaperone for toll-like receptors and is important in the innate function of macrophages. Immunity 26, 215–226 (2007).
Randow, F. & Seed, B. Endoplasmic reticulum chaperone gp96 is required for innate immunity but not cell viability. Nature Cell Biol. 3, 891–896 (2001).
Pearl, L. H. & Prodromou, C. Structure and mechanism of the Hsp90 molecular chaperone machinery. Annu. Rev. Biochem. 75, 271–294 (2006).
Gardner, R.G. et al. Endoplasmic reticulum degradation requires lumen to cytosol signaling. Transmembrane control of Hrd1p by Hrd3p. J. Cell Biol. 151, 69–82 (2000).
Kislinger, T. et al. Global survey of organ and organelle protein expression in mouse: combined proteomic and transcriptomic profiling. Cell 125, 173–186 (2006).
Yuan, B., Latek, R., Hossbach, M., Tuschl, T. & Lewitter, F. siRNA Selection Server: an automated siRNA oligonucleotide prediction server. Nucleic Acids Res. 32, W130–W134 (2004).
Elbashir, S. M. et al. Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature 411, 494–498 (2001).
DeLaBarre, B., Christianson, J. C., Kopito, R. R. & Brunger, A. T. Central pore residues mediate the p97/VCP activity required for ERAD. Mol. Cell 22, 451–462 (2006).
Christianson, J. C. & Green, W. N. Regulation of nicotinic receptor expression by the ubiquitin-proteasome system. EMBO J. 23, 4156–4165 (2004).
Ward, C. L. & Kopito, R. R. Intracellular turnover of cystic fibrosis transmembrane conductance regulator. Inefficient processing and rapid degradation of wild-type and mutant proteins. J. Biol. Chem. 269, 25710–25718 (1994).
Stephens, S., Dodd, R., Lerner, R., Pyhtila, B. & Nicchitta, C. Analysis of mRNA Partitioning Between the Cytosol and Endoplasmic Reticulum Compartments of Mammalian Cells. in Post-transcriptional gene regulation. Methods in Molecular Biology, Vol. 419. (ed. J. Wilusz), 197–214 (Humana Press, Totowa, NJ, 2007).
Hosokawa, N., You, Z., Tremblay, L. O., Nagata, K. & Herscovics, A. Stimulation of ERAD of misfolded null Hong Kong α1-antitrypsin by Golgi α2-mannosidases. Biochem. Biophys. Res. Commun. 362, 626–632 (2007).
Liu, Y., Choudhury, P., Cabral, C. M. & Sifers, R. N. Oligosaccharide modifications in the early secretory pathway directs the selection of a misfolded glycoprotein for degradation by the proteosome. J. Biol. Chem. 274, 5861–5867 (1999).
Acknowledgements
We thank J. Weissman, I. Biunno, V. Chau, C. Nicchitta, H. Ploegh, G. Semenza, J. Kelly and E. Wiertz for reagents and technical advice, members of the Kopito lab for helpful discussion, and S. Duttler and J. Olzmann for critical reading of the manuscript. This work was supported by grants from NIDDK, NIGMS, and the Cystic Fibrosis Foundation. JCC was supported by a postdoctoral fellowship from Cystic Fibrosis Research, Inc.
Author information
Authors and Affiliations
Contributions
J. C. C. performed all experiments and data analysis, with the exception of Table 1 and Supplementary Information, Fig. 4, which were carried out by T. A. S. and R. E. M.; J. C. C. and R. R. K. wrote the manuscript.
Corresponding author
Supplementary information
Supplementary Information
Supplementary figures S1, S2, S3, S4, S5 and table S1 (PDF 606 kb)
Rights and permissions
About this article
Cite this article
Christianson, J., Shaler, T., Tyler, R. et al. OS-9 and GRP94 deliver mutant α1-antitrypsin to the Hrd1–SEL1L ubiquitin ligase complex for ERAD. Nat Cell Biol 10, 272–282 (2008). https://doi.org/10.1038/ncb1689
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1689
This article is cited by
-
SEL1L-HRD1 interaction is required to form a functional HRD1 ERAD complex
Nature Communications (2024)
-
SEL1L preserves CD8+ T-cell survival and homeostasis by fine-tuning PERK signaling and the IL-15 receptor-mediated mTORC1 axis
Cellular & Molecular Immunology (2023)
-
USP2 promotes tumor immune evasion via deubiquitination and stabilization of PD-L1
Cell Death & Differentiation (2023)
-
Mechanisms of substrate processing during ER-associated protein degradation
Nature Reviews Molecular Cell Biology (2023)
-
Evolution patterns of NBS genes in the genus Dendrobium and NBS-LRR gene expression in D. officinale by salicylic acid treatment
BMC Plant Biology (2022)