Abstract
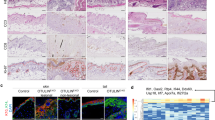
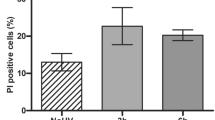
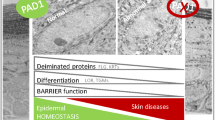
Caspase-14 belongs to a conserved family of aspartate-specific proteinases. Its expression is restricted almost exclusively to the suprabasal layers of the epidermis and the hair follicles1,2,3,4. Moreover, the proteolytic activation of caspase-14 is associated with stratum corneum formation, implicating caspase-14 in terminal keratinocyte differentiation and cornification5,6. Here, we show that the skin of caspase-14-deficient mice was shiny and lichenified, indicating an altered stratum-corneum composition. Caspase-14-deficient epidermis contained significantly more alveolar keratohyalin F-granules, the profilaggrin stores. Accordingly, caspase-14-deficient epidermis is characterized by an altered profilaggrin processing pattern and we show that recombinant caspase-14 can directly cleave profilaggrin in vitro. Caspase-14-deficient epidermis is characterized by reduced skin-hydration levels and increased water loss. In view of the important role of filaggrin in the structure and moisturization of the skin, the knockout phenotype could be explained by an aberrant processing of filaggrin. Importantly, the skin of caspase-14-deficient mice was highly sensitive to the formation of cyclobutane pyrimidine dimers after UVB irradiation, leading to increased levels of UVB-induced apoptosis. Removal of the stratum corneum indicate that caspase-14 controls the UVB scavenging capacity of the stratum corneum.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Eckhart, L. et al. Terminal differentiation of human keratinocytes and stratum corneum formation is associated with caspase-14 activation. J. Invest. Dermatol. 115, 1148–1151 (2000).
Lippens, S. et al. Epidermal differentiation does not involve the pro-apoptotic executioner caspases, but is associated with caspase-14 induction and processing. Cell Death Differ. 7, 1218–1224 (2000).
Lippens, S. et al. Vitamin D3 induces caspase-14 expression in psoriatic lesions and enhances caspase-14 processing in organotypic skin cultures. Am. J. Pathol. 165, 833–841 (2004).
Fischer, H. et al. Stratum corneum-derived caspase-14 is catalytically active. FEBS Lett. 577, 446–450 (2004).
Candi, E., Schmidt, R. & Melino, G. The cornified envelope: a model of cell death in the skin. Nature Rev. Mol. Cell Biol. 6, 328–340 (2005).
Lippens, S., Denecker, G., Ovaere, P., Vandenabeele, P. & Declercq, W. Death penalty for keratinocytes: apoptosis versus cornification. Cell Death Differ. 12, 1497–1508 (2005).
Rendl, M. et al. Caspase-14 expression by epidermal keratinocytes is regulated by retinoids in a differentiation-associated manner. J. Invest. Dermatol. 119, 1150–1155 (2002).
Mikolajczyk, J., Scott, F. L., Krajewski, S., Sutherlin, D. P. & Salvesen, G. S. Activation and substrate specificity of caspase-14. Biochemistry 43, 10560–10569 (2004).
Lippens, S. et al. Caspase-14 is expressed in the epidermis, the choroid plexus, the retinal pigment epithelium and thymic Hassall's bodies. Cell Death Differ. 10, 257–259 (2003).
Alibardi, L., Dockal, M., Reinisch, C., Tschachler, E. & Eckhart, L. Ultrastructural localization of caspase-14 in human epidermis. J. Histochem. Cytochem. 52, 1561–1574 (2004).
Matsuki, M. et al. Defective stratum corneum and early neonatal death in mice lacking the gene for transglutaminase 1 (keratinocyte transglutaminase). Proc. Natl Acad. Sci. USA 95, 1044–1049 (1998).
Smith, F. J. et al. Loss-of-function mutations in the gene encoding filaggrin cause ichthyosis vulgaris. Nature Genet. 38, 337–342 (2006).
Presland, R. B. et al. Loss of normal profilaggrin and filaggrin in flaky tail (ft/ft) mice: an animal model for the filaggrin-deficient skin disease ichthyosis vulgaris. J. Invest. Dermatol. 115, 1072–1081 (2000).
Scott, I. R. & Harding, C. R. Filaggrin breakdown to water binding compounds during development of the rat stratum corneum is controlled by the water activity of the environment. Dev. Biol. 115, 84–92 (1986).
Presland, R. B. et al. Evidence for specific proteolytic cleavage of the N-terminal domain of human profilaggrin during epidermal differentiation. J. Invest. Dermatol. 108, 170–178 (1997).
Palmer, C. N. et al. Common loss-of-function variants of the epidermal barrier protein filaggrin are a major predisposing factor for atopic dermatitis. Nature Genet. 38, 441–446 (2006).
Fischer, H. et al. Caspase-14 but not caspase-3 is processed during the development of fetal mouse epidermis. Differentiation 73, 406–413 (2005).
Hardman, M. J., Sisi, P., Banbury, D. N. & Byrne, C. Patterned acquisition of skin barrier function during development. Development 125, 1541–1552 (1998).
Lu, Y. P., Lou, Y. R., Peng, Q. Y., Xie, J. G. & Conney, A. H. Stimulatory effect of topical application of caffeine on UVB-induced apoptosis in the epidermis of p53 and Bax knockout mice. Cancer Res. 64, 5020–5027 (2004).
Jacks, T. et al. Tumor spectrum analysis in p53-mutant mice. Curr. Biol. 4, 1–7 (1994).
Kulms, D. & Schwarz, T. Molecular mechanisms of UV-induced apoptosis. Photodermatol. Photoimmunol. Photomed. 16, 195–201 (2000).
Rawlings, A. V. & Matts, P. J. Stratum corneum moisturization at the molecular level: an update in relation to the dry skin cycle. J. Invest. Dermatol. 124, 1099–1110 (2005).
Krysko, D. V. et al. Macrophages use different internalization mechanisms to clear apoptotic and necrotic cells. Cell Death Differ. 13, 2011–2022 (2006).
Schwarz, A. et al. Interleukin-12 suppresses ultraviolet radiation-induced apoptosis by inducing DNA repair. Nature Cell Biol. 4, 26–31 (2002).
Caldelari, R., Suter, M. M., Baumann, D., De Bruin, A. & Muller, E. Long-term culture of murine epidermal keratinocytes. J. Invest. Dermatol. 114, 1064–1065 (2000).
Presland, R. B., Haydock, P. V., Fleckman, P., Nirunsuksiri, W. & Dale, B. A. Characterization of the human epidermal profilaggrin gene. J. Biol. Chem. 267, 23772–23781 (1992).
Denecker, G. et al. Death receptor-induced apoptotic and necrotic cell death: differential role of caspases and mitochondria. Cell Death Differ. 8, 829–840 (2001).
Carmeliet, P. et al. Impaired myocardial angiogenesis and ischemic cardiomyopathy in mice lacking the vascular endothelial growth factor isoforms VEGF164 and VEGF188. Nature Med. 5, 495–502 (1999).
Schoonjans, L. et al. Improved generation of germline-competent embryonic stem cell lines from inbred mouse strains. Stem Cells 21, 90–97 (2003).
Chien, A. J., Presland, R. B. & Kuechle, M. K. Processing of native caspase-14 occurs at an atypical cleavage site in normal epidermal differentiation. Biochem. Biophys. Res. Commun. 296, 911–917 (2002).
Acknowledgements
We thank A. Bredan for editing the manuscript and E. van Damme, A. Meeus and W. Deckers for technical assistance. This work was supported in part by the Interuniversitaire Attractiepolen V, the Fonds voor Wetenschappelijk Onderzoek-Vlaanderen, the Epistem 6th framework EC-RTD grant and Ghent University GOA project. G.D. is a postdoctoral fellow at the Fonds voor Wetenschappelijk Onderzoek-Vlaanderen, E.H. has an Instituut voor de Aanmoediging van Innovatie door Wetenschap en Technologie (IWT) predoctoral grant and P.O. had an Emmanuel Verscheuren and an IWT predoctoral grant. R.P. was supported by R01 AR49183 from the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1, S2, S3, S4 and S5 (PDF 570 kb)
Rights and permissions
About this article
Cite this article
Denecker, G., Hoste, E., Gilbert, B. et al. Caspase-14 protects against epidermal UVB photodamage and water loss. Nat Cell Biol 9, 666–674 (2007). https://doi.org/10.1038/ncb1597
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1597
This article is cited by
-
Comparative genomics of monotremes provides insights into the early evolution of mammalian epidermal differentiation genes
Scientific Reports (2024)
-
Human skin specific long noncoding RNA HOXC13-AS regulates epidermal differentiation by interfering with Golgi-ER retrograde transport
Cell Death & Differentiation (2023)
-
A Review on Caspases: Key Regulators of Biological Activities and Apoptosis
Molecular Neurobiology (2023)
-
Study of the protective effects of cosmetic ingredients on the skin barrier, based on the expression of barrier-related genes and cytokines
Molecular Biology Reports (2022)
-
The Genetics of Eczema Herpeticum
Clinical Reviews in Allergy & Immunology (2022)