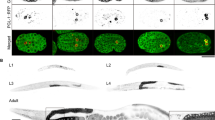
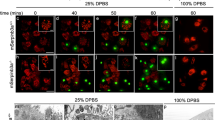
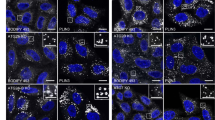
Abstract
Externalization of phosphatidylserine, which is normally restricted to the inner leaflet of plasma membrane, is a hallmark of mammalian apoptosis1,2,3,4. It is not known what activates and mediates the phosphatidylserine externalization process in apoptotic cells. Here, we report the development of an annexin V-based phosphatidylserine labelling method and show that a majority of apoptotic germ cells in Caenorhabditis elegans have surface-exposed phosphatidylserine, indicating that phosphatidylserine externalization is a conserved apoptotic event in worms. Importantly, inactivation of the gene encoding either the C. elegans apoptosis-inducing factor (AIF) homologue (WAH-1)5, a mitochondrial apoptogenic factor, or the C. elegans phospholipid scramblase 1 (SCRM-1), a plasma membrane protein, reduces phosphatidylserine exposure on the surface of apoptotic germ cells and compromises cell-corpse engulfment. WAH-1 associates with SCRM-1 and activates its phospholipid scrambling activity in vitro. Thus WAH-1, after its release from mitochondria during apoptosis, promotes plasma membrane phosphatidylserine externalization through its downstream effector, SCRM-1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Fadok, V. A. et al. Exposure of phosphatidylserine on the surface of apoptotic lymphocytes triggers specific recognition and removal by macrophages. J. Immunol. 148, 2207–2216 (1992).
Ashman, R. F., Peckham, D., Alhasan, S. & Stunz, L. L. Membrane unpacking and the rapid disposal of apoptotic cells. Immunol. Lett. 48, 159–166 (1995).
Martin, S. J. et al. Early redistribution of plasma membrane phosphatidylserine is a general feature of apoptosis regardless of the initiating stimulus: inhibition by overexpression of Bcl-2 and Abl. J. Exp. Med. 182, 1545–1556 (1995).
Verhoven, B., Schlegel, R. A. & Williamson, P. Mechanisms of phosphatidylserine exposure, a phagocyte recognition signal, on apoptotic T lymphocytes. J. Exp. Med. 182, 1597–1601 (1995).
Wang, X., Yang, C., Chai, J., Shi, Y. & Xue, D. Mechanisms of AIF-mediated apoptotic DNA degradation in Caenorhabditis elegans. Science 298, 1587–1592 (2002).
Gumienny, T. L., Lambie, E., Hartwieg, E., Horvitz, H. R. & Hengartner, M. O. Genetic control of programmed cell death in the Caenorhabditis elegans hermaphrodite germline. Development 126, 1011–1022 (1999).
Hedgecock, E. M., Sulston, J. E. & Thomson, J. N. Mutations affecting programmed cell deaths in the nematode Caenorhabditis elegans. Science 220, 1277–1279 (1983).
Susin, S. A. et al. Molecular characterization of mitochondrial apoptosis-inducing factor. Nature 397, 441–446 (1999).
Loeffler, M. et al. Dominant cell death induction by extramitochondrially targeted apoptosis-inducing factor. FASEB J. 15, 758–767 (2001).
Tenenhaus, C., Subramaniam, K., Dunn, M. A. & Seydoux, G. PIE-1 is a bifunctional protein that regulates maternal and zygotic gene expression in the embryonic germ line of Caenorhabditis elegans. Genes Dev. 15, 1031–1040 (2001).
Fadok, V. A., Xue, D. & Henson, P. If phosphatidylserine is the death knell, a new phosphatidylserine- specific receptor is the bellringer. Cell Death Differ. 8, 582–587 (2001).
Reddien, P. W. & Horvitz, H. R. The engulfment process of programmed cell death in caenorhabditis elegans. Annu. Rev. Cell Dev. Biol. 20, 193–221 (2004).
Ellis, R. E., Jacobson, D. M. & Horvitz, H. R. Genes required for the engulfment of cell corpses during programmed cell death in Caenorhabditis elegans. Genetics 129, 79–94 (1991).
Zhou, Z., Hartwieg, E. & Horvitz, H. R. CED-1 is a transmembrane receptor that mediates cell corpse engulfment in C. elegans. Cell 104, 43–56 (2001).
Wu, Y. C. & Horvitz, H. R. The C. elegans cell corpse engulfment gene ced-7 encodes a protein similar to ABC transporters. Cell 93, 951–960 (1998).
Wang, X. et al. Cell corpse engulfment mediated by C. elegans phosphatidylserine receptor through CED-5 and CED-12. Science 302, 1563–1566 (2003).
Zhou, Q. et al. Molecular cloning of human plasma membrane phospholipid scramblase. A protein mediating transbilayer movement of plasma membrane phospholipids. J. Biol. Chem. 272, 18240–18244 (1997).
Wiedmer, T., Zhou, Q., Kwoh, D. Y. & Sims, P. J. Identification of three new members of the phospholipid scramblase gene family. Biochim. Biophys. Acta. 1467, 244–253 (2000).
Frasch, S. C. et al. Regulation of phospholipid scramblase activity during apoptosis and cell activation by protein kinase Cdelta. J. Biol. Chem. 275, 23065–23073 (2000).
Zhou, Q., Sims, P. J. & Wiedmer, T. Identity of a conserved motif in phospholipid scramblase that is required for Ca2+-accelerated transbilayer movement of membrane phospholipids. Biochemistry 37, 2356–2360 (1998).
Ye, H. et al. DNA binding is required for the apoptogenic action of apoptosis inducing factor. Nature Struct Biol. 9, 680–684 (2002).
Vahsen, N. et al. Physical interaction of apoptosis-inducing factor with DNA and RNA. Oncogene 25, 1763–1774 (2006).
Balasubramanian, K. & Schroit, A. J. Aminophospholipid asymmetry: A matter of life and death. Annu. Rev. Physiol. 65, 701–734 (2003).
Gartner, A., Milstein, S., Ahmed, S., Hodgkin, J. & Hengartner, M. O. A conserved checkpoint pathway mediates DNA damage-induced apoptosis and cell cycle arrest in C. elegans. Mol. Cell 5, 435–443 (2000).
Woodle, M. C. & Papahadjopoulos, D. Liposome preparation and size characterization. Methods Enzymol. 171, 193–217 (1989).
Acknowledgements
We thank T. Blumenthal for anti-CstF-64 antibody and Geraldine Seydoux for the pTE5 construct. This research was supported by a Burroughs Wellcome Fund Career Award and a Human Frontier Science Program (HFSP) grant (RGP0016/2005-C) to D.X., a grant from the Ministry of Education, Culture, Sports, Science and Technology of Japan to S.M., and grants from the National Institutes of Health (NIH) to D.X., X.S.X. and Y.S.
Author information
Authors and Affiliations
Contributions
X.C.W. performed most of the experiments. X.C.W. and D.X. designed and interpreted most of the experiments. J.W. and X.S.W. performed the in vitro proteoliposome assays and related data analysis. K.G.A., T.K. and S.M. isolated scrm deletion alleles. L.G., Y.G.S., C.L.S., Y.S. and C.L.Y. contributed to the experiments. X.C.W., X.S.X. and D.X. wrote the paper and others commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1, S2, S3, S4, S5, Supplementary Methods, Supplementary Tables S1, S2, S3, S4 and References (PDF 5201 kb)
Rights and permissions
About this article
Cite this article
Wang, X., Wang, J., Gengyo-Ando, K. et al. C. elegans mitochondrial factor WAH-1 promotes phosphatidylserine externalization in apoptotic cells through phospholipid scramblase SCRM-1. Nat Cell Biol 9, 541–549 (2007). https://doi.org/10.1038/ncb1574
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1574
This article is cited by
-
Metronidazole Determination in Raw Milk with a Graphene Aerogel-Based Electrochemiluminescent Sensor and Its Effect on Cell Apoptosis
Food Analytical Methods (2021)
-
Molecular cloning and biochemical characterization of the phospholipid scramblase SCRM-1 from Caenorhabditis elegans
European Biophysics Journal (2020)
-
AEBP1 down regulation induced cell death pathway depends on PTEN status of glioma cells
Scientific Reports (2019)
-
Regulation of CED-3 caspase localization and activation by C. elegans nuclear-membrane protein NPP-14
Nature Structural & Molecular Biology (2016)
-
Programmed cell death and clearance of cell corpses in Caenorhabditis elegans
Cellular and Molecular Life Sciences (2016)