Abstract
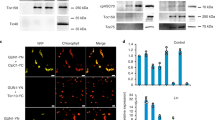
In contrast to animal and fungal cells, green plant cells contain one or multiple chloroplasts, the organelle(s) in which photosynthetic reactions take place. Chloroplasts are believed to have originated from an endosymbiotic event and contain DNA that codes for some of their proteins. Most chloroplast proteins are encoded by the nuclear genome and imported with the help of sorting signals that are intrinsic parts of the polypeptides. Here, we show that a chloroplast-located protein in higher plants takes an alternative route through the secretory pathway, and becomes N-glycosylated before entering the chloroplast.
This is a preview of subscription content, access via your institution
Access options




Similar content being viewed by others
References
Leister, D. Chloroplast research in the genomic age. Trends Genet. 19, 47–56 (2003).
Abdallah, F., Salamini, F. & Leister, D. A prediction of the size and evolutionary origin of the proteome of chloroplasts of Arabidopsis. Trends Plant Sci. 5, 141–142 (2000).
Keegstra, K. & Cline, K. Protein import and routing systems of chloroplasts. Plant Cell 11, 557–570 (1999).
Soll, J. Protein import into chloroplasts. Curr. Opin. Plant Biol. 5, 529–535 (2002).
Jarvis, P. & Soll, J. Toc, Tic, and chloroplast protein import. Biochim. Biophys. Acta 1541, 64–79 (2001).
Kleffmann, T. et al. The Arabidopsis thaliana chloroplast proteome reveals pathway abundance and novel protein functions. Curr. Biol. 14, 354–362 (2004).
Friso, G. et al. In-depth analysis of the thylakoid membrane proteome of Arabidopsis thaliana chloroplasts: new proteins, new functions, and a plastid proteome database. Plant Cell 16, 478–499 (2004).
Asatsuma, S. et al. Involvement of α-amylase I-1 in starch degradation in rice chloroplasts. Plant Cell Physiol. 46, 858–869 (2005).
Hewet-Emmett, D. & Tashian, R. E. Functional diversity, conservation, and convergence in the evolution of the α-, β-, and γ-carbonic anhydrase gene families. Mol. Phyl. Evol. 5, 50–77 (1996).
Emanuelsson, O., Nielsen H., Brunak, S. & von Heijne, G. Predicting subcellular localization of proteins based on their N-terminal amino acid sequence. J. Mol. Biol. 300, 1005–1016 (2000).
Nielsen, H., Engelbrecht, J., Brunak, S. & von Heijne, G. Identification of prokaryotic and eukaryotic signal peptides and prediction of their cleavage sites. Prot. Eng. 10, 1–6 (1997).
Monné, M., Nilsson, I., Elofsson, A. & von Heijne, G. Turns in transmembrane helices: determination of the minimal length of a “helical hairpin” and derivation of a fine-grained turn propensity scale. J. Mol. Biol. 293, 807–814 (1999).
Lerouge, P. et al. N-glycosylation biosynthesis in plants: recent developments and future trends. Plant Mol. Biol. 38, 31–48 (1998).
Faye, L., Gomord, V., Fitchette-Laine, A. C. & Chrispeels, M. J. Affinity purification of antibodies specific for Asn-linked glycans containing α1–3 fucose or β1–2 xylose. Anal. Biochem. 209, 104–108 (1993).
Rayon, C. et al. Characterization of N-glycans from Arabidopsis thaliana. Application to a fucose-deficient mutant. Plant Physiol. 119, 725–734 (1999).
Bonin, C., Potter, I., Vanzin, G. F. & Reiter, W. D. The MUR1 gene of Arabidopsis thaliana encodes an isoform of GDP-D-mannose-4,6-dehydratase, catalyzing the first step in the de novo synthesis of GDP-L-fucose. Proc. Natl Acad. Sci. USA 94, 2085–2090 (1997).
Ritzenthaler, C. et al. Reevaluation of the effects of brefeldin A on plant cells using tobacco bright yellow 2 cells expressing Golgi-targeted green fluorescent protein and COPI antisera. Plant Cell 14, 237–261 (2002).
Lee, M. H. et al. ADP-ribosylation factor 1 of Arabidopsis plays a critical role in intracellular trafficking and maintenance of endoplasmic reticulum morphology in Arabidopsis. Plant Physiol. 129, 1507–1520 (2002).
Zheng, H., Kunst, L., Hawes, C. & Moore, I. A GFP-based assay reveals a role for RHD3 in transport between the endoplasmic reticulum and Golgi apparatus. Plant J. 37, 398–414 (2004).
Sulli, C. & Schwartzbach, S. D. The polyprotein precursor to the Euglena light-harvesting chlorophyll a/b-binding protein is transported to the Golgi apparatus prior to chloroplast import and polyprotein processing. J. Biol. Chem. 270, 13084–13090 (1995).
Waller, R. F., Reed, M. B., Cowman, A. F. & McFaden, I. Protein trafficking to the plastid of Plasmodium falciparum is via the secretory pathway. EMBO J. 19, 1794–1802 (2000).
Delwiche, C. Tracing the thread of plastid diversity through the tapestry of life. Am. Nat. 154, S164–S177 (1999).
Foth, B. J. et al. Dissecting apicoplast targeting in the malaria parasite Plasmodium falciparum. Science 299, 705–708 (2003).
Kilian, O. & Kroth, P. G. Identification and characterization of a new conserved motif within the presequence of proteins targeted into complex diatom plastids. Plant J. 41, 175–183 (2005).
Moreau, P. et al. Lipid trafficking in plant cells. Prog. Lipid Res. 37, 371–391 (1998).
Xu, C., Fan, J., Riekhof, W., Froehlich, J. E. & Benning, C. A permease-like protein involved in ER to thylakoid lipid transfer in Arabidopsis. EMBO J. 22, 2370–2379 (2003).
Chen, M. H., Huang, L. F., Li, H.-M., Chen, Y. R. & Yu, S. M. Signal peptide-dependent targeting of a rice α-amylase and cargo proteins to plastids and extracellular compartments of plant cells. Plant Physiol. 135, 1367–1377 (2004).
Levitan, A. et al. Dual targeting of the protein disulfide isomerase RB60 to the chloroplast and the endoplasmic reticulum. Proc. Natl Acad. Sci. USA 102, 6225–6230 (2005).
Kunst, L. in Methods in Molecular Biology, Arabidopsis Protocols Vol 82 (eds Martinez-Zapater, J. & Salinas, J.) 43–53 (Humana Press Inc., Totowa, New Jersey, USA, 1998).
Kozak, M. Regulation of translation in eukaryotic systems. Annu. Rev. Cell. Biol. 8, 197–225 (1992).
Hermansson, M., Monné, M. & von Heijne, G. Formation of helical hairpins during membrane protein integration into the endoplasmic reticulum membrane. Role of the N- and C-terminal flanking regions. J. Mol. Biol. 313, 1171–1179 (2001).
Chiu, W. L. et al. Engineered GFP as a vital reporter in plants. Curr. Biol. 6, 325–330 (1996).
Gasparian, M. et al. Identification and characterization of an 18-kilodalton, VAMP-like protein in suspension-cultured carrot cells. Plant Physiol. 122, 25–33 (2000).
Acknowledgements
We thank A. Kraut for technical assistance in the transient expression of GFP fusions in plant cells; L. Faye for the gift of antibodies against xylose and fucose residues; C. Robinson for helpful discussion; and J. Brangeon and R. Boyer for technical help in the immunocytochemistry experiments. The authors are grateful to B. Martin for critical reading of the manuscript. This work was supported by grants from the Swedish National Research Council, FORMAS, Wallenberg and Kempe Foundations, and the Swedish Foundation for Strategic Research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Methods and Results plus Supplementary figures S1, S2 and S3 (PDF 2717 kb)
Rights and permissions
About this article
Cite this article
Villarejo, A., Burén, S., Larsson, S. et al. Evidence for a protein transported through the secretory pathway en route to the higher plant chloroplast. Nat Cell Biol 7, 1224–1231 (2005). https://doi.org/10.1038/ncb1330
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1330
This article is cited by
-
Genome-wide identification, evolution, and expression analysis of carbonic anhydrases genes in soybean (Glycine max)
Functional & Integrative Genomics (2023)
-
Chaperone-like protein DAY plays critical roles in photomorphogenesis
Nature Communications (2021)
-
Advances in understanding the physiological role and locations of carbonic anhydrases in C3 plant cells
Protoplasma (2021)
-
The role of carbonic anhydrase α-CA4 in the adaptive reactions of photosynthetic apparatus: the study with α-CA4 knockout plants
Protoplasma (2020)
-
Study of subcellular localization of Glycine max γ-tocopherol methyl transferase isoforms in N. benthamiana
3 Biotech (2020)