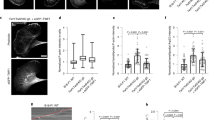
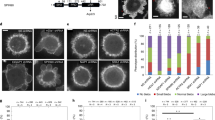
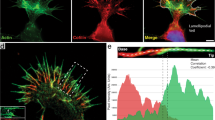
Abstract
Formins have important roles in the nucleation of actin and the formation of linear actin filaments, but their role in filopodium formation has remained elusive. Dictyostelium discoideum Diaphanous-related formin dDia2 is enriched at the tips of filopodia and interacts with profilin II and Rac1. An FH1FH2 fragment of dDia2 nucleated actin polymerization and removed capping protein from capped filament ends. Genetic studies showed that dDia2 is important for cell migration as well as the formation, elongation and maintenance of filopodia. Here we provide evidence that dDia2 specifically controls filopodial dynamics by regulating actin turnover at the barbed ends of actin filaments.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Evangelista, M., Zigmond, S. & Boone, C. Formins: signaling effectors for assembly and polarization of actin filaments. J. Cell Sci. 116, 2603–2611 (2003).
Wallar, B. J. & Alberts, A. S. The formins: active scaffolds that remodel the cytoskeleton. Trends Cell Biol. 13, 435–446 (2003).
Zigmond, S. H. Formin-induced nucleation of actin filaments. Curr. Opin. Cell Biol. 16, 99–105 (2004).
Petersen, J., Nielsen, O., Egel, R. & Hagan, I. M. FH3, a domain found in formins, targets the fission yeast formin Fus1 to the projection tip during conjugation. J. Cell Biol. 141, 1217–1228 (1998).
Alberts, A. S. Identification of a carboxyl-terminal diaphanous-related formin homology protein autoregulatory domain. J. Biol. Chem. 276, 2824–2830 (2001).
Shimada, A. et al. The core FH2 domain of diaphanous-related formins is an elongated actin binding protein that inhibits polymerization. Mol. Cell 13, 511–522 (2004).
Xu, Y. et al. Crystal structures of a formin homology-2 domain reveal a tethered dimer architecture. Cell 116, 711–723 (2004).
Evangelista, et al. Bni1p, a yeast formin linking cdc42p and the actin cytoskeleton during polarized morphogenesis. Science 276, 118–122 (1997).
Watanabe, N. et al. p140mDia, a mammalian homolog of Drosophila diaphanous, is a target protein for Rho small GTPase and is a ligand for profilin. EMBO J. 16, 3044–3056 (1997).
Chan, D. C., Bedford, M. T. & Leder, P. Formin binding proteins bear WWP/WW domains that bind proline-rich peptides and functionally resemble SH3 domains. EMBO J. 15, 1045–1054 (1996).
Sagot, I., Rodal, A. A., Moseley, J., Goode, B. L. & Pellman, D. An actin nucleation mechanism mediated by Bni1 and profilin. Nature Cell Biol. 4, 626–631 (2002).
Kovar, D. R., Kuhn, J. R., Tichy, A. L. & Pollard, T. D. The fission yeast cytokinesis formin Cdc12p is a barbed end actin filament capping protein gated by profilin. J. Cell Biol. 161, 875–887 (2003).
Evangelista, M., Pruyne, D., Amberg, D. C., Boone, C. & Bretscher, A. Formins direct Arp2/3-independent actin filament assembly to polarize cell growth in yeast. Nature Cell Biol. 4, 32–41 (2002).
Peng, J., Wallar, B. J., Flanders, A., Swiatek, P. J. & Alberts, A. S. Disruption of the Diaphanous-related formin Drf1 gene encoding mDia1 reveals a role for Drf3 as an effector for Cdc42. Curr. Biol. 13, 534–545 (2003).
Pellegrin, S. & Mellor, H. The Rho family GTPase Rif induces filopodia through mDia2. Curr. Biol. 15, 129–133 (2005).
Dai, J. & Sheetz, M. P. Membrane tether formation from blebbing cells. Biophys. J. 77, 3363–3370 (1999).
Romero, S. et al. Formin is a processive motor that requires profilin to accelerate actin assembly and associated ATP hydrolysis. Cell 119, 419–429 (2004).
Kovar, D. R. & Pollard, T. D. Insertional assembly of actin filament barbed ends in association with formins produces piconewton forces. Proc. Natl Acad. Sci. USA 101, 14725–14730 (2004).
Eichinger, L. et al. The genome of the social amoeba Dictyostelium discoideum. Nature 435, 43–57 (2005).
Kitayama, C. & Uyeda, T. Q. ForC, a novel type of formin family protein lacking an FH1 domain, is involved in multicellular development in Dictyostelium discoideum. J. Cell Sci. 116, 711–723 (2003).
Haugwitz, M., Noegel, A. A., Karakesisoglou, J. & Schleicher, M. Dictyostelium amoebae that lack G-actin-sequestering profilins show defects in F-actin content, cytokinesis, and development. Cell 79, 303–314 (1994).
Rivero, F., Dislich, H., Glöckner, G. & Noegel, A. A. The Dictyostelium discoideum family of Rho-related proteins. Nucleic Acids Res. 29, 1068–1079 (2001).
Faix, J. et al. Recruitment of cortexillin into the cleavage furrow is controlled by Rac1 and IQGAP-related proteins. EMBO J. 20, 3705–3715 (2001).
Dumontier, M., Höcht, P., Mintert, U. & Faix, J. Rac1 GTPases control filopodia formation, cell motility, endocytosis, cytokinesis and development in Dictyostelium. J. Cell Sci. 113, 2253–2265 (2000).
Small, J. V., Stradal, T., Vignal, E. & Rottner, K. The lamellipodium: where motility begins. Trends Cell Biol. 12, 112–120 (2002).
Han, Y. H. et al. Requirement of a vasodilator-stimulated phosphoprotein family member for cell adhesion, the formation of filopodia, and chemotaxis in Dictyostelium. J. Biol. Chem. 277, 49877–49887 (2002).
Svitkina, T. M. et al. Mechanism of filopodia initiation by reorganization of a dendritic network. J. Cell Biol. 160, 409–421 (2003).
Lebrand, C. et al. Critical role of Ena/VASP proteins for filopodia formation in neurons and in function downstream of Netrin-1. Neuron 42, 37–49 (2004).
Hug, C. et al. Capping protein levels influence actin assembly and cell motility in Dictyostelium. Cell 81, 591–600 (1995).
Faix, J. et al. The IQGAP-related protein DGAP1 interacts with Rac and is involved in the modulation of the F-actin cytoskeleton and control of cell motility. J. Cell Sci. 111, 3059–3071 (1998).
Sutoh, K. A transformation vector for Dictyostelium discoideum with a new selectable marker bsr. Plasmid 2, 150–154 (1993).
Hartmann, H., Noegel, A. A., Eckerskorn, C., Rapp, S. & Schleicher, M. Ca2+-independent F-actin capping proteins. J. Biol. Chem. 264, 12639–12647 (1989).
Faix, J. & Dittrich, W. DGAP1, a homologue of rasGTPase activating proteins that controls growth, cytokinesis, and development in Dictyostelium discoideum. FEBS Lett. 394, 251–257 (1996).
Weber, I. et al. Cytokinesis mediated through the recruitment of cortexillins into the cleavage furrow. EMBO J. 18, 586–594 (1999).
Eichinger, L. & Schleicher, M. Characterization of actin- and lipid-binding domains in severin, a Ca2+-dependent F-actin fragmenting protein. Biochemistry 31, 4779–4787 (1992).
Acknowledgements
We thank J. Segall for discussions and for reading the manuscript, W. Witke for CapG cDNA and D. Rieger and M. Borath for excellent technical assistance. This work was supported by a grant to M.S. from the Deutsche Forschungsgemeinschaft and a grant to J.F. from the Friedrich-Baur-Stiftung.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Schirenbeck, A., Bretschneider, T., Arasada, R. et al. The Diaphanous-related formin dDia2 is required for the formation and maintenance of filopodia. Nat Cell Biol 7, 619–625 (2005). https://doi.org/10.1038/ncb1266
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1266
This article is cited by
-
Deciphering the involvement of the Hippo pathway co-regulators, YAP/TAZ in invadopodia formation and matrix degradation
Cell Death & Disease (2023)
-
Myosin IIA and formin dependent mechanosensitivity of filopodia adhesion
Nature Communications (2019)
-
FMNL formins boost lamellipodial force generation
Nature Communications (2017)
-
Inhibition of breast cancer invasion by TIS21/BTG2/Pc3-Akt1-Sp1-Nox4 pathway targeting actin nucleators, mDia genes
Oncogene (2016)
-
Internetwork competition for monomers governs actin cytoskeleton organization
Nature Reviews Molecular Cell Biology (2016)