Abstract
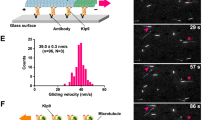
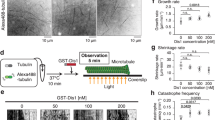
The motion of a chromosome during mitosis is mediated by a bundle of microtubules, termed a kinetochore fibre (K-fibre), which connects the kinetochore of the chromosome to a spindle pole. Once formed, mature K-fibres maintain a steady state length because the continuous addition of microtubule subunits onto microtubule plus ends at the kinetochore is balanced by their removal at their minus ends within the pole. This condition is known as 'microtubule poleward flux'1. Chromosome motion and changes in position are then driven by changes in K-fibre length, which in turn are controlled by changes in the rates at which microtubule subunits are added at the kinetochore and/or removed from the pole2. A key to understanding the role of flux in mitosis is to identify the molecular factors that drive it. Here we use Drosophila melanogaster S2 cells expressing α-tubulin tagged with green fluorescent protein, RNA interference, laser microsurgery and photobleaching to show that the kinetochore protein MAST/Orbit — the single CLASP orthologue in Drosophila — is an essential component for microtubule subunit incorporation into fluxing K-fibres.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Mitchison, T. J. Polewards microtubule flux in the mitotic spindle: evidence from photoactivation of fluorescence. J. Cell Biol. 109, 637–652 (1989).
Rogers, G. C. et al. Two mitotic kinesins cooperate to drive sister chromatid separation during anaphase. Nature 427, 364–370 (2004).
Carvalho, P., Tirnauer, J. S. & Pellman, D. Surfing on microtubule ends. Trends Cell Biol. 13, 229–237 (2003).
Lemos, C. L. et al. Mast, a conserved microtubule-associated protein required for bipolar mitotic spindle organization. EMBO J. 19, 3668–3682 (2000).
Maiato, H. et al. Human CLASP1 is an outer kinetochore component that regulates spindle microtubule dyanmics. Cell 113, 891–904 (2003).
Maiato, H. et al. MAST/Orbit has a role in microtubule-kinetochore attachment and is essential for chromosome alignment and maintenance of spindle bipolarity. J. Cell Biol. 157, 749–760 (2002).
Goshima, G. & Vale, R. D. The roles of microtubule-based motor proteins in mitosis: comprehensive RNAi analysis in the Drosophila S2 cell line. J. Cell Biol. 162, 1003–1016 (2003).
Maiato, H., Rieder, C. L. & Khodjakov, A. Kinetochore-driven formation of kinetochore fibers contributes to spindle assembly during mitosis in animals. J. Cell Biol. DOI: 10.1083/jcb.200407090.
Ganem, N. J. & Compton, D. A. The KinI kinesin Kif2a is required for bipolar spindle assembly through a functional relationship with MCAK. J. Cell Biol. 166, 473–478 (2004).
Lawrence, C. J. et al. A standardized kinesin nomenclature. J. Cell Biol. 167, 19–22 (2004).
Yin, H., You, L., Pasqualone, D., Kopski, K. M. & Huffaker, T. C. Stu1p is physically associated with β-tubulin and is required for structural integrity of the mitotic spindle. Mol. Biol. Cell 13, 1881–1892 (2002).
Sharp, D. J. MAST sails through mitosis. Curr. Biol. 12, R585–R587 (2002).
Desai, A. et al. KNL-1 directs assembly of the microtubule-binding interface of the kinetochore in C. elegans. Genes Dev. 17, 2421–2435 (2003).
Hoffman, D. B., Pearson, C. G., Yen, T. J., Howell, B. J. & Salmon, E. D. Microtubule dependent changes in the assembly of microtubule motor proteins and mitotic spindle checkpoint proteins at kinetochores. Mol. Biol. Cell 12, 1995–2009 (2001).
Tirnauer, J. S., Canman, J. C., Salmon, E. D. & Mitchison, T. J. EB1 targets to kinetochores with attached, polymerizing microtubules. Mol. Biol. Cell 13, 4308–4316 (2002).
Rogers, S. L., Rogers, G. C., Sharp, D. J. & Vale, R. D. Drosophila EB1 is important for proper assembly, dynamics, and positioning of the mitotic spindle. J. Cell Biol. 158, 873–884 (2002).
Waters, J. C., Mitchison, T. J., Rieder, C. L. & Salmon, E. D. The kinetochore microtubule minus-end disassembly associated with poleward flux produces a force that can do work. Mol. Biol. Cell 7, 1547–1558 (1996).
Czaban, B. B., Forer, A. & Bajer, A. S. Ultraviolet microbeam irradiation of chromosomal spindle fibres in Haemanthus katherinae endosperm. I. Behaviour of the irradiated region. J. Cell Sci. 105, 571–578 (1993).
Forer, A., Spurck, T. & Pickett-Heaps, J. Ultaviolet microbeam irradiations of spindle fibers in crane-fly spermatocytes and newt epithelial cells: resolution of previously conflicting observations. Protoplasma 197, 230–240 (1997).
Gordon, G. W. The Control of Mitotic Motility as Influenced by Ultraviolet Microbeam Irradiation of Kinetochore Fibers. Thesis, Univ. Pennsylvania (1980).
Wadsworth, P. & Khodjakov, A. E pluribus unum: towards a universal mechanism for spindle assembly. Trends Cell Biol. 14, 413–419 (2004).
Chen, W. & Zhang, D. Kinetochore fibre dynamics outside the contex of the spindle during anaphase. Nature Cell Biol. 6, 227–231 (2004).
LaFountain, J. R. Jr., Cohan, C. S., Siegel, A. J. & LaFountain, D. J. Direct visualization of microtubule flux during metaphase and anaphase in crane-fly spermatocytes. Mol. Biol. Cell 15, 5724–5732 (2004).
Mitchison, T. J. & Salmon, E. D. Poleward kinetochore fiber movement occurs during both metaphase and anaphase-A in newt lung cell mitosis. J. Cell Biol. 119, 569–582 (1992).
Brust-Mascher, I. & Scholey, J. M. Microtubule flux and sliding in mitotic spindles of Drosophila embryos. Mol. Biol. Cell 13, 3967–3975 (2002).
Maddox, P., Desai, A., Oegema, K., Mitchison, T. J. & Salmon, E. D. Poleward microtubule flux is a major component of spindle dynamics and anaphase A in mitotic Drosophila embryos. Curr. Biol. 12, 1670–1674 (2002).
Skibbens, R. V., Skeen, V. P. & Salmon, E. D. Directional instability of kinetochore motility during chromosome congression and segregation in mitotic newt lung cells: a push-pull mechanism. J. Cell Biol. 122, 859–875 (1993).
Khodjakov, A. & Rieder, C. L. Kinetochores moving away from their associated pole do not exert a significant pushing force on the chromosome. J. Cell Biol. 135, 315–327 (1996).
Maiato, H., Sunkel, C. E. & Earnshaw, W. C. Dissecting mitosis by RNAi in Drosophila tissue culture cells. Biol. Proced. 5, 153–161 (2003).
Acknowledgements
We thank G. Goshima, S. Rogers and R. Vale (University of California, San Francisco, CA) for the S2T cells and EB1 antibodies, and C. Sunkel (University of Porto, Portugal) for communicating results before publication. This work was supported by National Institutes of Health grants GMS 40198 (to C.L.R.), GMS 59363 (to A.K.) and a postdoctoral research fellowship from Fundação para a Ciência e a Tecnologia of Portugal (SFRH/BPD/1159/2002 to H.M.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Maiato, H., Khodjakov, A. & Rieder, C. Drosophila CLASP is required for the incorporation of microtubule subunits into fluxing kinetochore fibres. Nat Cell Biol 7, 42–47 (2005). https://doi.org/10.1038/ncb1207
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1207
This article is cited by
-
Mechanisms underlying spindle assembly and robustness
Nature Reviews Molecular Cell Biology (2023)
-
CLASP balances two competing cell division plane cues during leaf development
Nature Plants (2022)
-
Theoretical model of mitotic spindle microtubule growth for FRAP curve interpretation
BMC Systems Biology (2017)
-
MAPs: cellular navigators for microtubule array orientations in Arabidopsis
Plant Cell Reports (2014)
-
CLASPs prevent irreversible multipolarity by ensuring spindle-pole resistance to traction forces during chromosome alignment
Nature Cell Biology (2012)