Abstract
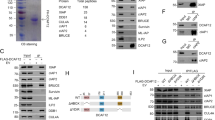
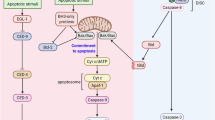
Some members of the inhibitor of apoptosis (IAP) family suppress apoptosis by neutralizing caspases. The current model suggests that all caspase-regulatory IAPs function as direct enzyme inhibitors, blocking effector caspases by binding to their catalytically active pockets. Here we show that IAPs are functionally non-equivalent and regulate effector caspases through distinct mechanisms. Whereas XIAP binds directly to the active-site pockets of effector caspases, we find that regulation of effector caspases by Drosophila IAP1 (DIAP1) requires an evolutionarily conserved IAP-binding motif (IBM) at the neo-amino terminus of the large caspase subunit. Remarkably, unlike XIAP, DIAP1-sequestered effector caspases remain catalytically active, suggesting that DIAP1 does not function as a bona fide enzyme inhibitor. Moreover, we demonstrate that the mammalian IAP c-IAP1 interacts with caspase-7 in an exclusively IBM-dependent, but active site pocket-independent, manner that is mechanistically similar to DIAP1. The importance of IBM-mediated regulation of effector-caspases in vivo is substantiated by the enhanced apoptotic potency of IBM-mutant versions of drICE, DCP-1 and caspase-7.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Salvesen, G. S. & Duckett, C. S. Apoptosis: IAP proteins: blocking the road to death's door. Nature Rev. Mol. Cell Biol. 3, 401–410 (2002).
Goyal, L., McCall, K., Agapite, J., Hartwieg, E. & Steller, H. Induction of apoptosis by Drosophila reaper, hid and grim through inhibition of IAP function. EMBO J. 19, 589–597 (2000).
Lisi, S., Mazzon, I. & White, K. Diverse domains of THREAD/DIAP1 are required to inhibit apoptosis induced by REAPER and HID in Drosophila. Genetics 154, 669–678 (2000).
Rodriguez, A. et al. Dark is a Drosophila homologue of Apaf-1/CED-4 and functions in an evolutionarily conserved death pathway. Nature Cell Biol. 1, 272–279 (1999).
Wang, S. L., Hawkins, C. J., Yoo, S. J., Muller, H. A. & Hay, B. A. The Drosophila caspase inhibitor DIAP1 is essential for cell survival and is negatively regulated by HID. Cell 98, 453–463 (1999).
Harlin, H., Reffey, S. B., Duckett, C. S., Lindsten, T. & Thompson, C. B. Characterization of XIAP-deficient mice. Mol. Cell. Biol. 21, 3604–3608 (2001).
Shi, Y. Mechanisms of caspase activation and inhibition during apoptosis. Mol. Cell 9, 459–470 (2002).
Shiozaki, E. N. et al. Mechanism of XIAP-mediated inhibition of caspase-9. Mol. Cell 11, 519–527 (2003).
Suzuki, Y., Nakabayashi, Y., Nakata, K., Reed, J. C. & Takahashi, R. X-linked inhibitor of apoptosis protein (XIAP) inhibits caspase-3 and -7 in distinct modes. J. Biol. Chem. 276, 27058–27063 (2001).
Silke, J. et al. Direct inhibition of caspase 3 is dispensable for the anti-apoptotic activity of XIAP. EMBO J. 20, 3114–3123 (2001).
Chai, J. et al. Structural basis of caspase-7 inhibition by XIAP. Cell 104, 769–780 (2001).
Huang, Y. et al. Structural basis of caspase inhibition by XIAP: differential roles of the linker versus the BIR domain. Cell 104, 781–790 (2001).
Riedl, S. J. et al. Structural basis for the inhibition of caspase-3 by XIAP. Cell 104, 791–800 (2001).
Zachariou, A. et al. IAP-antagonists exhibit non-redundant modes of action through differential DIAP1 binding. EMBO J. 22, 6642–6652 (2003).
Yan, N., Wu, J. W., Chai, J., Li, W. & Shi, Y. Molecular mechanisms of DrICE inhibition by DIAP1 and removal of inhibition by Reaper, Hid and Grim. Nature Struct. Mol. Biol. 11, 420–428 (2004).
Yokokura, T. et al. Dissection of DIAP1 functional domains via a mutant replacement strategy. J. Biol. Chem. DOI: 10.1074/jbc.M409691200 (2004).
Ditzel, M. et al. Degradation of DIAP1 by the N-end rule pathway is essential for regulating apoptosis. Nature Cell Biol. 5, 467–473 (2003).
Bachmair, A., Finley, D. & Varshavsky, A. In vivo half-life of a protein is a function of its amino-terminal residue. Science 234, 179–186 (1986).
Varshavsky, A. Ubiquitin fusion technique and its descendants. Methods Enzymol. 327, 578–593 (2000).
Hawkins, C. J., Wang, S. L. & Hay, B. A. A cloning method to identify caspases and their regulators in yeast: Identification of Drosophila IAP1 as an inhibitor of the Drosophila caspase DCP-1. Proc. Natl Acad. Sci. USA 96, 2885–2890 (1999).
Hawkins, C. J. et al. The Drosophila caspase DRONC cleaves following glutamate or aspartate and is regulated by DIAP1, HID, and GRIM. J. Biol. Chem. 275, 27084–27093. (2000).
Meier, P., Silke, J., Leevers, S. J. & Evan, G. I. The Drosophila caspase DRONC is regulated by DIAP1. EMBO J. 19, 598–611 (2000).
Wilson, R. et al. The DIAP1 RING finger mediates ubiquitination of Dronc and is indispensable for regulating apoptosis. Nature Cell Biol. 4, 445–450 (2002).
Schlegel, J. et al. CPP32/apopain is a key interleukin 1 β converting enzyme-like protease involved in Fas-mediated apoptosis. J. Biol. Chem. 271, 1841–1844 (1996).
Tenev, T., Zachariou, A., Wilson, R., Paul, A. & Meier, P. Jafrac2 is an IAP antagonist that promotes cell death by liberating Dronc from DIAP1. EMBO J. 21, 5118–5129 (2002).
Wu, J. W., Cocina, A. E., Chai, J., Hay, B. A. & Shi, Y. Structural analysis of a functional DIAP1 fragment bound to grim and hid peptides. Mol. Cell 8, 95–104 (2001).
Wu, G. et al. Structural basis of IAP recognition by Smac/DIABLO. Nature 408, 1008–1012 (2000).
Srinivasula, S. M. et al. A conserved XIAP-interaction motif in caspase-9 and Smac/DIABLO regulates caspase activity and apoptosis. Nature 410, 112–116 (2001).
Rigaut, G. et al. A generic protein purification method for protein complex characterization and proteome exploration. Nature Biotechnol. 17, 1030–1032 (1999).
Tenev, T. et al. Perinuclear localization of the protein-tyrosine phosphatase SHP-1 and inhibition of epidermal growth factor-stimulated STAT1/3 activation in A431 cells. Eur. J. Cell Biol 79, 261–271 (2000).
Acknowledgements
We thank A. Varshavsky and F. Levy for the ubiquitin fusion construct and B. Seraphin for the TAP construct. We thank F. Leulier, S. Schneider, A. Ashworth and J. Silke for critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Fig S1, Fig S2, Fig S3, Fig S4 (PDF 650 kb)
Rights and permissions
About this article
Cite this article
Tenev, T., Zachariou, A., Wilson, R. et al. IAPs are functionally non-equivalent and regulate effector caspases through distinct mechanisms. Nat Cell Biol 7, 70–77 (2005). https://doi.org/10.1038/ncb1204
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1204
This article is cited by
-
Drice restrains Diap2-mediated inflammatory signalling and intestinal inflammation
Cell Death & Differentiation (2022)
-
The Hh pathway promotes cell apoptosis through Ci-Rdx-Diap1 axis
Cell Death Discovery (2021)
-
Monoglyceride lipase mediates tumor-suppressive effects by promoting degradation of X-linked inhibitor of apoptosis protein
Cell Death & Differentiation (2020)
-
The Relevance of SNPs at 3′UTR Region of CASP7 and miR-371b-5p Associated Diseases: A Computational Analysis
Cell Biochemistry and Biophysics (2020)
-
Boolean network modeling of β-cell apoptosis and insulin resistance in type 2 diabetes mellitus
BMC Systems Biology (2019)