Abstract
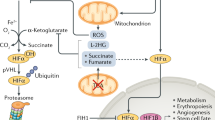
Hypoxia and acidosis occur in a wide variety of physiological and pathological settings that include muscle stress, tumour development and ischaemic disorders. A central element in the adaptive response to cellular hypoxia is HIF (hypoxia-inducible factor), a transcription factor that activates an array of genes implicated in oxygen homeostasis, tumour vascularization and ischaemic preconditioning1. HIF is activated by hypoxia, but undergoes degradation by the VHL (von Hippel-Lindau) tumour suppressor protein in the presence of oxygen2,3. Here, we demonstrate that hypoxia induction or normoxic acidosis can neutralize the function of VHL by triggering its nucleolar sequestration, a regulatory mechanism of protein function that is observed rarely4,5,6,7. VHL is confined to nucleoli until neutral pH conditions are re-instated. Nucleolar sequestration of VHL enables HIF to evade destruction in the presence of oxygen and activate its target genes. Our findings suggest that an increase in hydrogen ions elicits a transient and reversible loss of VHL function by promoting its nucleolar sequestration.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Semenza, G.L. HIF-1 and human disease: one highly involved factor. Genes Dev. 14, 1983–1991 (2000).
Maxwell, P.H. et al. The tumour suppressor protein VHL targets hypoxia-inducible factors for oxygen-dependent proteolysis. Nature 399, 271–275 (1999).
Jaakkola, P. et al. Targeting of HIF-alpha to the von Hippel-Lindau ubiquitylation complex by O2-regulated prolyl hydroxylation. Science 292, 468–472 (2001).
Shou, W. et al. Exit from mitosis is triggered by Tem1-dependent release of the protein phosphatase Cdc14 from nucleolar RENT complex. Cell 97, 233–244 (1999).
Visintin, R., Hwang, E.S. & Amon, A. Cfi1 prevents premature exit from mitosis by anchoring Cdc14 phosphatase in the nucleolus. Nature 398, 818–823 (1999).
Weber, J.D., Taylor, L.J., Roussel, M.F., Sherr, C.J. & Bar-Sagi, D. Nucleolar Arf sequesters Mdm2 and activates p53. Nature Cell Biol. 1, 20–26 (1999).
Tsai, R.Y. & McKay, R.D. A nucleolar mechanism controlling cell proliferation in stem cells and cancer cells. Genes Dev. 16, 2991–3003 (2002).
Wagner, P.D. Skeletal muscle angiogenesis. A possible role for hypoxia. Adv. Exp. Med. Biol. 502, 21–38 (2001).
Kaelin, W.G., Jr. Molecular basis of the VHL hereditary cancer syndrome. Nature Rev. Cancer 2, 673–682 (2002).
Harris, A.L. Hypoxia — a key regulatory factor in tumour growth. Nature Rev. Cancer 2, 38–47 (2002).
Currin, R.T., Gores, G.J., Thurman, R.G. & Lemasters, J.J. Protection by acidotic pH against anoxic cell killing in perfused rat liver: evidence for a pH paradox. Faseb J. 5, 207–210 (1991).
Kaku, D.A., Giffard, R.G. & Choi, D.W. Neuroprotective effects of glutamate antagonists and extracellular acidity. Science 260, 1516–1518 (1993).
Giffard, R.G., Monyer, H., Christine, C.W. & Choi, D.W. Acidosis reduces NMDA receptor activation, glutamate neurotoxicity, and oxygen-glucose deprivation neuronal injury in cortical cultures. Brain Res. 506, 339–342 (1990).
Morimoto, Y., Kemmotsu, O. & Alojado, E.S. Extracellular acidosis delays cell death against glucose-oxygen deprivation in neuroblastoma x glioma hybrid cells. Crit. Care Med. 25, 841–847 (1997).
Nielsen, O.B., de Paoli, F. & Overgaard, K. Protective effects of lactic acid on force production in rat skeletal muscle. J. Physiol. 536, 161–166 (2001).
Lee, S. et al. Transcription-dependent nuclear-cytoplasmic trafficking is required for the function of the von Hippel-Lindau tumor suppressor protein. Mol. Cell Biol. 19, 1486–1497 (1999).
Groulx, I. & Lee, S. Oxygen-dependent ubiquitination and degradation of hypoxia-inducible factor requires nuclear-cytoplasmic trafficking of the von Hippel-Lindau tumor suppressor protein. Mol. Cell Biol. 22, 5319–5336 (2002).
Kibel, A., Iliopoulos, O., DeCaprio, J.A. & Kaelin, W.G., Jr. Binding of the von Hippel-Lindau tumor suppressor protein to Elongin B and C. Science 269, 1444–1446 (1995).
Corless, C.L., Kibel, A.S., Iliopoulos, O. & Kaelin, W.G., Jr. Immunostaining of the von Hippel-Lindau gene product in normal and neoplastic human tissues. Hum. Pathol. 28, 459–464 (1997).
Warburg, O. The metabolism of tumors (R.R. Smith, New York, NY, USA, 1931).
Warburg, O. On the origin of cancer cells. Science 123, 309–314 (1956).
Sutherland, R.M. Cell and environment interactions in tumor microregions: the multicell spheroid model. Science 240, 177–184 (1988).
Iliopoulos, O., Kibel, A., Gray, S. & Kaelin, W.G., Jr. Tumour suppression by the human von Hippel-Lindau gene product. Nature Med. 1, 822–826 (1995).
Pause, A. et al. The von Hippel-Lindau tumor-suppressor gene product forms a stable complex with human CUL-2, a member of the Cdc53 family of proteins. Proc. Natl Acad. Sci. USA 94, 2156–2161 (1997).
Cockman, M.E. et al. Hypoxia inducible factor-alpha binding and ubiquitylation by the von Hippel-Lindau tumor suppressor protein. J. Biol. Chem. 275, 25733–25741 (2000).
Bonicalzi, M.E., Groulx, I., de Paulsen, N. & Lee, S. Role of exon 2-encoded beta -domain of the von Hippel-Lindau tumor suppressor protein. J. Biol. Chem. 276, 1407–1416 (2001).
Gunaratnam, L. et al. Hypoxia inducible factor activates the transforming growth factor-alpha/epidermal growth factor receptor growth stimulatory pathway in VHL(−/−) renal cell carcinoma cells. J. Biol. Chem. 278, 44966–44974 (2003).
Chavez, J.C. & LaManna, J.C. Activation of hypoxia-inducible factor-1 in the rat cerebral cortex after transient global ischemia: potential role of insulin-like growth factor-1. J. Neurosci. 22, 8922–8931 (2002).
Sun, Y. et al. VEGF-induced neuroprotection, neurogenesis, and angiogenesis after focal cerebral ischemia. J. Clin. Invest. 111, 1843–1851 (2003).
Bachant, J.B. & Elledge, S.J. Mitotic treasures in the nucleolus. Nature 398, 757–758 (1999).
Acknowledgements
We thank C. Lavigne for technical expertise. This work was supported by a grant from the Canadian Institutes of Health Research (CIHR). K.M. is supported by the Natural Science and Engineering Research Council of Canada (NSERC). S.L. is a Harold E. Johns investigator of the National Cancer Institute of Canada (NCIC).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Mekhail, K., Gunaratnam, L., Bonicalzi, ME. et al. HIF activation by pH-dependent nucleolar sequestration of VHL. Nat Cell Biol 6, 642–647 (2004). https://doi.org/10.1038/ncb1144
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1144
This article is cited by
-
Role of pH-sensing receptors in colitis
Pflügers Archiv - European Journal of Physiology (2024)
-
How protons pave the way to aggressive cancers
Nature Reviews Cancer (2023)
-
Nucleolar translocation of human DNA topoisomerase II by ATP depletion and its disruption by the RNA polymerase I inhibitor BMH-21
Scientific Reports (2021)
-
A translational program that suppresses metabolism to shield the genome
Nature Communications (2020)
-
Mechanosensation of cyclical force by PIEZO1 is essential for innate immunity
Nature (2019)