Abstract
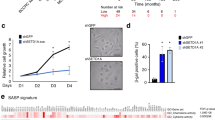
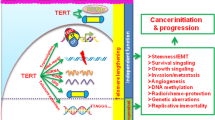
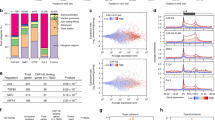
In contrast to cancer cells and embryonic stem cells, the lifespan of primary human cells is finite. After a defined number of population doublings, cells enter in an irreversible growth-arrested state termed replicative senescence1. Mutations of genes involved in immortalization can contribute to cancer2,3. In a genetic screen for cDNAs bypassing replicative senescence of normal human prostate epithelial cells (HPrEC), we identified CBX7, a gene that encodes a Polycomb protein, as shown by sequence homology, its interaction with Ring1 and its localization to nuclear Polycomb bodies. CBX7 extends the lifespan of a wide range of normal human cells and immortalizes mouse fibroblasts by downregulating expression of the Ink4a/Arf locus. CBX7 does not inter-function or colocalize with Bmi1, and both can exert their actions independently of each other as shown by reverse genetics. CBX7 expression is downregulated during replicative senescence and its ablation by short-hairpin RNA (shRNA) treatment inhibited growth of normal cells though induction of the Ink4a/Arf locus. Taken together, these data show that CBX7 controls cellular lifespan through regulation of both the p16Ink4a/Rb and the Arf/p53 pathways.
This is a preview of subscription content, access via your institution
Access options




Similar content being viewed by others
Change history
08 August 2022
A Correction to this paper has been published: https://doi.org/10.1038/s41556-022-00956-2
References
Hayflick, L. The limited in vitro lifespan of human diploid cell strains. Exp. Cell Res. 37, 614–621 (1965).
Campisi, J. Cellular senescence as a tumor-suppressor mechanism. Trends Cell Biol. 11, S27–S31 (2001).
Hanahan, D. & Weinberg, R.A. The hallmarks of cancer. Cell 100, 57–70 (2000).
Kiyono, T. et al. Both Rb/p16INK4a inactivation and telomerase activity are required to immortalize human epithelial cells. Nature 396, 84–88 (1998).
Jacobs, J.J. et al. Senescence bypass screen identifies TBX2, which represses Cdkn2a (p19(ARF)) and is amplified in a subset of human breast cancers. Nature Genet. 26, 291–299 (2000).
Jacobs, J.J.L., Kieboom, K., Marino, S., DePinho, R.A. & van Lohuizen, M. The oncogene and Polycomb-group gene bmi-1 regulates cell proliferation and senescence through the ink4a locus. Nature 397, 164–168 (1999).
Hannon, G.J. et al. MaRX: an approach to genetics in mammalian cells. Science 283, 1129–1130 (1999).
Maestro, R. et al. Twist is a potential oncogene that inhibits apoptosis. Genes Dev. 13, 2207–2217 (1999).
Kamijo, T.F. et al. Tumor suppression at the mouse INK4a locus mediated by the alternative reading frame product p19ARF. Cell 91, 649–659 (1997).
Paro, R. & Hogness, D.S. The Polycomb protein shares a homologous domain with a heterochromatin-associated protein of Drosophila. Proc. Natl Acad. Sci. USA 88, 263–267 (1991).
Min, J., Zhang, Y. & Xu, R.-M. Structural basis for specific binding of Polycomb chromodomain to histone H3 methylated at Lys 27. Genes Dev. 17, 1823–1828 (2003).
Jones, D.O., Cowell, I.G. & Singh, P.B. Mammalian chromodomain proteins: their role in genome organisation and expression. Bioessays 22, 124–137 (2000).
Satijn, D.P. & Otte, A.P. Polycomb group protein complexes: do different complexes regulate distinct target genes? Biochim. Biophys. Acta 1447, 1–16 (1999).
Satijn, D.P. et al. RING1 is associated with the polycomb group protein complex and acts as a transcriptional repressor. Mol Cell Biol. 17, 4105–4113 (1997).
Saurin, A.J. et al. The human polycomb group complex associates with pericentromeric heterochromatin to form a novel nuclear domain. J. Cell Biol. 142, 887–898 (1998).
Jacobs, J.J. & van Lohuizen, M. Polycomb repression: from cellular memory to cellular proliferation and cancer. Biochim. Biophys. Acta 1602, 151–161 (2002).
Serrano, M., Lin, A.W., McCurrach, M.E., Beach, D. & Lowe, S.W. Oncogenic ras provokes premature cell senescence associated with accumulation of p53 and p16INK4a. Cell 88, 593–602 (1997).
Quelle, D.E., Zindy, F., Ashmun, R.A. & Sherr, C.J. Alternative reading frames of the INK4a tumor suppressor gene encode two unrelated proteins capable of inducing cell cycle arrest. Cell 83, 993–1000 (1995).
Zhang, Y., Xiong, Y. & Yarbrough, W.G. ARF promotes MDM2 degradation and stabilizes p53: ARF-INK4a locus deletion impairs both the Rb and p53 tumor suppression pathways. Cell 92, 725–734 (1998).
Serrano, M. et al. Role of the INK4a locus in tumor suppression and cell mortality. Cell 85, 27–37 (1996).
Sharpless, N.E. & DePinho, R.A. The INK4A/ARF locus and its two gene products. Curr. Opin. Genet. Dev. 9, 22–30 (1999).
Alkema, M.J., Jacobs, H., van Lohuizen, M. & Berns, A. Perturbation of B and T cell development and predisposition to lymphomagenesis in E-Bmi1 transgenic mice require the Bmi1 RING finger. Oncogene 15, 899–910 (1997).
Sewalt, R.G. et al. Characterization of interactions between the mammalian polycomb-group proteins Enx1/EZH2 and EED suggests the existence of different mammalian polycomb-group protein complexes. Mol. Cell Biol. 18, 3586–3595 (1998).
Varambally, S. et al. The polycomb group protein EZH2 is involved in progression of prostate cancer. Nature 419, 624–629 (2002).
Gunster, M.J. et al. Differential expression of human Polycomb group proteins in various tissues and cell types. J. Cell Biochem. 81, 129–143 (2001).
Itahana, K. et al. Control of the replicative life span of human fibroblasts by p16 and the polycomb protein Bmi-1. Mol. Cell Biol. 23, 389–401 (2003).
Dimri, G.P. et al. The Bmi-1 oncogene induces telomerase activity and immortalizes human mammary epithelial cells. Cancer Res. 62, 4736–4745 (2002).
Satijn, D.P. et al. Interference with the expression of a novel human polycomb protein, hPc2, results in cellular transformation and apoptosis. Mol. Cell Biol. 17, 6076–6086 (1997).
Carnero, A., Hudson, J.D., Hannon, G.J. & Beach, D.H. Loss-of-function genetics in mammalian cells: the p53 tumor suppressor model. Nucleic Acids Res. 28, 2234–2241 (2000).
Brummelkamp, T.R., Bernards, R. & Agami, R. A system for stable expression of short interfering RNAs in mammalian cells. Science 296, 550–553 (2002).
Acknowledgements
We thank W. Bordogna, M. van Lohuizen, F. Fuks, D. Wotton and P. Freemont for materials, M. Serrano, A. Carnero, M. Collado and all members of D. Beach's laboratory for thoughtful comments, and P. Gómez-Puertas for help with bioinformatic analysis. This work was supported by grants from Cancer Research UK, The Wellcome Trust, and the Hugh and Catherine Stevens Fund. J.G. is recipient of a Long Term Fellowship from the Human Frontier Science Program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information, Fig. S1
Supplementary Information, Fig. S2 (PDF 801 kb)
Supplementary Information, Fig. S3
Supplementary Information, Fig. S4
Rights and permissions
About this article
Cite this article
Gil, J., Bernard, D., Martínez, D. et al. Polycomb CBX7 has a unifying role in cellular lifespan. Nat Cell Biol 6, 67–72 (2004). https://doi.org/10.1038/ncb1077
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1077
This article is cited by
-
Adipose-derived exosomal miR-421 targets CBX7 and promotes metastatic potential in ovarian cancer cells
Journal of Ovarian Research (2023)
-
CBX7 Rejuvenates Late Passage Dental Pulp Stem Cells by Maintaining Stemness and Pro-angiogenic Ability
Tissue Engineering and Regenerative Medicine (2023)
-
Context-specific Polycomb mechanisms in development
Nature Reviews Genetics (2022)
-
Cellular senescence in musculoskeletal homeostasis, diseases, and regeneration
Bone Research (2021)
-
A multidimensional systems biology analysis of cellular senescence in aging and disease
Genome Biology (2020)