Abstract
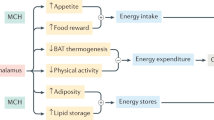
Gene-knockout studies of melanin-concentrating hormone (MCH) and its effect on feeding and energy balance have firmly established MCH as an orexigenic (appetite-stimulating) peptide hormone. Here we identify MCH as the ligand for the orphan receptor SLC-1. The rat SLC-1 is activated by nanomolar concentrations of MCH and is coupled to the G protein Gαi/o. The pattern of SLC-1 messenger RNA expression coincides with the distribution of MCH-containing nerve terminals and is consistent with the known central effects of MCH. Our identification of an MCH receptor could have implications for the development of new anti-obesity therapies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Rance, T. A. & Baker, B. I. Further observations on the distribution and properties of teleost melanin concentrating hormone. Gen. Comp. Endocrinol. 50, 423–431 (1983).
Kawauchi, H. et al. Characterization of melanin concentrating hormone in chum salmon pituitaries. Nature 305, 321–323 (1983).
Bittencourt, J. C. et al. The melanin-concentrating hormone system of the rat brain: an immuno- and hybridization histochemical characterization. J.Comp. Neurol. 319, 218–245 (1992).
Qu, D. et al. A role for melanin-concentrating hormone in the central regulation of feeding behaviour. Nature 380, 243–247 (1996).
Shimada, M., Tritos, N. A., Lowell, B. B., Flier, J. S. & Maratos-Flier, E. Mice lacking melanin-concentrating hormone are hypophagic and lean. Nature 396, 670–674 (1998).
Lakaye, B., Minet, A., Zorzi, W. & Grisar, T. Cloning of the rat brain cDNA encoding for the SLC-1 G-protein-coupled receptor reveals the presence of an intron in the gene. Biochim. Biophys. Acta 1401, 216–220 (1998).
Drozdz, R. in Proc. 23rd Eur. Peptide Symp. (ed. Braga, H. L. S.) 785 (Escom, Leiden, 1995).
Baker, B. I. Melanin concentrating hormone updated. Trends Endocrinol. Metab. 5, 120–126 (1994).
Singer, W. D., Brown, H. A. & Sternweis, P. C. Regulation of eukaryotic phosphatidylinositol-specific phospholipase C and phospholipase D. Annu. Rev. Biochem. 66, 475–509 (1997).
Wess, J. Molecular basis of receptor/G-protein-coupling selectivity. Pharmacol. Therap. 80, 231–264 (1998).
Zhang, J. et al. Molecular mechanisms of G-protein coupled receptors signaling: role of G-protein coupled receptor kinases and arrestins in receptor desensitization and resensitization. Receptors Channels 5, 193–199 (1997).
Barak, L. S., Ferguson, S. S., Zhang, J. & Caron, M. G. A β-arrestin/green fluorescent protein biosensor for detecting G protein-coupled receptor activation. J. Biol. Chem. 272, 27497–27500 (1997).
Kakinoki, Y., Somers, J. & Brautigan, D. L. Multisite phosphorylation and the nuclear localization of phosphatase inhibitor 2-green fluorescent protein fusion protein during S phase of the cell growth cycle. J. Biol. Chem. 272, 32308–32314 (1997).
Drmota, T., Gould, G. W. & Milligan, G. Real time visualization of agonist-mediated redistribution and internalization of a green fluorescent protein-tagged form of the thyrotropin-releasing hormone receptor. J. Biol. Chem. 273, 24000–24008 (1998).
Awaji, T. et al. Real-time optical monitoring of ligand-mediated internalization of α1b-adrenoceptor with green fluorescent protein. Mol. Endocrinol. 12, 1099–1111 (1998).
Drmota, T., Novotny, J., Gould, G. W., Svoboda, P. & Milligan, G. Visualization of distinct patterns of subcellular redistribution of the thyrotropin-releasing hormone receptor-1 and G(q)α/G(11)α induced by agonist stimulation. Biochem. J. 340, 529–538 (1999).
Fulwiller, C. E. & Saper, C. B. Subnuclear organization of the efferent connections of the parabrachial nucleus in the rat. Brain Res. 319, 229–259 (1984).
Fulwiller, C. E. & Saper, C. B. Cholecystokinin-immunoreactive innervation of the ventromedial hypothalamus in the rat: possible substrate for autonomic regulation of feeding. Neurosci. Lett. 53, 289–296 (1985).
Clapham D. E. & Neer, E. J. G protein βγ subunits. Annu. Rev. Pharmacol. Toxicol. 37, 167–203 (1997).
Camps, M. et al. Stimulation of phospholipase C by guanine-nucleotide-binding protein βγ subunits. Eur. J. Biochem. 206, 821–831 (1992).
Ghahremani, M. H., Cheng, P., Lembo, P. M. & Albert, P. R. Distinct roles for Gαi2, Gαi3, and Gβγ in modulation of forskolin- or Gs-mediated cAMP accumulation and calcium mobilization by dopamine D2S receptors. J. Biol. Chem. 274, 9238–9245 (1999 ).
Murthy, K. S., Coy, D. H. & Makhlouf, G. M. Somatostatin receptor-mediated signaling in smooth muscle. Activation of phospholipase C-β3 by G-βγ and inhibition of adenylyl cyclase by Gαi-1 and Gα-o. J. Biol. Chem. 271, 23458–23463 (1996).
Gunn, T. M. et al. The mouse mahogany locus encodes a transmembrane form of human attractin. Nature 398, 152–156 (1999).
Chambers, J. et al. Melanin-concentrating hormone is the cognate ligand for the orphan G-protein-coupled receptor SLC-1. Nature 400, 261–265 (1999).
Saito, Y. et al. Molecular characterization of the melanin-concentrating-hormone receptor. Nature 400, 265–269 (1999).
O’Donnell, D., Ahmad, S., Wahlestedt, C. & Walker, P. Expression of the novel galanin receptor subtype GALR2 in the adult rat CNS: distinct distribution from GALR1. J. Comp. Neurol. 409, 469–481 (1999).
Paxinos, G. & Watson, C. Rat Brain Atlas 4th edn (Academic, San Diego, 1998).
Acknowledgements
We thank G. Milligan for providing HEK 293 cells stably transfected with GFP-tagged TRH receptor-1; A. Beaudet for advice on receptor-internalization studies; and G. Keil for critical analysis of the manuscript.
Correspondence and requests for materials should be addressed to S.A.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lembo, P., Grazzini, E., Cao, J. et al. The receptor for the orexigenic peptide melanin-concentrating hormone is a G-protein-coupled receptor. Nat Cell Biol 1, 267–271 (1999). https://doi.org/10.1038/12978
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/12978
This article is cited by
-
Hypothalamic melanin-concentrating hormone neurons integrate food-motivated appetitive and consummatory processes in rats
Nature Communications (2023)
-
Multifaceted actions of melanin-concentrating hormone on mammalian energy homeostasis
Nature Reviews Endocrinology (2021)
-
Breeding history and candidate genes responsible for black skin of Xichuan black-bone chicken
BMC Genomics (2020)
-
Hypothalamus-hippocampus circuitry regulates impulsivity via melanin-concentrating hormone
Nature Communications (2019)
-
SNAPshots of the MCHR1: a Comparison Between the PET-Tracers [18F]FE@SNAP and [11C]SNAP-7941
Molecular Imaging and Biology (2019)