Abstract
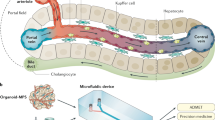
Tissue function depends on hierarchical structures extending from single cells (∼10 μm) to functional subunits (100 μm–1 mm) that coordinate organ functions. Conventional cell culture disperses tissues into single cells while neglecting higher-order processes. The application of semiconductor-driven microtechnology in the biomedical arena now allows fabrication of microscale tissue subunits that may be functionally improved1 and have the advantages of miniaturization2. Here we present a miniaturized, multiwell culture system for human liver cells with optimized microscale architecture that maintains phenotypic functions for several weeks. The need for such models is underscored by the high rate of pre-launch and post-market attrition of pharmaceuticals due to liver toxicity3. We demonstrate utility through assessment of gene expression profiles, phase I/II metabolism, canalicular transport, secretion of liver-specific products and susceptibility to hepatotoxins. The combination of microtechnology and tissue engineering may enable development of integrated tissue models in the so-called 'human on a chip'4.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Voldman, J., Gray, M.L. & Schmidt, M.A. Microfabrication in biology and medicine. Annu. Rev. Biomed. Eng. 1, 401–425 (1999).
Khetani, S.R. & Bhatia, S.N. Engineering tissues for in vitro applications. Curr. Opin. Biotechnol. 17, 524–531 (2006).
Kaplowitz, N. Idiosyncratic drug hepatotoxicity. Nat. Rev. Drug Discov. 4, 489–499 (2005).
Viravaidya, K. & Shuler, M.L. Incorporation of 3T3–L1 cells to mimic bioaccumulation in a microscale cell culture analog device for toxicity studies. Biotechnol. Prog. 20, 590–597 (2004).
Chen, C.S., Mrksich, M., Huang, S., Whitesides, G.M. & Ingber, D.E. Geometric control of cell life and death. Science 276, 1425–1428 (1997).
Bhatia, S.N., Balis, U.J., Yarmush, M.L. & Toner, M. Effect of cell-cell interactions in preservation of cellular phenotype: cocultivation of hepatocytes and nonparenchymal cells. FASEB J. 13, 1883–1900 (1999).
Pritchard, J.F. et al. Making better drugs: Decision gates in non-clinical drug development. Nat. Rev. Drug Discov. 2, 542–553 (2003).
Gebhardt, R. et al. New hepatocyte in vitro systems for drug metabolism: metabolic capacity and recommendations for application in basic research and drug development, standard operation procedures. Drug Metab. Rev. 35, 145–213 (2003).
Guillouzo, A. Liver cell models in in vitro toxicology. Environ. Health Perspect. 106 Suppl 2, 511–532 (1998).
LeCluyse, E.L. Human hepatocyte culture systems for the in vitro evaluation of cytochrome P450 expression and regulation. Eur. J. Pharm. Sci. 13, 343–368 (2001).
Sivaraman, A. et al. A microscale in vitro physiological model of the liver: predictive screens for drug metabolism and enzyme induction. Curr. Drug Metab. 6, 569–591 (2005).
Hewitt, N.J. et al. Primary hepatocytes: current understanding of the regulation of metabolic enzymes and transporter proteins, and pharmaceutical practice for the use of hepatocytes in metabolism, enzyme induction, transporter, clearance, and hepatotoxicity studies. Drug Metab. Rev. 39, 159–234 (2007).
Donato, M.T., Jimenez, N., Castell, J.V. & Gomez-Lechon, M.J. Fluorescence-based assays for screening nine cytochrome P450 (P450) activities in intact cells expressing individual human P450 enzymes. Drug Metab. Dispos. 32, 699–706 (2004).
Wilkening, S., Stahl, F. & Bader, A. Comparison of primary human hepatocytes and hepatoma cell line Hepg2 with regard to their biotransformation properties. Drug Metab. Dispos. 31, 1035–1042 (2003).
Whitesides, G.M., Ostuni, E., Takayama, S., Jiang, X. & Ingber, D.E. Soft lithography in biology and biochemistry. Annu. Rev. Biomed. Eng. 3, 335–373 (2001).
Khetani, S.R., Szulgit, G., Del Rio, J.A., Barlow, C. & Bhatia, S.N. Exploring interactions between rat hepatocytes and nonparenchymal cells using gene expression profiling. Hepatology 40, 545–554 (2004).
Madan, A. et al. Effects of prototypical microsomal enzyme inducers on cytochrome P450 expression in cultured human hepatocytes. Drug Metab. Dispos. 31, 421–431 (2003).
Waring, J.F. et al. Isolated human hepatocytes in culture display markedly different gene expression patterns depending on attachment status. Toxicol. In Vitro 17, 693–701 (2003).
Richert, L. et al. Gene expression in human hepatocytes in suspension after isolation is similar to the liver of origin, is not affected by hepatocyte cold storage and cryopreservation, but is strongly changed after hepatocyte plating. Drug Metab. Dispos. 34, 870–879 (2006).
Rodriguez-Antona, C., Donato, M.T., Pareja, E., Gomez-Lechon, M.J. & Castell, J.V. Cytochrome P-450 mRNA expression in human liver and its relationship with enzyme activity. Arch. Biochem. Biophys. 393, 308–315 (2001).
Isley, W.L. Hepatotoxicity of thiazolidinediones. Expert Opin. Drug Saf. 2, 581–586 (2003).
Kamali, F. The effect of probenecid on paracetamol metabolism and pharmacokinetics. Eur. J. Clin. Pharmacol. 45, 551–553 (1993).
Pirotte, J.H. Apparent potentiation of hepatotoxicity from small doses of acetaminophen by phenobarbital. Ann. Intern. Med. 101, 403 (1984).
Corlu, A. et al. The coculture: a system for studying the regulation of liver differentiation/proliferation activity and its control. Cell Biol. Toxicol. 13, 235–242 (1997).
Eschbach, E. et al. Microstructured scaffolds for liver tissue cultures of high cell density: morphological and biochemical characterization of tissue aggregates. J. Cell. Biochem. 95, 243–255 (2005).
Allen, J.W., Hassanein, T. & Bhatia, S.N. Advances in bioartificial liver devices. Hepatology 34, 447–455 (2001).
Naughton, B.A., Sibanda, B., Weintraub, J.P., San Roman, J. & Kamali, V. A stereotypic, transplantable liver tissue-culture system. Appl. Biochem. Biotechnol. 54, 65–91 (1995).
Kaihara, S. et al. Survival and function of rat hepatocytes cocultured with nonparenchymal cells or sinusoidal endothelial cells on biodegradable polymers under flow conditions. J. Pediatr. Surg. 35, 1287–1290 (2000).
Kane, B.J., Zinner, M.J., Yarmush, M.L. & Toner, M. Liver-specific functional studies in a microfluidic array of primary Mammalian hepatocytes. Anal. Chem. 78, 4291–4298 (2006).
Zeilinger, K. et al. Three-dimensional co-culture of primary human liver cells in bioreactors for in vitro drug studies: effects of the initial cell quality on the long-term maintenance of hepatocyte-specific functions. Altern. Lab. Anim. 30, 525–538 (2002).
Acknowledgements
We are grateful to Emanuele Ostuni and Surface Logix, Inc. for design and fabrication of the PDMS stencils, Howard Green for providing 3T3-J2 fibroblasts, Jennifer Koh for assistance with pilot studies, David Eddington for assistance with microfabrication, Taylor Sittler for helpful discussions regarding compound selection, Elise Liu for assistance with biochemical assays and Sandra March for assistance with RNA isolation. Funding was generously provided by a National Science Foundation (NSF) graduate fellowship (S.R.K.), NSF CAREER, National Institutes of Health National Institute of Diabetes and Digestive and Kidney Diseases, Deshpande Center at MIT, the David and Lucile Packard Foundation, the Massachusetts Technology Transfer Center, and the Center for Environmental Health Sciences at MIT.
Author information
Authors and Affiliations
Contributions
S.R.K. designed and performed the experiments, analyzed the data and wrote the manuscript. S.N.B. designed the experiments, analyzed the data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
S.R.K. and S.N.B. have stock in Hepregen.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5, Supplementary Table 1, Supplementary Methods (PDF 379 kb)
Rights and permissions
About this article
Cite this article
Khetani, S., Bhatia, S. Microscale culture of human liver cells for drug development. Nat Biotechnol 26, 120–126 (2008). https://doi.org/10.1038/nbt1361
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt1361
This article is cited by
-
Advances and challenges in organ-on-chip technology: toward mimicking human physiology and disease in vitro
Medical & Biological Engineering & Computing (2024)
-
Human disease models in drug development
Nature Reviews Bioengineering (2023)
-
Micropatterned primary hepatocyte co-culture (HEPATOPAC) for fatty liver disease modeling and drug screening
Scientific Reports (2023)
-
A human iPSC-derived hepatocyte screen identifies compounds that inhibit production of Apolipoprotein B
Communications Biology (2023)
-
Numerical Simulation of Mixing in Active Micromixers Using SPH
Transport in Porous Media (2023)