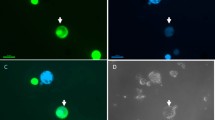
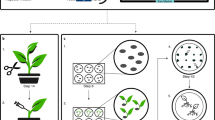
Abstract
Progress in the genetic manipulation of cereals using protoplast technology has been limited by a lack of reproducible plant regeneration from protoplasts of these important species. Here we report that plant regeneration can be achieved efficiently and reproducibly from protoplasts of rice isolated from cell suspension cultures. Regeneration from 10—20% of protoplast–derived colonies of two varieties was obtained rapidly after direct transfer to a hormone–free medium. Green plants were produced at a high frequency through somatic embryogenesis. The protoplast origin of the regenerated plants is unequivocally demonstrated by the inability of contaminating intact cells to divide.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Lörz, H., Baker, B., and Schell, J. 1985. Gene transfer to cereal cells mediated by protoplast transformation. Mol. Gen. Genet. 199:178–182.
Potrykus, I., Saul, M.W., Petruska, J., Paszkowski, J., and Shillito, R.D. 1985. Direct gene transfer to cells of a graminaceous monocot. Mol. Gen. Genet. 199:183–188.
Fromm, M.E., Taylor, L.P., and Walbot, V. 1986. Stable transformation of maize after gene transfer by electroporation. Nature 319:791–793.
Vasil, V. and Vasil, I.K. 1980. Isolation and culture of cereal protoplasts. II. Embryogenesis and plantlet formation from protoplasts of Pennisetum americanum. Theor. Appl. Genet. 56:97–99.
Lu, C.Y., Vasil, V., and Vasil, I.K. 1981. Isolation and culture of protoplasts of Panicum maximum Jacq. (Guinea grass): Somatic embryogenesis and plantlet formation. Z. Pflanzenphysiol. 104:311–318.
Vasil, V., Wang, D.Y., and Vasil, I.K. 1983. Plant regeneration from protoplasts of Napier Grass (Pennisetum purpureum Schum.). Z. Pflanzenphysiol. 111:233–239.
Heyser, J.W. 1984. Callus and shoot regeneration from protoplasts of Proso Millet (Panicum miliaceum L.). Z. Pflanzenphysiol. 113:293–299.
Vasil, I.K. 1983. Isolation and culture of protoplasts of grasses. Int. Review of Cytology, Suppl. 16:79–88.
Harms, C.T. 1982. Maize and cereal protoplasts—facts and perspectives, p. 373–384. In: Maize for Biological Research. W. F. Sheridan (ed.). Plant Molecular Biology Association, Charlottesville, Virginia.
Fujimura, T., Sakurai, M., Akagi, H., Negishi, T., and Hirose, A. 1985. Regeneration of rice plants from protoplasts. Plant Tissue Culture Letters 2:74–75.
Coulibaly, M.Y. and Demarly, Y. 1986. Regeneration of plantlets from protoplasts of rice, Oryza sativa L. Z. Pflanzenzuchtg. 96:79–81.
Yamada, Y., Yang, Z.Q., and Tang, D.T. 1986. Plant regeneration from protoplast-derived callus of rice (Oryza sativa L.). Plant Cell Reports 5:85–88.
Thompson, J.A., Abdullah, R., and Cocking, E.C. 1986. Protoplast culture of rice (Oryza sativa L.) using media solidified with agarose. Plant Science in press.
Ahuja, P.S., Pental, D., and Cocking, E.C. 1982. Plant regeneration from leaf base callus and cell suspensions of Triticum aestivum. Z. Pflanzenzuchtg. 89:139–144.
Ho, W.J. and Vasil, I.K. 1983. Somatic embryogenesis in sugarcane (Saccharum officinarum L.) I. The morphology of callus formation and the ontogeny of somatic embryos. Protoplasma 118:169–180.
Gray, D.J., Conger, B.V., and Hanning, G.E. 1984. Somatic embryogenesis in suspension and suspension-derived callus cultures of Dactylis glomerata. Protoplasma 122:196–202.
Wernicke, W., Brettell, R., Wakizuka, T., and Potrykus, I. 1981. Adventitious embryoid and root formation from rice leaves. Z. Pflanzenphysiol. 103:361–365.
Thompson, J.A. 1983. Protoplast isolation from morphogenic cell cultures of rice (Oryza sativa L.). Experientia Suppl. 45:2–3.
Murashige, T. and Skoog, F. 1962. A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol. Plant. 15:473–497.
Linsmaier, E.M. and Skoog, F. 1965. Organic growth factor requirements of tobacco tissue cultures. Physiol. Plant. 8:100–127.
Müller, A.J. and Grafe, R. 1978. Isolation and characterization of cell lines of Nicotiana tabacum lacking nitrate reductase. Mol. Gen. Genet. 161:67–76.
Frearson, E.M., Power, J.B., and Cocking, E.C. 1973. The isolation, culture and regeneration of Petunia leaf protoplasts. Dev. Biol. 33:130–137.
Kao, K.N. 1977. Chromosomal behaviour in somatic hybrids of soybean—Nicotiana glauca. Molec. Gen. Genet. 150:225–230.
Widholm, J.M. 1972. The use of fluorescein diacetate and phenosafranine for determining viability of cultured plant cells. Stain Technology 47:189–194.
Chu, C.C., Wang, C.C., Sun, C.S., Hsu, C., Yin, K.C., Chu, C.Y., and Bi, F.Y. 1975. Establishment of an efficient medium for anther culture of rice through comparative experiments on the nitrogen sources. Sci. Sinica 18:659–668.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Abdullah, R., Cocking, E. & Thompson, J. Efficient Plant Regeneration from Rice Protoplasts Through Somatic Embryogenesis. Nat Biotechnol 4, 1087–1090 (1986). https://doi.org/10.1038/nbt1286-1087
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt1286-1087
This article is cited by
-
In vitro rooting of leguminous plants: Difficulties, alternatives, and strategies for improvement
Horticulture, Environment, and Biotechnology (2016)
-
Isolation and Characterization of a Novel SOMATIC EMBRYOGENESIS RECEPTOR KINASE Gene Expressed in the Fern Adiantum capillus-veneris During Shoot Regeneration In Vitro
Plant Molecular Biology Reporter (2015)
-
Small brown planthopper resistance loci in wild rice (Oryza officinalis)
Molecular Genetics and Genomics (2014)
-
Mitochondrial VDAC and hexokinase together modulate plant programmed cell death
Protoplasma (2013)
-
Maturation of Anthurium andraeanum cv. Eidibel somatic embryos from nodal segments
In Vitro Cellular & Developmental Biology - Plant (2013)