Abstract
Dendrimers are branched, synthetic polymers with layered architectures that show promise in several biomedical applications. By regulating dendrimer synthesis, it is possible to precisely manipulate both their molecular weight and chemical composition, thereby allowing predictable tuning of their biocompatibility and pharmacokinetics. Advances in our understanding of the role of molecular weight and architecture on the in vivo behavior of dendrimers, together with recent progress in the design of biodegradable chemistries, has enabled the application of these branched polymers as anti-viral drugs, tissue repair scaffolds, targeted carriers of chemotherapeutics and optical oxygen sensors. Before such products can reach the market, however, the field must not only address the cost of manufacture and quality control of pharmaceutical-grade materials, but also assess the long-term human and environmental health consequences of dendrimer exposure in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Buhleier, E., Wehner, W. & Vögtle, F. “Cascade”- and “nonskid-chain-like” syntheses of molecular cavity topologies. Synthesis (Mass.) 155–158 (1978).
Denkewalter, R.G., Kolc, J. & Lukasavage, W.J. Macromolecular highly branched homogeneous compound based on lysine units. US Patent 4,289,872, (1981).
Tomalia, D.A. et al. A new class of polymers-starburst-dendritic macromolecules. Polym. J. 17, 117–132 (1985).
Newkome, G.R., Yao, Z., Baker, G.R. & Gupta, V.K. Micelles. Part 1. Cascade molecules: a new approach to micelles. A [27]-arborol. J. Org. Chem. 50, 2003–2004 (1985).
Fréchet, J.M.J. & Tomalia, D.A. (eds.) Dendrimers and Other Dendritic Polymers. (John Wiley & Sons, Chichester, New York, USA, 2001).
Tomalia, D.A., Naylor, A.M. & Goddard, W.A. Starburst dendrimers: molecular-level control of size, shape, surface chemistry, topology, and flexibility from atoms to macroscopic matter. Angew. Chem. Int. Edn. Engl. 29, 138–175 (1990).
de Brabander-van den Berg, E.M.M. & Meijer, E.W. Poly(propylene imine) dendrimers: large-scale synthesis by hetereogeneously catalyzed hydrogenations. Angew. Chem. Int. Edn Engl. 32, 1308–1311 (1993).
Hawker, C.J. & Fréchet, J.M.J. Preparation of polymers with controlled molecular architecture. A new convergent approach to dendritic macromolecules. J. Am. Chem. Soc. 112, 7638–7647 (1990).
Sadler, K. & Tam, J.P. Peptide dendrimers: applications and synthesis. J. Biotechnol. 90, 195–229 (2002).
Ihre, H., Hult, A. & Söderlind, E. Synthesis, characterization, and 1H NMR self-diffusion studies of dendritic aliphatic polyesters based on 2,2-bis(hydroxymethyl)propionic acid and 1,1,1-tris(hydroxyphenyl)ethane. J. Am. Chem. Soc. 118, 6388–6395 (1996).
Grinstaff, M.W. Biodendrimers: new polymeric biomaterials for tissue engineering. Chemistry 8, 2838–2846 (2002).
Turnbull, W.B. & Stoddart, J.F. Design and synthesis of glycodendrimers. J. Biotechnol. 90, 231–255 (2002).
Nilsen, T.W., Grayzel, J. & Prensky, W. Dendritic nucleic acid structures. J. Theor. Biol. 187, 273–284 (1997).
Li, Y. et al. Controlled assembly of dendrimer-like DNA. Nat. Mater. 3, 38–42 (2004).
Liu, M., Kono, K. & Fréchet, J.M.J. Water-soluble dendritic unimolecular micelles: their potential as drug delivery agents. J. Control. Release 65, 121–131 (2000).
Stevelmans, S. et al. Synthesis, characterization, and guest-host properties of inverted unimolecular dendritic micelles. J. Am. Chem. Soc. 118, 7398–7399 (1996).
Mammen, M., Choi, S.K. & Whitesides, G.M. Polyvalent interactions in biological systems: implications for design and use of multivalent ligands and inhibitors. Angew. Chem. Int. Edn Engl. 37, 2754–2794 (1998).
Lundquist, J.J. & Toone, E.J. The cluster glycoside effect. Chem. Rev. 102, 555–578 (2002).
André, S., Liu, B., Gabius, H.J. & Roy, R. First demonstration of differential inhibition of lectin binding by synthetic tri- and tetravalent glycoclusters from cross-coupling of rigidified 2-propynyl lactoside. Org. Biomol. Chem. 1, 3909–3916 (2003).
Jiang, Y.H. et al. SPL7013 gel as a topical microbicide for prevention of vaginal transmission of SHIV89.6P in macaques. AIDS Res. Hum. Retroviruses 21, 207–213 (2005).
Hecht, S. & Fréchet, J.M.J. Dendritic encapsulation of function: applying nature's site isolation principle from biomimetics to materials science. Angew. Chem. Int. Edn. Engl. 40, 74–91 (2001).
Kojima, C., Kono, K., Maruyama, K. & Takagishi, T. Synthesis of polyamidoamine dendrimers having poly(ethylene glycol) grafts and their ability to encapsulate anticancer drugs. Bioconjug. Chem. 11, 910–917 (2000).
Morgan, M.T. et al. Dendritic molecular capsules for hydrophobic compounds. J. Am. Chem. Soc. 125, 15485–15489 (2003).
Rozhkov, V., Wilson, D. & Vinogradov, S. Phosphorescent Pd porphyrin-dendrimers: tuning core accessibility by varying the hydrophobicity of the dendritic matrix. Macromolecules 35, 1991–1993 (2002).
Cloninger, M.J. Biological applications of dendrimers. Curr. Opin. Chem. Biol. 6, 742–748 (2002).
Stiriba, S.E., Frey, H. & Haag, R. Dendritic polymers in biomedical applications: from potential to clinical use in diagnostics and therapy. Angew. Chem. Int. Edn. Engl. 41, 1329–1334 (2002).
Boas, U. & Heegaard, P.M.H. Dendrimers in drug research. Chem. Soc. Rev. 33, 43–63 (2004).
Gillies, E.R. & Fréchet, J.M.J. Dendrimers and dendritic polymers in drug delivery. Drug Discov. Today 10, 35–43 (2005).
Allen, T.M. & Cullis, P.R. Drug delivery systems: entering the mainstream. Science 303, 1818–1822 (2004).
Malik, N., Evagorou, E.G. & Duncan, R. Dendrimer-platinate: a novel approach to cancer chemotherapy. Anticancer Drugs 10, 767–776 (1999).
Matsumura, Y. & Maeda, H. A new concept for macromolecular therapeutics in cancer chemotherapy: mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res. 46, 6387–6392 (1986).
Duncan, R. Polymer conjugates for tumour targeting and intracytoplasmic delivery. The EPR effect as a common gateway? Pharm. Sci. Technol. Today 2, 441–449 (1999).
Kukowska-Latallo, J.F. et al. Nanoparticle targeting of anticancer drug improves therapeutic response in animal model of human epithelial cancer. Cancer Res. 65, 5317–5324 (2005).
Wooley, K.L., Hawker, C.J. & Fréchet, J.M.J. Unsymmetrical three-dimensional macromolecules: preparation and characterization of strongly dipolar dendritic macromolecules. J. Am. Chem. Soc. 115, 11496–11505 (1993).
Gillies, E.R. & Fréchet, J.M.J. Designing macromolecules for therapeutic applications: polyester dendrimer-poly(ethylene oxide) “bow-tie” hybrids with tunable molecular weight and architecture. J. Am. Chem. Soc. 124, 14137–14146 (2002).
Steffensen, M.B. & Simanek, E.E. Synthesis and manipulation of orthogonally protected dendrimers: building blocks for library synthesis. Angew. Chem. Int. Edn. Engl. 43, 5178–5180 (2004).
Li, Y., Cu, Y.T.H. & Luo, D. Multiplexed detection of pathogen DNA with DNA-based fluorescence nanobarcodes. Nat. Biotechnol. 23, 885–889 (2005).
Haensler, J. & Szoka, F.C. Polyamidoamine cascade polymers mediate efficient transfection of cells in culture. Bioconjug. Chem. 4, 372–379 (1993).
Tang, M.X., Redemann, C.T. & Szoka, F.C. In vitro gene delivery by degraded polyamidoamine dendrimers. Bioconjug. Chem. 7, 703–714 (1996).
Vincent, L. et al. Efficacy of dendrimer-mediated angiostatin and TIMP-2 gene delivery on inhibition of tumor growth and angiogenesis: in vitro and in vivo studies. Int. J. Cancer 105, 419–429 (2003).
Wiener, E.C. et al. Dendrimer-based metal chelates: a new class of magnetic resonance imaging contrast agents. Magn. Reson. Med. 31, 1–8 (1994).
Margerum, L.D. et al. Gadolinium(III) DO3A macrocycles and polyethylene glycol coupled to dendrimers. Effect of molecular weight on physical and biological properties of macromolecular magnetic resonance imaging contrast agents. J. Alloys Compd. 249, 185–190 (1997).
Kobayashi, H. & Brechbiel, M.W. Dendrimer-based macromolecular MRI contrast agents: characteristics and application. Mol. Imaging 2, 1–10 (2003).
Ziemer, L.S., Lee, W.M.F., Vinogradov, S.A., Sehgal, C. & Wilson, D.F. Oxygen distribution in murine tumors: characterization using oxygen-dependent quenching of phosphorescence. J. Appl. Physiol. 98, 1503–1510 (2005).
Dunphy, I., Vinogradov, S.A. & Wilson, D.F. Oxyphor R2 and G2: phosphors for measuring oxygen by oxygen-dependent quenching of phosphorescence. Anal. Biochem. 310, 191–198 (2002).
Briñas, R.P., Troxler, T., Hochstrasser, R.M. & Vinogradov, S.A. Phosphorescent oxygen sensor with dendritic protection and two-photon absorbing antenna. J. Am. Chem. Soc. 127, 11851–11862 (2005).
Supattapone, S., Nguyen, H.O.B., Cohen, F.E., Prusiner, S.B. & Scott, M.R. Elimination of prions by branched polyamines and implications for therapeutics. Proc. Natl. Acad. Sci. USA 96, 14529–14534 (1999).
Roy, R. & Baek, M.G. Glycodendrimers: novel glycotope isosteres unmasking sugar coding. Case study with T-antigen markers from breast cancer MUC1 glycoprotein. J. Biotechnol. 90, 291–309 (2002).
Bourne, N. et al. Dendrimers, a new class of candidate topical microbicides with activity against herpes simplex virus infection. Antimicrob. Agents Chemother. 44, 2471–2474 (2000).
Wathier, M., Jung, P.J., Camahan, M.A., Kim, T. & Grinstaff, M.W. Dendritic macromers as in situ polymerizing biomaterials for securing cataract incisions. J. Am. Chem. Soc. 126, 12744–12745 (2004).
Velazquez, A.J. et al. New dendritic adhesives for sutureless ophthalmic surgical procedures. In vitro studies of corneal laceration repair. Arch. Ophthalmol. 122, 867–870 (2004).
Drobnik, J. & Rypacek, F. Soluble synthetic polymers in biological systems. Adv. Polym. Sci. 57, 1–50 (1984).
Roberts, J.C., Bhalgat, M.K. & Zera, R.T. Preliminary biological evaluation of polyamidoamine (PAMAM) Starburst dendrimers. J. Biomed. Mater. Res. 30, 53–65 (1996).
Malik, N. et al. Dendrimers: relationship between structure and biocompatibility in vitro, and preliminary studies on the biodistribution of 125I-labelled polyamidoamine dendrimers in vivo. J. Control. Release 65, 133–148 (2000).
Jevprasesphant, R. et al. The influence of surface modification on the cytotoxicity of PAMAM dendrimers. Int. J. Pharm. 252, 263–266 (2003).
De Jesús, O.L.P., Ihre, H.R., Gagne, L., Fréchet, J.M.J. & Szoka, F.C. Polyester dendritic systems for drug delivery applications: in vitro and in vivo evaluation. Bioconjug. Chem. 13, 453–461 (2002).
Gillies, E.R., Dy, E., Fréchet, J.M.J. & Szoka, F.C. Biological evaluation of polyester dendrimer: poly(ethylene oxide) “bow-tie” hybrids with tunable molecular weight and architecture. Mol. Pharm. 2, 129–138 (2005).
Fuchs, S. et al. A surface-modified dendrimer set for potential application as drug delivery vehicles: synthesis, in vitro toxicity, and intracellular localization. Chemistry 10, 1167–1192 (2004).
Chen, H.T., Neerman, M.F., Parrish, A.R. & Simanek, E.E. Cytotoxicity, hemolysis, and acute in vivo toxicity of dendrimers based on melamine, candidate vehicles for drug delivery. J. Am. Chem. Soc. 126, 10044–10048 (2004).
Hong, S. et al. Interaction of poly(amidoamine) dendrimers with supported lipid bilayers and cells: hole formation and the relation to transport. Bioconjug. Chem. 15, 774–782 (2004).
Kuo, J.H.S., Jan, M.S. & Chiu, H.W. Mechanism of cell death induced by cationic dendrimers in RAW 264.7 murine macrophage-like cells. J. Pharm. Pharmacol. 57, 489–495 (2005).
Neerman, M.F., Zhang, W., Parrish, A.R. & Simanek, E.E. In vitro and in vivo evaluation of a melamine dendrimer as a vehicle for drug delivery. Int. J. Pharm. 281, 129–132 (2004).
Seebach, D., Herrmann, G.F., Lengweiler, U.D., Bachmann, B.M. & Amrein, W. Synthesis and enzymatic degradation of dendrimers from (R)-3-hydroxy-butanoic acid and trimesic acid. Angew. Chem. Int. Edn. Engl. 35, 2795–2797 (1996).
Ihre, H.R., De Jesús, O.L.P., Szoka, F.C. & Fréchet, J.M.J. Polyester dendritic systems for drug delivery applications: design, synthesis, and characterization. Bioconjug. Chem. 13, 443–452 (2002).
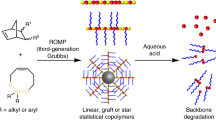
Lee, C.C., Grayson, S.M. & Fréchet, J.M.J. Synthesis of narrow-polydispersity degradable dendronized aliphatic polyesters. J. Polym. Sci. Part A: Polym. Chem. 42, 3563–3578 (2004).
Zhang, W. et al. Evaluation of multivalent dendrimers based on melamine: kinetics of thiol-disulfide exchange depends on the structure of the dendrimer. J. Am. Chem. Soc. 125, 5086–5094 (2003).
Rendle, P.M. et al. Glycodendriproteins: a synthetic glycoprotein mimic enzyme with branched sugar-display potently inhibits bacterial aggregation. J. Am. Chem. Soc. 126, 4750–4751 (2004).
Córdova, A. & Janda, K.D. Synthesis and catalytic antibody functionalization of dendrimers. J. Am. Chem. Soc. 123, 8248–8259 (2001).
Haba, K. et al. Single-triggered trimeric prodrugs. Angew. Chem. Int. Edn. Engl. 44, 716–720 (2005).
Bracci, L. et al. Synthetic peptides in the form of dendrimers become resistant to protease activity. J. Biol. Chem. 278, 46590–46595 (2003).
Hussain, M. et al. A novel anionic dendrimer for improved cellular delivery of antisense oligonucleotides. J. Control. Release 99, 139–155 (2004).
Smet, M., Liao, L.X., Dehaen, W. & McGrath, D.V. Photolabile dendrimers using o-nitrobenzyl ether linkages. Org. Lett. 2, 511–513 (2000).
Watanabe, S., Sato, M., Sakamoto, S., Yamaguchi, K. & Iwamura, M. New dendritic caged compounds: synthesis, mass spectrometric characterization, and photochemical properties of dendrimers with á-carboxy-2-nitrobenzyl caged compounds at their periphery. J. Am. Chem. Soc. 122, 12588–12589 (2000).
Amir, R.J., Pessah, N., Shamis, M. & Shabat, D. Self-immolative dendrimers. Angew. Chem. Int. Edn. Engl. 42, 4494–4499 (2003).
Shum, P., Kim, J.M. & Thompson, D.H. Phototriggering of liposomal drug delivery systems. Adv. Drug Deliv. Rev. 53, 273–284 (2001).
Szalai, M.L., Kevwitch, R.M. & McGrath, D.V. Geometric disassembly of dendrimers: dendritic amplification. J. Am. Chem. Soc. 125, 15688–15689 (2003).
de Groot, F.M.H., Albrecht, C., Koekkoek, R., Beusker, P.H. & Scheeren, H.W. “Cascade-release dendrimers” liberate all end groups upon a single triggering event in the dendritic core. Angew. Chem. Int. Edn. Engl. 42, 4490–4494 (2003).
Li, S., Szalai, M.L., Kevwitch, R.M. & McGrath, D.V. Dendrimer disassembly by benzyl ether depolymerization. J. Am. Chem. Soc. 125, 10516–10517 (2003).
Nishikawa, M., Takakura, Y. & Hashida, M. Pharmacokinetic evaluation of polymeric carriers. Adv. Drug Deliv. Rev. 21, 135–155 (1996).
Yamaoka, T., Tabata, Y. & Ikada, Y. Distribution and tissue uptake of poly(ethylene glycol) with different molecular weights after intravenous administration to mice. J. Pharm. Sci. 83, 601–606 (1994).
Bohrer, M.P., Deen, W.M., Robertson, C.R., Troy, J.L. & Brenner, B.M. Influence of molecular configuration on the passage of macromolecules across the glomerular capillary wall. J. Gen. Physiol. 74, 583–593 (1979).
Ohlson, M. et al. Effects of filtration rate on the glomerular barrier and clearance of four differently shaped molecules. Am. J. Physiol. Renal Physiol. 281, F103–F113 (2001).
Brochard-Wyart, F. & de Gennes, P.G. Injection threshold for a star polymer inside a nanopore. C. R. l'Acadamie. Sci. Ser. II Univers 323, 473–479 (1996).
Lee, C.C., Yoshida, M., Fréchet, J.M.J., Dy, E.E. & Szoka, F.C. In vitro and in vivo evaluation of hydrophilic dendronized linear polymers. Bioconjug. Chem. 16, 535–541 (2005).
Duncan, R. The dawning era of polymer therapeutics. Nat. Rev. Drug Discov. 2, 347–360 (2003).
Drummond, D.C., Meyer, O., Hong, K., Kirpotin, D.B. & Papahadjopoulos, D. Optimizing liposomes for delivery of chemotherapeutic agents to solid tumors. Pharmacol. Rev. 51, 691–744 (1999).
King, H.D. et al. Monoclonal antibody conjugates of doxorubicin prepared with branched peptide linkers: inhibition of aggregation by methoxytriethyleneglycol chains. J. Med. Chem. 45, 4336–4343 (2002).
Choe, Y.H. et al. Anticancer drug delivery systems: multi-loaded 4N-acyl poly(ethylene glycol) prodrugs of ara-C. II. Efficacy in ascites and solid tumors. J. Control. Release 79, 55–70 (2002).
Pasut, G., Scaramuzza, S., Schiavon, O., Mendichi, R. & Veronese, F.M. PEG-epirubicin conjugates with high drug loading. J. Bioact. Compat. Polym. 20, 213–230 (2005).
Defoort, J.P., Nardelli, B., Huang, W., Ho, D.D. & Tam, J.P. Macromolecular assemblage in the design of a synthetic AIDS vaccine. Proc. Natl. Acad. Sci. USA 89, 3879–3883 (1992).
Voit, B. New developments in hyperbranched polymers. J. Polym. Sci. Part A: Polym. Chem. 38, 2505–2525 (2000).
Sunder, A., Heinemann, J. & Frey, H. Controlling the growth of polymer trees: concepts and perspectives for hyperbranched polymers. Chemistry 6, 2499–2506 (2000).
Schlüter, A.D. & Rabe, J.P. Dendronized polymers: synthesis, characterization, assembly at interfaces, and manipulation. Angew. Chem. Int. Edn. Engl. 39, 864–883 (2000).
Gössl, I., Shu, L., Schlüter, A.D. & Rabe, J.P. Molecular structure of single DNA complexes with positively charged dendronized polymers. J. Am. Chem. Soc. 124, 6860–6865 (2002).
Acknowledgements
We are grateful for financial support of dendrimer drug carrier research from the National Institutes of Health (GM 65361 and EB 002047).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
F.C.S. is an inventor on US patent no. 2,661,025, “Self-assembling polynucleotide delivery systems comprising dendrimer polycations.” The University of California has licensed this patent to Qiagen, from which the University of California and F.C.S. receive royalty income.
Supplementary information
Rights and permissions
About this article
Cite this article
Lee, C., MacKay, J., Fréchet, J. et al. Designing dendrimers for biological applications. Nat Biotechnol 23, 1517–1526 (2005). https://doi.org/10.1038/nbt1171
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt1171
This article is cited by
-
Current analytical approaches for characterizing nanoparticle sizes in pharmaceutical research
Journal of Nanoparticle Research (2024)
-
Recent Development and Future Aspects: Nano-Based Drug Delivery System in Cancer Therapy
Topics in Catalysis (2024)
-
Technological advances in the use of viral and non-viral vectors for delivering genetic and non-genetic cargos for cancer therapy
Drug Delivery and Translational Research (2023)
-
Future adoption and consumption of green and sustainable nanoproducts—classifications and synthesis
Nanotechnology for Environmental Engineering (2023)
-
Synthesis and Biological Studies of Cationic Organoiron Star-Shaped Dendrimers with Attached Ketoprofen and Mefenamic Acid at the Peripheries
Journal of Inorganic and Organometallic Polymers and Materials (2023)