Abstract
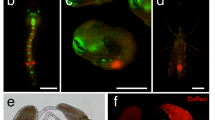
Genetic manipulation of mosquito species that serve as vectors for human malaria is a prerequisite to the implementation of gene transfer technologies for the control of vector-borne diseases. Here we report on the development of transgenic sexing lines for the mosquito Anopheles stephensi, the principal vector of human malaria in Asia. Male mosquitoes, expressing enhanced green fluorescent protein (EGFP) under the control of the β2-tubulin promoter, are identified by their fluorescent gonads in as early as their 3rd instar larval stage, and can be efficiently separated from females using both manual methods and automated sorting machines. Importantly, β2-EGFP males are not impaired in their mating ability and viable fluorescent spermatozoa are also detected in spermathecae of wild-type females mated with transgenic males. The transgenic mosquito lines described here combine most of the features desired and required for a safe application of transgenic methodologies to malaria-control programs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Catteruccia, F. et al. Stable germline transformation of the malaria mosquito Anopheles stephensi. Nature 405, 959–962 (2000).
Grossman, G.L. et al. Germline transformation of the malaria vector Anopheles gambiae, with the piggyBac transposable element. Insect Mol. Biol. 10, 597–604 (2001).
Perera, O.P., Harrell, I.R. & Handler, A.M. Germ-line transformation of the South American malaria vector, Anopheles albimanus, with a piggyBac/EGFP transposon vector is routine and highly efficient. Insect Mol. Biol. 11, 291–297 (2002).
Alphey, L. et al. Malaria control with genetically manipulated insect vectors. Science 298, 119–121 (2002).
Benedict, M.Q. & Robinson, A.S. The first releases of transgenic mosquitoes: an argument for the sterile insect technique. Trends Parasitol. 19, 349–355 (2003).
Knipling, E.F. Sterile-male method of population control. Science 130, 902–904 (1959).
Knipling, E.F. et al. Genetic control of insects of public health importance. Bull. World Health Organ. 38, 421–438 (1968).
Alphey, L. & Andreasen, M. Dominant lethality and insect population control. Mol. Biochem. Parasitol. 121, 173–178 (2002).
Krafsur, E.S., Whitten, C.J. & Novj, J.E. Screwworm eradication in North and Central America. Parasitol. Today 3, 131–137 (1987).
Lindquist, D.A., Abusowa, M. & Hall, J.R. The New World screwworm fly in Lybia: a review of its introduction and eradication. Med. Vet. Entomol. 6, 2–8 (1992).
Robinson, A.S. Genetic sexing strains in medfly, Ceratitis capitata, sterile insect technique programmes. Genetica 116, 5–13 (2002).
Vreysen, M.J. et al. Glossina austeni (Diptera: Glossinidae) eradicated on the island of Unguja, Zanzibar, using the sterile insect technique. J. Econ. Entomol. 93, 123–135 (2000).
Rendon, P., McInnis, D., Lance, D. & Stewart, J. Medfly (Diptera: Tephritidae) genetic sexing: large-scale field comparison of males-only and bisexual sterile fly releases in Guatemala. J. Econ. Entomol. 97, 1547–1553 (2004).
Caceres, C. Mass rearing of temperature sensitive genetic sexing strains in the Mediterranean fruit fly (Ceratitis capitata). Genetica 116, 107–116 (2002).
Lofgren, C.S. et al. Release of chemosterilized males for the control of Anopheles albimanus in El Salvador. 3. Field methods and population control. Am. J. Trop. Med. Hyg. 23, 288–297 (1974).
Patterson, R.S., Weidhaas, D.E., Ford, H.R. & Lofgren, C.S. Suppression and elimination of an island population of Culex pipiens quinquefasciatus with sterile males. Science 168, 1368–1370 (1970).
Curtis, C.F. Genetic sex separation in Anopheles arabiensis and the production of sterile hybrids. Bull. World Health Organ. 56, 453–454 (1978).
Lines, J.D. & Curtis, C.F. Genetic sexing systems in Anopheles arabiensis Patton (Diptera: Culicidae). J. Econ. Entomol. 78, 848–851 (1985).
Seawright, J.A., Kaiser, P.E., Dame, D.A. & Lofgren, C.S. Genetic method for the preferential elimination of females of Anopheles albimanus. Science 200, 1303–1304 (1978).
Sharma, V.P., Patterson, R.S. & Ford, H.R. A device for the rapid separation of male and female mosquito pupae. Bull. World Health Organ. 47, 429–432 (1972).
Kemphues, K.J., Raff, E.C., Raff, R.A. & Kaufman, T.C. Mutation in a testis-specific beta-tubulin in Drosophila: analysis of its effects on meiosis and map location of the gene. Cell 21, 445–451 (1980).
Horn, C. & Wimmer, E.A. A versatile vector set for animal transgenesis. Dev. Genes Evol. 210, 630–637 (2000).
Handler, A.M. & Harrell, R.A., II Germline transformation of Drosophila melanogaster with the piggyBac transposon vector. Insect Mol. Biol. 8, 449–457 (1999).
Furlong, E.E., Profitt, D. & Scott, M.P. Automated sorting of live transgenic embryos. Nat. Biotechnol. 19, 153–156 (2001).
Okanda, F.M. et al. Behavioural determinants of gene flow in malaria vector populations: Anopheles gambiae males select large females as mates. Malar. J. 1, 10 (2002).
Handler, A.M., Zimowska, G.J. & Horn, C. Post-integration stabilization of a transposon vector by terminal sequence deletion in Drosophila melanogaster. Nat. Biotechnol. 22, 1150–1154 (2004).
Acknowledgements
We wish to thank Charles Godfray and Chris Curtis for helpful suggestions, Elisa Petris and Ann Cronin for help with the injections, and Oliver Billker for help with the microphotographs. We also wish to thank Rico Boongarts from Union Biometrica for assisting us in the automated sorting experiments. The project was supported by the Wellcome Trust and BBSRC. F.C. was sponsored by the Wellcome Trust, J.P.B. by the FP6-EU BioMolPar program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Schematic representation of transformation vector pPB[DsRed]β2EGFP. (PDF 91 kb)
Supplementary Table 1
β2-EGFP expression as a marker to predict sex. (PDF 64 kb)
Rights and permissions
About this article
Cite this article
Catteruccia, F., Benton, J. & Crisanti, A. An Anopheles transgenic sexing strain for vector control. Nat Biotechnol 23, 1414–1417 (2005). https://doi.org/10.1038/nbt1152
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt1152
This article is cited by
-
Transcriptional profiling and physiological roles of Aedes aegypti spermathecal-related genes
BMC Genomics (2020)
-
A drug-inducible sex-separation technique for insects
Nature Communications (2020)
-
Improvement and use of CRISPR/Cas9 to engineer a sperm-marking strain for the invasive fruit pest Drosophila suzukii
BMC Biotechnology (2019)
-
High-resolution transcriptional profiling of Anopheles gambiae spermatogenesis reveals mechanisms of sex chromosome regulation
Scientific Reports (2019)
-
A synthetic male-specific sterilization system using the mammalian pro-apoptotic factor in a malaria vector mosquito
Scientific Reports (2019)