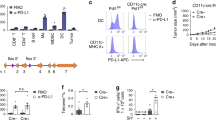
Abstract
Tumor vaccines represent a promising therapeutic approach, but thus far have achieved only limited success in the clinic. The major challenge is to find a means of overcoming inhibitory immune regulatory mechanisms and eliciting effective T-cell responses to antigens preferentially expressed by tumor cells. Here we show that the stimulatory capacity of dendritic cells (DCs) and the magnitude of adaptive immunity are critically regulated by the suppressor of cytokine signaling (SOCS) 1 in DCs. Silencing SOCS1 in antigen-presenting DCs strongly enhances antigen-specific anti-tumor immunity. Our findings indicate that SOCS1 represents an inhibitory mechanism for qualitatively and quantitatively controlling antigen presentation by DCs and the magnitude of adaptive immunity. This study has implications for understanding the regulation of antigen presentation and for developing more effective tumor vaccines by silencing the critical brake in antigen presentation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Steinman, R.M., Hawiger, D. & Nussenzweig, M.C. Tolerogenic dendritic cells. Annu. Rev. Immunol. 21, 685–711 (2003).
Pardoll, D. Does the immune system see tumors as foreign or self? Annu. Rev. Immunol. 21, 807–839 (2003).
Alegre, M.L., Frauwirth, K.A. & Thompson, C.B. T-cell regulation by CD28 and CTLA-4. Nat. Rev. Immunol. 1, 220–228 (2001).
Timmerman, J.M. & Levy, R. Dendritic cell vaccines for cancer immunotherapy. Annu. Rev. Med. 50, 507–529 (1999).
Chen, L. Co-inhibitory molecules of the B7–CD28 family in the control of T-cell immunity. Nat. Rev. Immunol. 4, 336–347 (2004).
Leach, D.R., Krummel, M.F. & Allison, J.P. Enhancement of antitumor immunity by CTLA-4 blockade. Science 271, 1734–1736 (1996).
Hodi, F.S. et al. Biologic activity of cytotoxic T lymphocyte-associated antigen 4 antibody blockade in previously vaccinated metastatic melanoma and ovarian carcinoma patients. Proc. Natl. Acad. Sci. USA 100, 4712–4717 (2003).
Phan, G.Q. et al. Cancer regression and autoimmunity induced by cytotoxic T lymphocyte-associated antigen 4 blockade in patients with metastatic melanoma. Proc. Natl. Acad. Sci. USA 100, 8372–8377 (2003).
Kubo, M., Hanada, T. & Yoshimura, A. Suppressors of cytokine signaling and immunity. Nat. Immunol. 4, 1169–1176 (2003).
Alexander, W.S. & Hilton, D.J. The Role of Suppressors of Cytokine Signaling (SOCS) Proteins in Regulation of the Immune Response. Annu. Rev. Immunol. 22, 503–529 (2004).
Marine, J.C. et al. SOCS1 deficiency causes a lymphocyte-dependent perinatal lethality. Cell 98, 609–616 (1999).
Alexander, W.S. et al. SOCS1 is a critical inhibitor of interferon gamma signaling and prevents the potentially fatal neonatal actions of this cytokine. Cell 98, 597–608 (1999).
Naka, T. et al. SOCS-1/SSI-1-deficient NKT cells participate in severe hepatitis through dysregulated cross-talk inhibition of IFN-gamma and IL-4 signaling in vivo. Immunity 14, 535–545 (2001).
Metcalf, D., Mifsud, S., Di Rago, L. & Alexander, W.S. The lethal effects of transplantation of Socs1−/− bone marrow cells into irradiated adult syngeneic recipients. Proc. Natl. Acad. Sci. USA 100, 8436–8441 (2003).
Hanada, T. et al. Suppressor of cytokine signaling-1 is essential for suppressing dendritic cell activation and systemic autoimmunity. Immunity 19, 437–450 (2003).
Kinjyo, I. et al. SOCS1/JAB is a negative regulator of LPS-induced macrophage activation. Immunity 17, 583–591 (2002).
Hill, J.A. et al. Immune modulation by silencing IL-12 production in dendritic cells using small interfering RNA. J. Immunol. 171, 691–696 (2003).
Livak, K.J. & Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25, 402–408 (2001).
Chong, M.M. et al. Suppressor of cytokine signaling-1 is a critical regulator of interleukin-7-dependent CD8+ T cell differentiation. Immunity 18, 475–487 (2003).
Sledz, C.A., Holko, M., de Veer, M.J., Silverman, R.H. & Williams, B.R. Activation of the interferon system by short-interfering RNAs. Nat. Cell Biol. 5, 834–839 (2003).
Rubinson, D.A. et al. A lentivirus-based system to functionally silence genes in primary mammalian cells, stem cells and transgenic mice by RNA interference. Nat. Genet. 33, 401–406 (2003).
Schroers, R. & Chen, S.Y. Lentiviral transduction of human dendritic cells. Methods Mol. Biol. 246, 451–459 (2004).
Srivastava, P. Interaction of heat shock proteins with peptides and antigen presenting cells: chaperoning of the innate and adaptive immune responses. Annu. Rev. Immunol. 20, 395–425 (2002).
van Elsas, A. et al. Elucidating the autoimmune and antitumor effector mechanisms of a treatment based on cytotoxic T lymphocyte antigen-4 blockade in combination with a B16 melanoma vaccine: comparison of prophylaxis and therapy. J. Exp. Med. 194, 481–489 (2001).
Porgador, A. et al. Predominant role for directly transfected dendritic cells in antigen presentation to CD8+ T cells after gene gun immunization. J. Exp. Med. 188, 1075–1082 (1998).
Garg, S. et al. Genetic tagging shows increased frequency and longevity of antigen-presenting, skin-derived dendritic cells in vivo. Nat. Immunol. 4, 907–912 (2003).
Menges, M. et al. Repetitive injections of dendritic cells matured with tumor necrosis factor alpha induce antigen-specific protection of mice from autoimmunity. J. Exp. Med. 195, 15–21 (2002).
Pasare, C. & Medzhitov, R. Toll pathway-dependent blockade of CD4+CD25+ T cell-mediated suppression by dendritic cells. Science 299, 1033–1036 (2003).
Yang, Y., Huang, C.T., Huang, X. & Pardoll, D.M. Persistent Toll-like receptor signals are required for reversal of regulatory T cell-mediated CD8 tolerance. Nat. Immunol. 5, 508–515 (2004).
You, Z. et al. Induction of vigorous helper and cytotoxic T cell as well as B cell responses by DCs expressing a modified antigen targeting receptor-mediated internalization pathway. J. Immunol. 165, 4581–4592 (2000).
Huang, X.F. et al. A broadly applicable, personalized heat shock protein-mediated oncolytic tumor vaccine. Cancer Res. 63, 7321–7329 (2003).
Chen, J., Yang, A., Bai, X., Cong, Y. & Chen, S-Y. Inactivation of HIV-1 chemokine co-receptor CXCR4 by a novel intraline approach. Nat. Med. 3, 1110–1116 (1997).
Brummelkamp, T.R., Bernards, R. & Agami, R. A system for stable expression of short interfering RNAs in mammalian cells. Science 296, 550–553 (2002).
Acknowledgements
We thank Lisa Rollins, Xiao-Tong Song, Wenhong Ren, Melissa Aldrich, Denise Sanders, Natasha Lapteva and Xue F. Huang for technical assistance and valuable suggestions. This work was supported by grants from the National Institutes of Health (NIH) (R01CA90427, R01AI48480 and R01AI48711), and the Leukemia and Lymphoma Society SCOR award. L.S. was supported by a US Army Prostate Cancer Research Program Postdoctoral fellowship, and K.E. was supported by a predoctoral NIH training grant (T32-AI07495).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Transfection of DCs by siRNA oligos. (PDF 144 kb)
Supplementary Fig. 2
Q-RT-PCR analysis of SOCS1 levels in oligo-transfected DCs. (PDF 67 kb)
Supplementary Fig. 3
Q-RT-PCR analysis of SOCS1 levels during BM-DC culture. (PDF 40 kb)
Supplementary Fig. 4
Transduction of DCs by LV-SOCS1-siRNA. (PDF 672 kb)
Supplementary Fig. 5
Q-RT-PCR analysis of SOCS1 levels in YFP+ LV-transduced DCs. (PDF 65 kb)
Supplementary Fig. 6
Flow cytometric analysis of CD86. (PDF 163 kb)
Supplementary Fig. 7
Flow cytometric analysis of CD40. (PDF 155 kb)
Rights and permissions
About this article
Cite this article
Shen, L., Evel-Kabler, K., Strube, R. et al. Silencing of SOCS1 enhances antigen presentation by dendritic cells and antigen-specific anti-tumor immunity. Nat Biotechnol 22, 1546–1553 (2004). https://doi.org/10.1038/nbt1035
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt1035
This article is cited by
-
H. pylori modulates DC functions via T4SS/TNFα/p38-dependent SOCS3 expression
Cell Communication and Signaling (2020)
-
The silencing of indoleamine 2,3-dioxygenase 1 (IDO1) in dendritic cells by siRNA-loaded lipid nanoparticles enhances cell-based cancer immunotherapy
Scientific Reports (2019)
-
SOCS1 and its Potential Clinical Role in Tumor
Pathology & Oncology Research (2019)
-
A safe and potent anti-CD19 CAR T cell therapy
Nature Medicine (2019)
-
Recent challenges and advances in genetically-engineered cell therapy
Journal of Pharmaceutical Investigation (2018)