Abstract
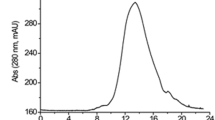
A glycosylation variant of human tissue-type plasminogen activator (tPA) designated longer-acting tissue-type plasminogen activator (LAtPA) was extensively purified from the milk of a transgenic goat by a combination of acid fractionation, hydrophobic interaction chromatography and immunoaffinity chromatography. This scheme provided greater than 8,000-fold purification of the protein, a cumulative yield of 25% and purity greater than 98% as judged by SDS gel electrophoresis. SDS gel electrophoresis revealed that the transgenic enzyme was predominantly the “two chain” form of the protease. The specific activity of the purified transgenic protein, based on the average of the values obtained for three different preparations, was 610,000 U/mg as judged by amidolytic activity assay. This was approximately 84% of the value observed for the recombinant enzyme produced in mouse C127 cells. Analysis of the transgenic protein indicated that it had a significantly different carbohydrate composition from the recombinant enzyme produced in C127 cells. Molecular size analysis of the oligosaccharides from the transgenic and C127 cell-derived LAtPA preparations confirmed their differences and showed that the mouse cell-derived preparation contained larger, complex-type N-linked oligosaccharide structures than the material produced in goat mammary tissue.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bachmann, F. and Kruithof, E.K.O. 1984. Tissue plasminogen activator: Chemical and physiological aspects. Semin. Thromb. Hemostatis 10: 6–17.
Vehar, G.A., Spellman, M.W., Keyt, B.A., Ferguson, C.K., Keck, R.G., Chloupek, R.C., Harris, R., Bennett, W.F., Builder, S.E. and Hancock, W.S. 1986. Characterization studies of human tissue-type plasminogen activator produced by recombinant DNA technology. Cold Spring Harbor Quant. Biol. 51: 551–end562.
Collen, D. 1988. Fibrin-specific thrombolytic therapy. Thromb. Res. Suppl. VIII: 3–14.
Wun, T.C. and Capuano, A. 1985. Spontaneous fibrinolysis in whole human plasma: Identification of tissue activator-related protein as the major plasminogen activator causing spontaneous activity in vitro. J. Biol. Chem. 260: 5061–5066.
Van der Werf, F., Ludbrook, P.A., Bergmann, S.R., Tiefenbrunn, A.J., Fox, K.A.A., Hilaire de Geest, C.B., Verstraete, M., Collen, D. and Sobel, B.E. 1984. Coronary thrombolysis with tissue-type plasminogen activator in patients with evolving myocardial infarction. N. Eng. J. Med. 310: 609–613.
Collen, D., Topol, E.J., Tiefenbrunn, A.J., Gold, H.K., Weisfield, M.L., Sobel, B.E., Leinbach, R.C., Brinker, J.A., Ludbrook, P.A., Yasuda, I., Bulkev, B.H., Robison, A.K., Hutter, A.M., Jr., Bell, W.R., Spadaro, J.J., Jr., Khaw, B.A. and Grossbard, E.B. 1984. Coronary thrombolysis with recombinant human tissue-type plasminogen activator: A prospective, randomized, placebo-controlled trial. Circulation 70: 1012–1017.
Braunwald, E. 1988. Thrombolytic reperfusion of acute myocardial infarction: Resolved and unresolved issues. J. Am. Coll. Cardiol. 12: 85A–92A.
Gordon, K., Lee, E., Vitale, J., Smith, A.E., Westphal, H. and Hennighausen, L. 1987. Production of human tissue-type plasminogen activator in transgenic mice. Bio/Technology 5: 1183–1187.
Pittius, C.W., Hennighausen, L., Lee, E., Westphal, H., Nicols, E., Vitale, J. and Gordon, K. 1988. A milk protein gene promoter directs the expression of human tissue plasminogen activator complementary DNA to the mammary gland in transgenic mice. Proc. Natl. Acad. Sci. U.S.A. 85: 5874–5878.
Clark, A.J., Bessos, H., Bishop, J.O., Brown, P., Harris, S., Lathe, R., McCenaghan, M., Prowse, C., Simons, J.P., Whitelaw, C.B.A. and Wilmut, I. 1989. Expression of human anti-hemophilic factor IX in the milk of transgenic sheep. Bio/Technology 7: 487–492.
Lau, D., Kuzma, G., Wei, C.M., Livingston, D.J. and Hsiung, N. 1987. A modified human tissue plasminogen activator with extended half-life in vivo. Bio/Technology 5: 953–958.
Pennica, D., Holmes, W.E., Kohr, W.J., Harkins, R.N., Vehar, G.A., Ward, C.A., Bennett, W.F., Yelverton, E., Seeburg, P.H., Heyneker, H.L. and Goeddel, D.V. 1983. Cloning and expression of human tissue-type plasminogen activator cDNA in E. coli. Nature 301: 214–220.
Wallen, P., Pohl, G., Bergsdorf, N., Ranby, M., Ny, T. and Jornvall, H. 1983. Purification and characterization of a melanoma cell plasminogen activator. Eur. J. Biochem. 132: 681–686.
Pfeiffer, G., Schmidt, M., Strube, K.H. and Geyer, R. 1989. Carbohydrate structure of recombinant human uterine tissue plasminogen activator expressed in mouse epithelial cells. Eur. J. Biochem. 186: 273–286.
Cole, E.S., Nichols, E.H., Poisson, L., Harnois, M.L., Lee, K. and Livingston, D.J. 1991. The effect of complex oligosaccharides on clearance of plasminogen activator. In preparation.
Tilley, D.A., Singer, A., Harris-Brandts, M. and Moscarello, M.A. 1991. The major oligosaccharide of bovine α-lactalbumin carries terminal β-linked GalNAc. Glycoconjugate J. 8: 249.
Nakata, N., Furakawa, K., Greenwalt, D.E. and Kobata, A. 1991. Bovine milk epithelial glycoprotein IV (CD36) contains novel sialylate tri- and tetra- antennary complex-type sugar chains having a β-N-acetyl-galactosamine linked to an N-acetylglucosamine residue. Glycoconjugate J. 8: 250.
Ranby, M., Bergsdorf, N., Pohl, G. and Wallen, P. 1982. Isolation of two variants of native one-chain tissue plasminogen activator. FEBS Lett. 146: 289–292.
Einarson, M., Brandt, J. and Kaplan, L. 1985. Large scale purification of human tissue type plasminogen activator using monoclonal antibodies. Biochem. Biophys. Acta 830: 1–10.
Rijken, D.C., Emeis, J.J. and Gerwig, G.J. 1985. On the composition and function of the carbohydrate moiety of tissue-type plasminogen activator from human melanoma cells. Thromb. Hemostasis 54: 788–791.
Laemmli, U.K. 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685.
Hardy, M.R., Townsend, R.R. and Lee, Y.C. 1988. Monosaccharide analysis of glycoconjugates by anion exchange chromatography with pulsed amperometric detection. Anal. Biochem. 170: 54–62.
Powell, L.D. and Hart, I.G.W. 1986. Quantitation of picomole levels of N-acetyl and N-glycolylneuraminic acids by HPLC-adaption of the thiobarbituric acid assay. Anal. Biochem. 157: 179–185.
Hirani, S., Bernasconi, R.J. and Rasmussen, J.R. 1987. Use of N-glycanase to release asparagine linked oligosaccharides for structural analysis. Anal. Biochem. 162: 485–492.
Basa, L.J. and Spellman, M.W. 1990. Analysis of glycoprotein oligosaccharides by high pH anion-exchange chromatography. Chromatography 499: 205–220.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Denman, J., Hayes, M., O'Day, C. et al. Transgenic Expression of a Variant of Human Tissue-Type Plasminogen Activator in Goat Milk: Purification and Characterization of the Recombinant Enzyme. Nat Biotechnol 9, 839–843 (1991). https://doi.org/10.1038/nbt0991-839
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0991-839
This article is cited by
-
Transgenesis applied to goat: current applications and ongoing research
Transgenic Research (2012)
-
Animal transgenesis: state of the art and applications
Journal of Applied Genetics (2007)
-
Molecular Farming in Plants: A Current Perspective
Journal of Plant Biochemistry and Biotechnology (2001)