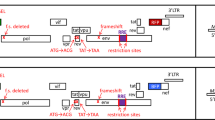
Abstract
Over two-thirds of the current gene therapy protocols use retroviral gene transfer systems. We have developed an efficient retroviral-based method that allows rapid identification of gene transfer in living mammalian cells. Cells were generated containing a gene for an improved (humanized, red-shifted) version of the Aequorea victoria green fluorescent protein (hRGFP) from a retroviral vector. The hRGFP gene was used to produce an amphotropic vector producer cell line that demonstrated vibrant green fluorescence after excitation with blue light. A375 melanoma cells transduced with the retroviral vector demonstrated stable green fluorescence. Both PA317 murine fibroblasts and A375 human cell lines containing the vector were easily detected by FACS analysis. These vectors represent a substantial improvement over currently available gene transfer marking systems. Bright, long-term expression of the hRGFP gene in living eukaryotic cells will advance the study of gene transfer, gene expression, and gene product function in vitro and in vivo particularly for human gene therapy applications.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Prasher, D.C. 1995. Using GFP to see the light. Trends Genetics 11: 320–323.
Chalfie, M., Tu, Y., Euskirchen, G., Ward, W.W., and Prasher, D.C. 1994. Green Fluorescent Protein as a marker for gene expression. Science 263: 802–805.
Prasher, D.C., Eckenrode, V.K., Ward, W.W., Prendergrast, F.G., and Cormier, M.J. 1992. Primary structure of the Aequorea victoria green-fluorescent protein. Gene 111: 229–233.
Cody, C.W., Prasher, D.C., Westler, W.M., Prendergast, F.G., and Ward, W.W. 1993. Chemical structure of the hexapeptide chromophore of the Aequorea green fluorescent protein. Biochemistry 32: 1212–1218.
Inouye, S. and Tsuji, F.I. 1994. Aequorea green fluorescent protein expression of the gene and fluorescence characteristics of the recombinant protein. FEBS Lett 341: 277–280.
Shimomura, O. 1979. Structure of the chromophore of Aequorea green fluorescent protein. FEBS Lett. 104: 220–222.
Heim, R., Prasher, D.C., and Tsien, R.T. 1994. Wavelength mutations and posttrans-lational autoxidation of green fluorescent protein. Proc. Natl. Acad. Sci. USA 91: 12501–12504.
Ward, W.W. and Bokman, S.H. 1982. Reversible denaturation of Aequorea green fluorescent protein: Physical separation and characterization of the renatured protein. Biochemistry 21: 4535–4540.
Perozzo, M.A., Ward, K.B., Thompson, R.B., and Ward, W.W. 1988. X-ray diffraction and time resolved fluorescence analyses of Aequorea green fluorescent protein crystals. J. Biol. Chem. 263: 7713–7716.
Deschamps, J.R., Miller, C.E., and Ward, K.B. 1988. Rapid purification of recombinant green fluorescent protein using the hydrophobe properties of an HPLC size-exclusion column. Prot Expres. Purific. 6: 555–558.
Heim, R., Cubitt, A.B., and Tsien, R.Y. 1995. Improved green fluorescence. Nature 373: 63–664.
Zototukhin, S., Potter, M., Hauswirth, W., Guy, J., and Muzyczka, N. 1996. A “humanized” green fluorescent protein cONA adapted for high level expression in mammalian cells. J. Virol. In press.
Delagrave, S., Hawtin, R.E., Silva, C.M., Yang, M.M., and Youvan, D.C. 1995. Red-shifted excitation mutants of the Green Fluorescent Protein. Bio/Technology 13: 151–154.
Rizzuto, R., Brinin, M., Pizo, P., Murgia, M., and Pozzan, T. 1995. Chimeric green fluorescent protein as a tool for visualizing subcellular organelles in living cells. Current Biol. 5: 635–642.
Kaether, C. and Gerdes, H. 1995. Visualization of protein transport along the secretory pathway using green fluorescent protein. FEBS Lett. 36: 267–271.
Ogawa, H., Inouye, S., Tsuji, F.I., Yasada, K., and Umesono, K. 1995. Localization, trafficking, and temperature-dependence of the Aequoreagreen fluorescent protein in cultured vertebrate cells. Proc. Natl. Acad. Sci. USA 92: 11899–11903.
Olson, K.R., Mcintosh, R., and Olmstread, J.B. 1995. Analysis of MAP 4 function in living cells using green fluorescent protein (GFP) chimeras. J Cell Biol. 130: 639–650.
Marshall, J., Molloy, R., Moss, G.W., Howe, J.R., and Hughes, T.E. 1995. The jellyfish green fluorescent protein: A new tool for studying ion channel expression and function. Neuron 14: 211–215.
Ikawa, M., Kominami, K., Yoshimura, Y., Tanaka, K., Nishimune, Y., and Okabe, M. 1995. Green fluorescent protein as a marker in transgenic mice. Develop. Growth Differ. 37: 455–459.
Miller, A.D. and Buttimore, C. 1986. Redesign of retrovirus packaging cell lines to avoid recombination leading to helper virus production. Molec. Cell Biol. 6: 2895–2902.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Levy, J., Muldoon, R., Zolotukhin, S. et al. Retroviral transfer and expression of a humanized, red-shifted green fluorescent protein gene into human tumor cells. Nat Biotechnol 14, 610–614 (1996). https://doi.org/10.1038/nbt0596-610
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0596-610
This article is cited by
-
The rat sodium iodide symporter gene permits more effective radioisotope concentration than the human sodium iodide symporter gene in human and rodent cancer cells
Cancer Gene Therapy (2003)
-
Generation of high-titer retroviral vector-producing macrophages as vehicles for in vivo gene transfer
Gene Therapy (2001)
-
In vitro neurogenesis by progenitor cells isolated from the adult human hippocampus
Nature Medicine (2000)
-
Accurate estimation of transduction efficiency necessitates a multiplex real-time PCR
Gene Therapy (2000)
-
Immune response to green fluorescent protein: implications for gene therapy
Gene Therapy (1999)