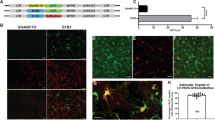
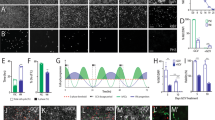
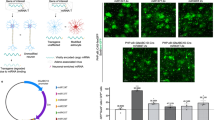
Abstract
Ex vivo gene transfer is emerging as a promising therapeutic approach to human neurodegenerative diseases. By combining efficient methodologies for cell amplification and gene delivery, large numbers of cells can be generated with the capacity to synthesize therapeutic molecules. These cells can then be transplanted into the degenerating central nervous system (CNS). Applying this approach to human diseases will require the development of suitable cellular vehicles, as well as safe gene delivery systems capable of tightly controlled transgene expression. For such brain repair technologies, human neural progenitors may be extremely valuable, because of their human CNS origin and developmental potential. We have used these cells to develop a system for the regulated expression of a gene of therapeutic potential. We report the construction of a single adenovirus encoding human tyrosine hydroxylase 1 (hTH-1) under the negative control of the tetracycline-based gene regulatory system. Human neural progenitors infected with this vector produced large amounts of hTH-1. Most importantly, doxycycline allowed a reversible switch of transgene transcription both in vitro and in vivo. This system may be applied to the development of therapies for human neurodegenerative diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Marsden, C.D. & Parkes, J.D. "On-off" effects in patients with Parkinson's disease on chronic levodopa therapy. Lancet 1, 292–296 (1976).
Lindvall, O. Neural transplantation: a hope for patients with Parkinson's disease. Neuroreport 8, iii–x ( 1997).
Raymon, H.K., Thode, S. & Gage, F.H. Application of ex vivo gene therapy in the treatment of Parkinson's disease. Exp. Neurol. 144, 82 –91 (1997).
Horellou, P., Sabate, O., Buc-Caron, M.H. & Mallet, J. Adenovirus-mediated gene transfer to the central nervous system for Parkinson's disease. Exp. Neurol. 144, 131– 138 (1997).
Uchida, K. & Kohsaka, S. Current advances in neural transplantation. Human Cell 2, 150–155 (1989).
Wolff, J.A. et al. Grafting fibroblasts genetically modified to produce L-dopa in a rat model of Parkinson disease. Proc. Natl. Acad. Sci. USA 86 , 9011–9014 (1989).
Horellou, P., Brundin, P., Kalen, P., Mallet, J. & Björklund, A. In vivo release of dopa and dopamine from genetically engineered cells grafted to the denervated rat striatum. Neuron 5, 393–402 ( 1990).
Fisher, L.J., Jinnah, H.A., Kale, L.C., Higgins, G.A. & Gage, F.H. Survival and function of intrastriatally grafted primary fibroblasts genetically modified to produce L-dopa. Neuron 6, 371–380 (1991).
Whittemore, S.R., Eaton, M.J. & Onifer, S.M. Gene therapy and the use of stem cells for central nervous system regeneration. Adv. Neurol. 72, 113–119 (1997).
Martinez, S.A. & Björklund, A. Immortalized neural progenitor cells for CNS gene transfer and repair. Trends Neurosci. 20, 530–538 ( 1997).
Fisher, L.J. Neural precursor cells: applications for the study and repair of the central nervous system. Neurobiol. Dis. 4, 1– 22 (1997).
Groves, A.K. et al. Repair of demyelinated lesions by transplantation of purified O-2A progenitor cells. Nature 362, 453– 455 (1993).
Snyder, E.Y., Taylor, R.M. & Wolfe, J.H. Neural progenitor cell engraftment corrects lysosomal storage throughout the MPS VII mouse brain. Nature 374, 367–370 (1995).
Buc, C.M. Neuroepithelial progenitor cells explanted from human fetal brain proliferate and differentiate in vitro. Neurobiol. Dis. 2, 37–47 (1995).
Sabate, O. et al. Transplantation to the rat brain of human neural progenitors that were genetically modified using adenoviruses. Nat. Genet. 9, 256–260 (1995).
Miller, N. & Whelan, J. Progress in transcriptionally targeted and regulatable vectors for genetic therapy. Hum. Gene Ther. 8, 803–815 (1997).
Obeso, J.A., Grandas, F., Herrero, M.T. & Horowski, R. The role of pulsatile versus continuous dopamine receptor stimulation for functional recovery in Parkinson's disease. Eur. J. Neurosci. 6, 889–897 (1994).
Saez, E., No, D., West, A. & Evans, R.M. Inducible gene expression in mammalian cells and transgenic mice. Curr. Opin. Biotechnol. 8, 608–616 ( 1997).
Gossen, M. & Bujard, H. Tight control of gene expression in mammalian cells by tetracycline-responsive promoters. Proc. Natl. Acad. Sci .USA 89, 5547–5551 (1992).
Gossen, M. et al. Transcriptional activation by tetracyclines in mammalian cells. Science 268, 1766–1769 ( 1995).
Corti, O., Horellou, P., Colin, P., Cattaneo, E. & Mallet, J. Intracerebral tetracycline-dependent regulation of gene expression in grafts of neural precursors. Neuroreport 7, 1655–1659 (1996).
Mayford, M. et al. Control of memory formation through regulated expression of a CaMKII transgene. Science 274, 1678– 1683 (1996).
Yu, J.S., Sena, E.M., Paulus, W., Breakefield, X.O. & Reeves, S.A. Retroviral delivery and tetracycline-dependent expression of IL-1beta-converting enzyme (ICE) in a rat glioma model provides controlled induction of apoptotic death in tumor cells. Cancer Res. 56, 5423–5427 (1996).
Adra, C.N., Boer, P.H. & McBurney, M.W. Cloning and expression of the mouse pgk-1 gene and the nucleotide sequence of its promoter. Gene 60, 65–74 (1987).
McGeady, M.L., Wood, T.G., Maizel, J.V. & Vande, W.G. Sequences upstream from the mouse c-mos oncogene may function as a transcription termination signal. DNA 5, 289–298 (1986).
Ungerstedt, U. & Arbuthnott, G.W. Quantitative recording of rotational behavior in rats after 6-hydroxydopamine lesions of the nigrostriatal dopamine system. Brain Res. 24, 485–493 (1970).
Harding, T.C., Geddes, B.J., Murphy, D., Knight, D. & Uney, J.B. Switching transgene expression in the brain using an adenoviral tetracycline-regulatable system. Nat. Biotechnol. 16 , 553–555 (1998).
StLouis, D. & Verma, I.M. An alternative approach to somatic cell gene therapy. Proc. Natl. Acad. Sci. USA 85, 3150–3154 (1988).
Scharfmann, R., Axelrod, J.H. & Verma, I.M. Long-term in vivo expression of retrovirus-mediated gene transfer in mouse fibroblast implants. Proc. Natl. Acad. Sci. USA 88, 4626–4630 ( 1991).
Palmer, T.D., Rosman, G.J., Osborne, W.R. & Miller, A.D. Genetically modified skin fibroblasts persist long after transplantation but gradually inactivate introduced genes. Proc. Natl. Acad. Sci. USA 88, 1330–1334 ( 1991).
Kay, M.A. et al. Expression of humans alpha 1-antitrypsin in dogs after autologous transplantation of retroviral transduced hepatocytes. Proc. Natl. Acad. Sci. USA 89, 89–93 ( 1992).
Lim, B., Apperley, J.F., Orkin, S.H. & Williams, D.A. Long-term expression of human adenosine deaminase in mice transplanted with retrovirus-infected hematopoietic stem cells. Proc. Natl. Acad. Sci. USA. 86, 8892–8896 (1989).
Moullier, P., Marechal, V., Danos, O. & Heard, J.M. Continuous systemic secretion of a lysosomal enzyme by genetically modified mouse skin fibroblasts. Transplantation 56, 427– 432 (1993).
Kaplitt, M.G. et al. Preproenkephalin promoter yields region-specific and long-term expression in adult brain after direct in vivo gene transfer via a defective herpes simplex viral vector. Proc. Natl. Acad. Sci. USA 91, 8979–8983 (1994).
Kojima, H. et al. Adenovirus-mediated transduction with human glial cell line-derived neurotrophic factor gene prevents 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced dopamine depletion in striatum of mouse brain. Biochem. Biophys. Res. Commun. 238, 569–573 (1997).
Geny, C. et al. Long-term delayed vascularization of human neural transplants to the rat brain. J. Neurosci. 14, 7553– 7562 (1994).
Kistner, A. et al. Doxycycline-mediated quantitative and tissue-specific control of gene expression in transgenic mice. Proc. Natl. Acad. Sci. USA 93, 10933–10938 ( 1996).
A-Mohammadi, S., Alvarez, V.L., Ashworth, L.J. & Hawkins, R.E. Delay in resumption of the activity of tetracycline-regulatable promoter following removal of tetracycline analogues. Gene. Ther. 4, 993–997 (1997).
Brundin, P. et al. Human fetal dopamine neurons grafted in a rat model of Parkinson's disease: immunological aspects, spontaneous and drug-induced behaviour, and dopamine release. Exp. Brain. Res. 70, 192 –208 (1988).
Freese, A. et al. Phenotypic recovery in parkinsonian rats: relationship between numbers of transduced cells and behavior using an adenoviral vector expressing tyrosine hydroxylase. Society for Neuroscience, Abstracts 23 , 540 (1997).
Pedersen, E.B., Zimmer, J. & Finsen, B. Triple immunosuppression protects murine intracerebral, hippocampal xenografts in adult rat hosts: effects on cellular infiltration, major histocompatibility complex antigen induction and blood-brain barrier leakage. Neuroscience 78, 685– 701 (1997).
Stratford, P.L., Makeh, I., Perricaudet, M. & Briand, P. Widespread long-term gene transfer to mouse skeletal muscles and heart. J. Clin. Invest. 90, 626–630 (1992).
Reinhard, J.J., Smith, G.K. & Nichol, C.A. A rapid and sensitive assay for tyrosine-3-monooxygenase based upon the release of 3[H2O] and adsorption of [3H]-tyrosine by charcoal. Life Sci. 39, 2185–2189 (1986).
Dumas, S., Horellou, P., Helin, C. & Mallet, J. Co-expression of tyrosine hydroxylase messenger RNA 1 and 2 in human ventral mesencephalon revealed by digoxigenin- and biotin-labelled oligodeoxyribonucleotides. J. Chem. Neuroanat. 5, 11–18 (1992).
Lanièce, P. et al. A novel rat tyrosine hydroxylase mRNA species generated by alternative splicing. J. Neurochem. 66, 1819– 1825 (1996).
Acknowledgements
We thank Gilles Pitiot for helpful discussions and critical review of the manuscript. We also thank Amelia Sanchez-Capelo for her unremitting contribution to the construction of AdPGK♦tet♦hTH-1. We thank Novartis for generously providing cyclosporine A. This work was supported by grants from the Association Francaise contre les Myopathies, the Centre National de la Recherche Scientifique, the Conséil Régional d'Ile de France, EC contract No. 951012, the European Science Foundation, the Institut pour la Recherche sur la Moelle Epiniere and Retina France.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Corti, O., Sabaté, O., Horellou, P. et al. A single adenovirus vector mediates doxycycline-controlled expression of tyrosine hydroxylase in brain grafts of human neural progenitors. Nat Biotechnol 17, 349–354 (1999). https://doi.org/10.1038/7901
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/7901
This article is cited by
-
Mifepristone-inducible transgene expression in neural progenitor cells in vitro and in vivo
Gene Therapy (2016)
-
Constitutive Gs activation using a single-construct tetracycline-inducible expression system in embryonic stem cells and mice
Stem Cell Research & Therapy (2011)
-
Genetic Modification of Neural Stem Cells
Molecular Therapy (2008)
-
Human neural progenitors deliver glial cell line-derived neurotrophic factor to parkinsonian rodents and aged primates
Gene Therapy (2006)
-
Liver-specific expression of interferon γ following adenoviral gene transfer controls hepatitis B virus replication in mice
Gene Therapy (2005)