Abstract
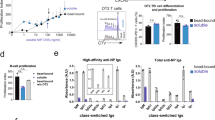
Monoclonal antibodies (MAbs) specific for surface antigens of lymphocytes are being used to target and deplete tumorous or normal lymphocytes in vivo. Here, we report evidence for the existence of antigenic epitopes on IgE that are accessible on IgE-secreting B cells but not on other cells bearing IgE. Among 42 murine MAbs specific for human IgE, two were shown by fluorescence flow cytometric analyses to bind to IgE-secreting cell lines but not to IgE bound to high-affinity IgE. Fc receptors (Fc∈RI) on basophils or low-affinity IgE receptors (Fc∈RII) on other cell types. Neither could they induce histamine release from basophils of various donors even under very permissive conditions. These antibodies may be useful for targeting IgE-secreting B cells in patients suffering from IgE-mediated allergies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Ishizaka, K. and Ishizaka, T. 1989. Allergy. p. 867–888. In: Fundamental Immunology 2nd Ed. Paul, W.E. (Ed.). Raven Press, Ltd., New York.
Eisen, H.N. 1980. Antibody-mediated immediate-type hypersensitivity. p. 468–492. In: Microbiology. Davis, B. D., Dulbecco, R., Eisen, H. N., and Ginsberg, H. S. (Eds.). Harper & Row Publishers, Philadelphia.
Frick, O.L. 1987. Immediate hypersensitivity. p. 197–227. In: Basic and Clinical Immunology 6th Ed. Stites, D. P., Stobo, J. D., and Wells, J. V. (Eds.). Appleton and Lange, Norwalk, Connecticut.
Metzger, H., Alcaraz, G., Hohman, R., Kinet, J.-P., Pribluda, V. and Quarto, R. 1986. The receptor with high affinity for immunoglobulin E. Ann. Rev. Immunol. 4: 419–470.
Ishizaka, T. and Ishizaka, K. 1984. Activation of mast cells for mediator release through IgE receptors. Prog. Allergy 34: 188–235.
Lichtenstein, L.M., Levy, D.A. and Ishizaka, K. 1970. In vitro reverse anaphylaxis: characteristics of anti-IgE mediated histamine release. Immunology 19: 831–842.
Spiegelberg, H.L. 1984. Structure and function of Fc receptors for IgE on lymphocytes, monocytes, and macrophages. Adv. Immunol. 35: 61–88.
Gordon, J., Flores-Romo, L., Cairns, J.A., Millsum, M.J., Lane, P.J., Johnson, G.D. and MacLennon, I.C.M. 1989. CD23: a multifunctional receptor/lymphokine?. Immunol. Today 10: 153–157.
Yaoita, Y., Kumagai, Y., Okumura, K. and Honjo, T. 1982. Expression of lymphocyte surface IgE does not require switch recombination. Nature 297: 697–699.
MacKenzie, T. and Dosch, H.-M. 1989. Clonal and molecular characteristics of the human IgE-committed B cell subset. J. Exp. Med. 169: 407–430.
Ishida, N., Ueda, S., Hayashida, H., Miyata, T. and Honjo, T. 1982. The nucleotide sequence of the mouse immunoglobulin epsilon gene: comparison with the human epsilon gene sequence. EMBO J. 1: 1117–1123.
MacGlashan, Jr., D.W. and Lichtenstein, L.M. 1980. The purification of human basophils. J. Immunol. 124: 2519–2521.
Gordon, J., Webb, A.J., Walker, L., Guy, G.R. and Rowe, M. 1986. Evidence for an association between CD23 and the receptor for a low molecular weight B cell growth factor. Eur. J. Immunol. 16: 1627–1630.
Sun, L.K., Liou, R.S. and Chang, T.W. 1989. Expression of a mouse/human chimeric IgE. J. Cell Biol. 109: 289a.
Hamburger, R.N. 1975. Peptide inhibition of the Prausnitz-Küstner reaction. Science 189: 389–390.
Stanworth, D.R. and Burt, D.S. 1986. Anti-∈-chain antibodies as probes in the study of mast-cell triggering. Mol. Immunol. 23: 1231–1235.
Rousseaux-Prevost, R., Rousseaux, J. and Bazin, H. 1987. Studies of the IgE binding site to rat mast cell receptor with proteolytic fragments and with a monoclonal antibody directed against epsilon heavy chain: evidence that the combining sites are located in the C∈3 domain. Mol. Immunol. 24: 187–196.
Helm, B., Marsh, P., Vercelli, D., Padlan, E., Gould, H. and Geha, R. 1988. The mast cell binding site on human immunoglobulin E. Nature 331: 180–184.
Hook, W.A., Berenstein, E.H. and Siraganian, R.P. 1987. Monoclonal antibodies defining epitopes on IgE. Fed. Proc. 46: 1346.
Baniyash, M., Eshhar, Z. and Rivnay, B. 1986. Relationships between epitopes on IgE recognized by defined monoclonal antibodies and by the Fc∈ receptor on basophils. J. Immunol. 136: 588–593.
Vercelli, D., Helm, B., Marsh, P., Padlan, E., Geha, R.S. and Gould, H., 1989. B-cell binding site on human immunoglobulin E. Nature 338: 649–651.
Burrows, B., Martinez, F.D., Halonen, M., Barbee, R.A. and Cline, M.G. 1989. Association of asthma with serum IgE and skin-test reactivity to allergens. New Engl. J. Med. 320: 271–277.
Levy, D.A. and Chen, J., 1970. IgE-deficient person. New Engl. J. Med. 283: 541–542.
Ishizaka, K. 1988. IgE-binding factors and regulation of the IgE antibody response. Ann. Rev. Immunol. 6: 513–534.
Delespesse, G., Sarfati, M. and Hofstetter, H., 1989. IgE-binding factors. Immunol. Today 10: 159–164.
Coffman, R.L. and Carty, J. 1986. A T cell activity that enhances polyclonal IgE production and its inhibition by interferon-γ. J. Immunol. 136: 949–954.
Paul, W.E. and Ohara, J. 1987. B-cell stimulatory factor-1/interleukin 4. Ann. Rev. Immunol. 5: 429–459.
Finkelman, F.D., Katona, L.M., Urban, Jr., J.F., Snapper, C.M., Ohara, J. and Paul, W.E. 1986. Suppression of in vivo polyclonal IgE response by monoclonal antibody to the lymphocyte B cell stimulatory factor I. Proc. Nad. Acad. Sci. USA 83: 9675–9678.
Meeker, T.C., Lowder, J., Maloney, D.G., Miller, R.A., Thielemans, K., Warnke, R. and Levy, R. 1985. A clinical trial of anti-idiotype therapy for B cell malignancy. Blood 65: 1349–1363.
Hale, G., Dyer, M.J.S., Clark, M.R., Phillips, J.M., Marcus, R., Riechmann, L., Winter, G. and Waldmann, H. 1988. Remission induction in non-Hodgkin's lymphoma with reshaped monoclonal antibody CAMPATH-1H. Lancet ii: 1394–1399.
Ortho Multicenter Transplant Study Group. 1985. A randomized clinical trial of OKT3 monoclonal antibody for acute rejection of cadaveric renal transplants. New Engl. J. Med. 313: 337–342.
Cobbold, S.P., Jayasuriya, A., Nash, A., Prospero, T.D. and Waldmann, H. 1984. Therapy with monoclonal antibodies by elimination of T cell subsets in vivo . Nature 312: 548–551.
Waldor, M.K., Sriram, S., Hardy, R., Herzenberg, L.A., Herzenberg, L.A., Lanier, L., Lim, M. and Steinman, L. 1985. Reversal of experimental allergic encephalomyelitis with monoclonal antibody to a T-cell subset marker. Science 227: 415–417.
Sriram, S., Carroll, L., Fortin, S., Cooper, S. and Ranges, G. 1988. In vivo immunomodulation by monoclonal anti-CD4 antibody II. Effect on T cell response to myelin basic protein and experimental allergic encephalomyelitis. J. Immunol. 141: 464–468.
Cosimi, A.B., Burton, R.C., Colvin, R.B., Goldstein, G., Delmonico, F.L., LaQuaglia, M.P., Tolkoff-Rubin, N., Rubin, R.H., Herrin, J.T. and Russell, P.S. 1981. Treatment of acute renal allograft rejection with OKT3 monoclonal antibody. Transplantation 32: 535–539.
Waldmann, H. 1989. Manipulation of T-cell responses with monoclonal antibody. Ann. Rev. Immunol. 7: 407–444.
Beatty, J.D., Beatty, B.G. and Vlahos, W.G. 1987. Measurement of monoclonal antibody affinity by non-competitive enzyme immunoassay. J. Immunol. Meth. 100: 173–179.
Siraganian, R.P. and Hook, W.A. 1980. Histamine release and assay methods for the study of human allergy. p. 808–821. Manual of Clinical Chemistry 2nd Ed. Rose, N. R. and Friedman, H. (Eds.). American Soc. Microbiol., Washington, D.C.
MacGlashan, Jr., D.W., Peters, S.P., Warner, J. and Lichtenstein, L.M. 1986. Characteristics of human basophil sulfidopeptide leukotriene release: releasability defined as the ability of the basophil to respond to dimeric cross-links. J. Immunol. 136: 2231.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chang, T., Davis, F., Sun, NC. et al. Monoclonal Antibodies Specific for Human IgE-Producing B Cells: A Potential Therapeutic for IgE-Mediated Allergic Diseases. Nat Biotechnol 8, 122–126 (1990). https://doi.org/10.1038/nbt0290-122
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0290-122
This article is cited by
-
Combining Anti-IgE Monoclonal Antibodies and Oral Immunotherapy for the Treatment of Food Allergy
Clinical Reviews in Allergy & Immunology (2022)
-
Skewed Exposure to Environmental Antigens Complements Hygiene Hypothesis in Explaining the Rise of Allergy
Acta Biotheoretica (2017)
-
Severe asthma and the omalizumab option
Clinical and Molecular Allergy (2008)
-
Allergy Vaccines
Clinical Immunotherapeutics (1996)
-
Novel approaches in immunotherapy
Clinical Reviews in Allergy (1994)