Abstract
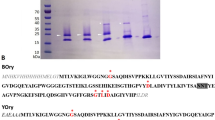
Escherichia coli cells transformed with the expression plasmid pLlec9 synthesize leucoagglutinating phytohemagglutinin (PHA–L) encoded by a nuclear gene of Phaseolus vulgaris. Pure, unglycosylated PHA–L can be isolated by affinity chromatography from lysates of these bacteria. The E. coli synthesized PHA–L (Eco PHA–L) appears to be tetrameric and resembles deglycosylated, processed PHA–L from bean seed in its subunit molecular weight. Eco PHA–L agglutinates human peripheral blood lymphocytes at concentrations as low as 0.5 μg/ml, and its specific mitogenicity exceeds that of commercially available PHA–L. At levels up to 450 μg/ml, Eco PHA–L does not agglutinate human type–0 erythrocytes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Yachnin, S. and Svenson, R.H. 1972. The immunological and physico-chemical properties of mitogenic proteins derived from Phaseolus vulgaris. Immunology 22: 871–883.
Osborn, T.C., Ausloos, K.A., Brown, J.W.S., and Bliss, F.A. 1983. Bean lectins III. Evidence for greater complexity in the structural model of Phaseolus vulgaris lectin. Plant Sci. Lett. 31: 193–203.
Chilson, O.P., Boylston, A.W., and Crumpton, M.J. 1984. Phaseolus vulgaris phytohemagglutinin (PHA) binds to the human T-lymphocyte antigen receptor. EMBO J. 3: 3239–3245.
Hoffman, L.M. and Donaldson, D.D. 1985. Characterization of two Phaseolus vulgaris phytohemagglutinin genes closely linked on the chromosome. EMBO J. 4: 883–889.
Bollini, R., Ceriotti, A., Daminati, M.G., and Vitale, A. 1985. Glycosylation is not needed for the intracellular transport of phytohemagglutinin in developing Phaseolus vulgaris cotyledons and for the maintenance of its biological activities. Physiol. Plant. 65: 15–22.
Tkacz, J.S. and Lampen, J.O. 1975. Tunicamycin inhibition of polyisoprenyl N-acetylglucosaminyl pyrophosphate formation in calf-liver microsomes. Biochem. Biophys. Res. Commun. 65: 248–257.
Allen, L.W., Svenson, R.H., and Yachnin, S. 1969. Purification of mitogenic proteins derived from Phaseolus vulgaris: isolation of potent and weak phytohemagglutinins possessing mitogenic activity. Proc. Natl. Acad. Sci. USA 63: 334–341.
Bollini, R. and Chrispeels, M.J. 1983. In vivo and in vitro processing of seed reserve protein in the endoplasmic reticulum: evidence for two glycosylation steps. J. Cell Biol. 96: 999–1007.
Leavitt, R.D., Felsted, R.L., and Bachur, N.R. 1977. Biological and biochemical properties of Phaseolus vulgaris isolectins. J. Biol. Chem. 252: 2961–2966.
Vitale, A., Ceriotti, A., and Bollini, R. 1985. Molecular analysis of a phytohemagglutinin-defective cultivar of Phaseolus vulgaris L. Planta 166: 201–207.
Grey, G.L., Baldridge, J.S., McKeown, K.S., Heynecker, H.L., and Chang, C.N. 1985. Periplasmic production of correctly processed human growth hormone in Escherichia coli: natural and bacterial signal peptides are interchangeable. Gene 39: 247–254.
Felsted, R.L., Leavitt, R.D., and Bachur, N.R. 1975. Purification of the phytohemagglutinin family of proteins from red kidney beans (Phaseolus vulgaris) by affinity chromatography. Biochim. Biophys. Acta 405: 72–81.
Edge, A.S.B., Faltynek, C.R., Hof, L., Reichert, L.E., and Weber, P. 1981. Deglycosylation of glycoproteins by trifluoromethanesulfonic acid. Anal. Biochem. 118: 131–137.
Laemmli, U.K. 1970. Cleavage of structural proteins during the assembly of the lead of bacteriophage T4 . Nature 227: 680–685.
Towbin, H., Staehelin, T., and Gordon, J. 1979. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc. Natl. Acad. Sci. USA 76: 4350–4354.
Mierendorf, R. 1985. Immunodetection using alkaline phosphatase conjugated second antibodies. Promega Notes 3.
Oh, Y.H. and Conrad, R.A. 1971. Some properties of mitogenic components isolated from phytohemagglutinin by a preparative gel electrophoresis. Arch. Biochem. Biophys. 146: 525–530.
Miller, J.B., Hsu, R., Heinrikson, R., and Yachnin, S. 1975. Extensive homology between the subunits of the phytohemagglutinin proteins derived from Phaseolus vulgaris. Proc. Natl. Acad. Sci. USA 72: 1388–1391.
Talmadge, K., Stahl, S., and Gilbert, W. 1980. Eukaryotic signal sequence transports insulin antigen in Escherichia coli. Proc. Natl. Acad. Sci. USA 77: 3369–3373.
Smith, K.A. 1984. Interleukin 2. Ann. Rev. Immunol. 2: 319–333.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hoffman, L., Donaldson, D. Synthesis of Mitogenic Phytohemagglutinin–L in Escherichia Coli. Nat Biotechnol 5, 157–160 (1987). https://doi.org/10.1038/nbt0287-157
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0287-157