Abstract
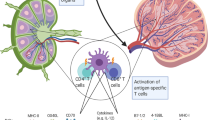
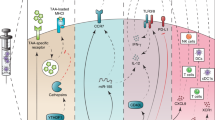
Vaccination with dendritic cells (DCs) pulsed with antigenic peptides derived from various tumor antigens has great, but as yet significantly unrealized, potential in cancer treatment. Here, we describe a strategy for prolonged presentation of an MHC class I–restricted self-peptide on DCs through linkage of it to a cell penetrating peptide (CPP). DCs loaded with a peptide derived from tyrosinase-related protein 2 (TRP2) covalently linked to a CPP1 sequence retained full capacity to stimulate T cells for at least 24 h, completely protected immunized mice from subsequent tumor challenge, and significantly inhibited lung metastases in a 3-day tumor model. DCs pulsed with TRP2 alone failed to provide any of these protections. In addition, we demonstrate that both CD4+ and CD8+ T cells were required for potent antitumor immunity. This CPP-based approach may be generally applicable to enhance the efficacy of DC-based peptide vaccines against cancer and other diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Wang, R.-F. & Rosenberg, S.A. Human tumor antigens for cancer vaccine development. Immunol. Rev. 170, 85–100 (1999).
Boon, T. & Van der Bruggen, P. Human tumor antigens recognized by T lymphocytes. J. Exp. Med. 183, 725–729 (1996).
Gilboa, E. The makings of a tumor rejection antigen. Immunity 11, 263–270 (1999).
Houghton, A.N., Gold, J.S. & Blachere, N.E. Immunity against cancer: lessons learned from melanoma. Curr. Opin. Immunol. 13, 134–140 (2001).
Dallal, R.M. & Lotze, M.T. The dendritic cell and human cancer vaccines. Curr. Opin. Immunol. 12, 583–588 (2000).
Schuler, G. & Steinman, R.M. Dendritic cells as adjuvants for immune-mediated resistance to tumors. J. Exp. Med. 186, 1183–1187 (1997).
Banchereau, J. & Steinman, R.M. Dendritic cells and the control of immunity. Nature 392, 245–252 (1998).
Celluzzi, C.M., Mayordomo, J.I., Storkus, W.J., Lotze, M.T. & Falo, L.D., Jr. Peptide-pulsed dendritic cells induce antigen-specific CTL-mediated protective tumor immunity. J. Exp. Med. 183, 283–287 (1996).
Paglia, P., Chiodoni, C., Rodolfo, M. & Colombo, M.P. Murine dendritic cells loaded in vitro with soluble protein prime cytotoxic T lymphocytes against tumor antigen in vivo. J. Exp. Med. 183, 317–322 (1996).
Young, J.W. & Inaba, K. Dendritic cells as adjuvants for class I major histocompatibility complex-restricted antitumor immunity. J. Exp. Med. 183, 7–11 (1996).
Nestle, F.O. et al. Vaccination of melanoma patients with peptide- or tumor lysate-pulsed dendritic cells. Nat. Med. 4, 328–332 (1998).
Thurner, B. et al. Vaccination with mage-3A1 peptide-pulsed mature, monocyte-derived dendritic cells expands specific cytotoxic T cells and induces regression of some metastases in advanced stage IV melanoma. J. Exp. Med. 190, 1669–1678 (1999).
Bellone, M. et al. Relevance of the tumor antigen in the validation of three vaccination strategies for melanoma. J. Immunol. 165, 2651–2656 (2000).
Schreurs, M.W. et al. Dendritic cells break tolerance and induce protective immunity against a melanocyte differentiation antigen in an autologous melanoma model. Cancer Res. 60, 6995–7001 (2000).
Parkhurst, M.R. et al. Improved induction of melanoma reactive CTL with peptides from the melanoma antigen gp100 modified at HLA-A0201 binding residues. J. Immunol. 157, 2539–2548 (1996).
Rosenberg, S.A. et al. Immunologic and therapeutic evaluation of a synthetic tumor-associated peptide vaccine for the treatment of patients with metastatic melanoma. Nat. Med. 4, 321–327 (1998).
Slansky, J.E. et al. Enhanced antigen-specific antitumor immunity with altered peptide ligands that stabilize the MHC–peptide–TCR complex. Immunity 13, 529–538 (2000).
Labeur, M.S. et al. Generation of tumor immunity by bone marrow-derived dendritic cells correlates with dendritic cell maturation stage. J. Immunol. 162, 168–175 (1999).
Cella, M., Engering, A., Pinet, V., Pieters, J. & Lanzavecchia, A. Inflammatory stimuli induce accumulation of MHC class II complexes on dendritic cells. Nature 388, 782–787 (1997).
Ludewig, B., Odermatt, B., Ochsenbein, A.F., Zinkernagel, R.M. & Hengartner, H. Role of dendritic cells in the induction and maintenance of autoimmune diseases. Immunol. Rev. 169, 45–54 (1999).
Frankel, A.D. & Pabo, C.O. Cellular uptake of the tat protein from human immunodeficiency virus. Cell 55, 1189–1193 (1988).
Elliott, G. & O'Hare, P. Intercellular trafficking and protein delivery by a herpesvirus structural protein. Cell 88, 223–233 (1997).
Phelan, A., Elliott, G. & O'Hare, P. Intercellular delivery of functional p53 by the herpesvirus protein VP22 [see comments]. Nat. Biotechnol. 16, 440–443 (1998).
Lin, Y.Z., Yao, S.Y., Veach, R.A., Torgerson, T.R. & Hawiger, J. Inhibition of nuclear translocation of transcription factor NF-kappa B by a synthetic peptide containing a cell membrane-permeable motif and nuclear localization sequence. J. Biol. Chem. 270, 14255–14258 (1995).
Rojas, M., Donahue, J.P., Tan, Z. & Lin, Y.Z. Genetic engineering of proteins with cell membrane permeability. Nat. Biotechnol. 16, 370–375 (1998).
Fawell, S. et al. Tat-mediated delivery of heterologous proteins into cells. Proc. Natl. Acad. Sci. USA 91, 664–668 (1994).
Kim, D.T. et al. Introduction of soluble proteins into the MHC class I pathway by conjugation to an HIV tat peptide. J. Immunol. 159, 1666–1668 (1997).
Schwarze, S.R., Ho, A., Vocero-Akbani, A. & Dowdy, S.F. In vivo protein transduction: delivery of a biologically active protein into the mouse. Science 285, 1569–1572 (1999).
Lindgren, M., Hallbrink, M., Prochiantz, A. & Langel, U. Cell-penetrating peptides. Trends Pharmacol. Sci. 21, 99–103 (2000).
Caron, N.J. et al. Intracellular delivery of a Tat-eGFP fusion protein into muscle cells. Mol. Ther. 3, 310–318 (2001).
Wang, R.-F., Appella, E., Kawakami, Y., Kang, X. & Rosenberg, S.A. Identification of TRP-2 as a human tumor antigen recognized by cytotoxic T lymphocytes. J. Exp. Med. 184, 2207–2216 (1996).
Bloom, M.B. et al. Identification of tyrosinase-related protein 2 as a tumor rejection antigen for the B16 melanoma. J. Exp. Med. 185, 453–460 (1997).
Parkhurst, M.R. et al. Identification of a shared HLA-A*0201-restricted T-cell epitope from the melanoma antigen tyrosinase-related protein 2 (TRP2). Cancer Res. 58, 4895–4901 (1998).
Zeh, H.J., Perry-Lalley, D., Dudley, M.E., Rosenberg, S.A. & Yang, J.C. High avidity CTLs for two self-antigens demonstrate superior in vitro and in vivo antitumor efficacy. J. Immunol. 162, 989–994 (1999).
Overwijk, W.W. et al. Vaccination with a recombinant vaccinia virus encoding a “self” antigen induces autoimmune vitiligo and tumor cell destruction in mice: requirement for CD4+ T lymphocytes. Proc. Natl. Acad. Sci. USA 96, 2982–2987 (1999).
Bowne, W.B. et al. Coupling and uncoupling of tumor immunity and autoimmunity. J. Exp. Med. 190, 1717–1722 (1999).
Winzler, C. et al. Maturation stages of mouse dendritic cells in growth factor-dependent long-term cultures. J. Exp. Med. 185, 317–328 (1997).
Srivastava, P.K. Immunotherapy of human cancer: lessons from mice. Nat. Immunol. 1, 363–366 (2000).
Suhrbier, A. et al. Peptide epitope induced apoptosis of human cytotoxic T lymphocytes. Implications for peripheral T cell deletion and peptide vaccination. J. Immunol. 150, 2169–2178 (1993).
Casares, N. et al. Immunization with a tumor-associated CTL epitope plus a tumor-related or unrelated Th1 helper peptide elicits protective CTL immunity. Eur. J. Immunol. 31, 1780–1789 (2001).
Wang, R.-F. The role of MHC class II-restricted tumor antigens and CD4+ T cells in antitumor immunity. Trends Immunol. 22, 269–276 (2001).
Greenberg, P.D. Adoptive T cell therapy of tumors: mechanisms operative in the recognition and elimination of tumor cells. Adv. Immunol. 49, 281–355 (1991).
Pardoll, D.M. & Topalian, S.L. The role of CD4+ T cell responses in antitumor immunity. Curr. Opin. Immunol. 10, 588–594 (1998).
Hung, K. et al. The central role of CD4+ T cells in the antitumor immune response. J. Exp. Med. 188, 2357–2368 (1998).
Toes, R.E., Ossendorp, F., Offringa, R. & Melief, C.J. CD4 T cells and their role in antitumor immune responses. J. Exp. Med. 189, 753–756 (1999).
Specht, J.M. et al. Dendritic cells retrovirally transduced with a model antigen gene are therapeutically effective against established pulmonary metastases. J. Exp. Med. 186, 1213–1221 (1997).
Acknowledgements
We thank Malcolm Brenner for his helpful suggestions and critical reading of the manuscript and Tihui Fu for assistance in some experiments.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Web Figure 1.
Phenotype characterization and MLR assay of DCs. A) Surface phenotype of mature and immature DCs generated different culture conditions. DCs were stained anti-CD11c-PE along with anti-I-A-FITC, anti-B7-1-FITC and anti-B7-2-FITC, respectively, and analyzed by flow cytometry. Double positive staining of mutaure DC for CD11c and co-stimulatory B7.1, B7.2 and MHC class II molecules was 55-70% while double positive staining for immature DC was only about 15-20%. B) Allogeneic MLR of mature, immature DCs and splenocytes. DCs were cocultured with allogeneic BALB/c T cells isolated from bulk splenocytes by passing cell through an immunoaffinity column. [3H]-thymidine incorporation from triplicate wells were obtained after subtraction of the background counts from irradiated stimulators and T cells alone, and are plotted as the average counts for each stimulator:effector ratio. (GIF 100 kb)
Rights and permissions
About this article
Cite this article
Wang, RF., Wang, H. Enhancement of antitumor immunity by prolonging antigen presentation on dendritic cells. Nat Biotechnol 20, 149–154 (2002). https://doi.org/10.1038/nbt0202-149
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0202-149
This article is cited by
-
Mechanistic insights into the efficacy of cell penetrating peptide-based cancer vaccines
Cellular and Molecular Life Sciences (2018)
-
High immunogenic potential of p53 mRNA-transfected dendritic cells in patients with primary breast cancer
Breast Cancer Research and Treatment (2011)
-
Generation in vivo of peptide-specific cytotoxic T cells and presence of regulatory T cells during vaccination with hTERT (class I and II) peptide-pulsed DCs
Journal of Translational Medicine (2009)
-
Efficient MHC Class I Presentation by Controlled Intracellular Trafficking of Antigens in Octaarginine-modified Liposomes
Molecular Therapy (2008)
-
A simple but effective cancer vaccine consisting of an antigen and a cationic lipid
Cancer Immunology, Immunotherapy (2008)