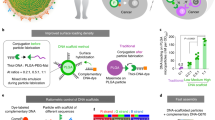
Abstract
Therapeutic ex vivo T-cell expansion is limited by low rates and T-cell products of limited functionality. Here we describe a system that mimics natural antigen-presenting cells (APCs) and consists of a fluid lipid bilayer supported by mesoporous silica micro-rods. The lipid bilayer presents membrane-bound cues for T-cell receptor stimulation and costimulation, while the micro-rods enable sustained release of soluble paracrine cues. Using anti-CD3, anti-CD28, and interleukin-2, we show that the APC-mimetic scaffolds (APC-ms) promote two- to tenfold greater polyclonal expansion of primary mouse and human T cells compared with commercial expansion beads (Dynabeads). The efficiency of expansion depends on the density of stimulatory cues and the amount of material in the starting culture. Following a single stimulation, APC-ms enables antigen-specific expansion of rare cytotoxic T-cell subpopulations at a greater magnitude than autologous monocyte-derived dendritic cells after 2 weeks. APC-ms support over fivefold greater expansion of restimulated CD19 CAR-T cells than Dynabeads, with similar efficacy in a xenograft lymphoma model.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
04 July 2018
In the version of this supplementary file originally posted online, the y axis in Supplementary Figure 4 was labeled "ng per bead," instead of "pg per bead." The error has been corrected in this file as of 4 July 2018.
References
Rosenberg, S.A. & Restifo, N.P. Adoptive cell transfer as personalized immunotherapy for human cancer. Science 348, 62–68 (2015).
June, C.H., Riddell, S.R. & Schumacher, T.N. Adoptive cellular therapy: a race to the finish line. Sci. Transl. Med. 7, 280ps7 (2015).
Fesnak, A.D., June, C.H. & Levine, B.L. Engineered T cells: the promise and challenges of cancer immunotherapy. Nat. Rev. Cancer 16, 566–581 (2016).
Maude, S.L. et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. N. Engl. J. Med. 371, 1507–1517 (2014).
Lee, D.W. et al. T cells expressing CD19 chimeric antigen receptors for acute lymphoblastic leukaemia in children and young adults: a phase 1 dose-escalation trial. Lancet 385, 517–528 (2015).
Brentjens, R.J. et al. Safety and persistence of adoptively transferred autologous CD19-targeted T cells in patients with relapsed or chemotherapy refractory B-cell leukemias. Blood 118, 4817–4828 (2011).
Turtle, C.J. et al. CD19 CAR-T cells of defined CD4+:CD8+ composition in adult B cell ALL patients. J. Clin. Invest. 126, 2123–2138 (2016).
Neelapu, S.S. et al. Kte-C19 (anti-CD19 CAR T Cells) induces complete remissions in patients with refractory diffuse large B-cell lymphoma (DLBCL): results from the pivotal phase 2 Zuma-1. Blood 128, LBA-6 (2016).
Huppa, J.B. & Davis, M.M. T-cell-antigen recognition and the immunological synapse. Nat. Rev. Immunol. 3, 973–983 (2003).
Lee, K.-H. et al. The immunological synapse balances T cell receptor signaling and degradation. Science 302, 1218–1222 (2003).
Alarcón, B., Mestre, D. & Martínez-Martín, N. The immunological synapse: a cause or consequence of T-cell receptor triggering? Immunology 133, 420–425 (2011).
Minguet, S., Swamy, M., Alarcón, B., Luescher, I.F. & Schamel, W.W.A. Full activation of the T cell receptor requires both clustering and conformational changes at CD3. Immunity 26, 43–54 (2007).
Hasan, A.N., Selvakumar, A. & O'Reilly, R.J. Artificial antigen presenting cells: an off the shelf approach for generation of desirable T-cell populations for broad application of adoptive immunotherapy. Adv. Genet. Eng. 4, 130 (2015).
Hollyman, D. et al. Manufacturing validation of biologically functional T cells targeted to CD19 antigen for autologous adoptive cell therapy. J. Immunother. 32, 169–180 (2009).
Maus, M.V. et al. Ex vivo expansion of polyclonal and antigen-specific cytotoxic T lymphocytes by artificial APCs expressing ligands for the T-cell receptor, CD28 and 4-1BB. Nat. Biotechnol. 20, 143–148 (2002).
Zappasodi, R. et al. The effect of artificial antigen-presenting cells with preclustered anti-CD28/-CD3/-LFA-1 monoclonal antibodies on the induction of ex vivo expansion of functional human antitumor T cells. Haematologica 93, 1523–1534 (2008).
Perica, K. et al. Enrichment and expansion with nanoscale artificial antigen presenting cells for adoptive immunotherapy. ACS Nano 9, 6861–6871 (2015).
Mandal, S. et al. Polymer-based synthetic dendritic cells for tailoring robust and multifunctional T cell responses. ACS Chem. Biol. 10, 485–492 (2015).
Steenblock, E.R. & Fahmy, T.M. A comprehensive platform for ex vivo T-cell expansion based on biodegradable polymeric artificial antigen-presenting cells. Mol. Ther. 16, 765–772 (2008).
Fadel, T.R. et al. A carbon nanotube-polymer composite for T-cell therapy. Nat. Nanotechnol. 9, 639–647 (2014).
Sunshine, J.C., Perica, K., Schneck, J.P. & Green, J.J. Particle shape dependence of CD8+ T cell activation by artificial antigen presenting cells. Biomaterials 35, 269–277 (2014).
Fadel, T.R. et al. Enhanced cellular activation with single walled carbon nanotube bundles presenting antibody stimuli. Nano Lett. 8, 2070–2076 (2008).
Meyer, R.A. et al. Biodegradable nanoellipsoidal artificial antigen presenting cells for antigen specific T-cell activation. Small 11, 1519–1525 (2015).
Steenblock, E.R., Fadel, T., Labowsky, M., Pober, J.S. & Fahmy, T.M. An artificial antigen-presenting cell with paracrine delivery of IL-2 impacts the magnitude and direction of the T cell response. J. Biol. Chem. 286, 34883–34892 (2011).
Novartis CTL019 Oncologic Drugs Advisory Committee Briefing Document (2017).
Li, Y. & Kurlander, R.J. Comparison of anti-CD3 and anti-CD28-coated beads with soluble anti-CD3 for expanding human T cells: differing impact on CD8 T cell phenotype and responsiveness to restimulation. J. Transl. Med. 8, 104 2010).
Jin, C. et al. Allogeneic lymphocyte-licensed DCs expand T cells with improved antitumor activity and resistance to oxidative stress and immunosuppressive factors. Mol. Ther. Methods Clin. Dev. 1, 14001 (2014).
Hendriks, J. et al. CD27 is required for generation and long-term maintenance of T cell immunity. Nat. Immunol. 1, 433–440 (2000).
Hunder, N.N. et al. Treatment of metastatic melanoma with autologous CD4+ T cells against NY-ESO-1. N. Engl. J. Med. 358, 2698–2703 (2008).
Mackensen, A. et al. Phase I study of adoptive T-cell therapy using antigen-specific CD8+ T cells for the treatment of patients with metastatic melanoma. J. Clin. Oncol. 24, 5060–5069 (2006).
Rooney, C.M. et al. Infusion of cytotoxic T cells for the prevention and treatment of Epstein-Barr virus-induced lymphoma in allogeneic transplant recipients. Blood 92, 1549–1555 (1998).
Satthaporn, S. et al. Dendritic cells are dysfunctional in patients with operable breast cancer. Cancer Immunol. Immunother. 53, 510–518 (2004).
Wölfl, M. & Greenberg, P.D. Antigen-specific activation and cytokine-facilitated expansion of naive, human CD8+ T cells. Nat. Protoc. 9, 950–966 (2014).
Grakoui, A. et al. The immunological synapse: a molecular machine controlling T cell activation. Science 285, 221–227 (1999).
Kim, J. et al. Injectable, spontaneously assembling, inorganic scaffolds modulate immune cells in vivo and increase vaccine efficacy. Nat. Biotechnol. 33, 64–72 (2015).
Li, W.A. et al. The effect of surface modification of mesoporous silica micro-rod scaffold on immune cell activation and infiltration. Biomaterials 83, 249–256 (2016).
Jerabek, H., Pabst, G., Rappolt, M. & Stockner, T. Membrane-mediated effect on ion channels induced by the anesthetic drug ketamine. J. Am. Chem. Soc. 132, 7990–7997 (2010).
Torres, A.J., Contento, R.L., Gordo, S., Wucherpfennig, K.W. & Love, J.C. Functional single-cell analysis of T-cell activation by supported lipid bilayer-tethered ligands on arrays of nanowells. Lab Chip 13, 90–99 (2013).
Gattinoni, L. et al. Acquisition of full effector function in vitro paradoxically impairs the in vivo antitumor efficacy of adoptively transferred CD8+ T cells. J. Clin. Invest. 115, 1616–1626 (2005).
Cohen, C.J. et al. Isolation of neoantigen-specific T cells from tumor and peripheral lymphocytes. J. Clin. Invest. 125, 3981–3991 (2015).
Strønen, E. et al. Targeting of cancer neoantigens with donor-derived T cell receptor repertoires. Science 352, 1337–1341 (2016).
Chauvin, J.-M. HLA anchor optimization of the melan-A-HLA-A2 epitope within a long peptide is required for efficient cross-priming of human tumor-reactive T cells. J. Immunol. 188, 2102–2110 (2012).
Turtle, C.J. & Riddell, S.R. Artificial antigen-presenting cells for use in adoptive immunotherapy. Cancer J. 16, 374–381 (2010).
Eggermont, L.J., Paulis, L.E., Tel, J. & Figdor, C.G. Towards efficient cancer immunotherapy: advances in developing artificial antigen-presenting cells. Trends Biotechnol. 32, 456–465 (2014).
Puu, G. & Gustafson, I. Planar lipid bilayers on solid supports from liposomes--factors of importance for kinetics and stability. Biochim. Biophys. Acta 1327, 149–161 (1997).
Anderson, N.A., Richter, L.J., Stephenson, J.C. & Briggman, K.A. Characterization and control of lipid layer fluidity in hybrid bilayer membranes. J. Am. Chem. Soc. 129, 2094–2100 (2007).
Collins, M.D. & Keller, S.L. Tuning lipid mixtures to induce or suppress domain formation across leaflets of unsupported asymmetric bilayers. Proc. Natl. Acad. Sci. USA 105, 124–128 (2008).
Reich, C. et al. Asymmetric structural features in single supported lipid bilayers containing cholesterol and GM1 resolved with synchrotron X-Ray reflectivity. Biophys. J. 95, 657–668 (2008).
Longo, G.S., Schick, M. & Szleifer, I. Stability and liquid-liquid phase separation in mixed saturated lipid bilayers. Biophys. J. 96, 3977–3986 (2009).
Kwong, B., Liu, H. & Irvine, D.J. Induction of potent anti-tumor responses while eliminating systemic side effects via liposome-anchored combinatorial immunotherapy. Biomaterials 32, 5134–5147 (2011).
Koo, H. et al. Bioorthogonal copper-free click chemistry in vivo for tumor-targeted delivery of nanoparticles. Angew. Chem. Int. Ed. 51, 11836–11840 (2012).
Desai, R.M., Koshy, S.T., Hilderbrand, S.A., Mooney, D.J. & Joshi, N.S. Versatile click alginate hydrogels crosslinked via tetrazine-norbornene chemistry. Biomaterials 50, 30–37 (2015).
Imai, C. et al. Chimeric receptors with 4-1BB signaling capacity provoke potent cytotoxicity against acute lymphoblastic leukemia. Leukemia 18, 676–684 (2004).
Acknowledgements
This work was supported by the National Institutes of Health (NIH) (1R01EB015498 U01 CA214369) and the Wyss Institute for Biologically Inspired Engineering at Harvard University. D.K.Y.Z. was supported by the Canadian Institutes of Health Research (CIHR-DFSA). S.T.K. was supported by an HHMI ISRF. This work was performed in part at the Center for Nanoscale Systems (CNS), a member of the National Nanotechnology Coordinated Infrastructure Network (NNCI), which is supported by the National Science Foundation under NSF award no. 1541959. CNS is part of Harvard University. We thank the National Institutes of Health (NIH) Tetramer Core Facility for the SIINFEKL/H-2K(b) biotinylated monomer, Alexa Fluor 647-labeled SIINFEKL/H-2K(b) tetramer, CLGGLLTMV/HLA-A*0201 biotinylated monomer, Alexa Fluor 647-labeled CLGGLLTMV/HLA-A*02:01 tetramer, GLCTLVAML/HLA-A*02:01 biotinylated monomer, and Alexa Fluor 647-labeled GLCTLVAML/HLA-A*0201 tetramer, N. Shastri for the B3Z cell line, and G. Freeman for the T2 cell line. We thank Unum Therapeutics for the luciferized Raji cell line and the 19BBz CAR-T cells. We also thank C. Stamoulis from Boston Children′s Hospital and the Harvard Catalyst for her help with statistical analysis; Harvard Catalyst is supported, in part, by the NIH (UL1 TR001102). Lastly, we thank R. Bates, M. Pezone, B. Schultes, L. Edwards, G. Motz, T. Barnitz, T. Hickman, K. McGinness, J. Ritz, M. Maus, T. Snyder, and W.A. Li for valuable scientific discussions.
Author information
Authors and Affiliations
Contributions
A.S.C. and D.J.M. conceived and designed the experiments. A.S.C., D.K.Y.Z., and S.T.K. performed the experiments. A.S.C., D.K.Y.Z., and D.J.M. analyzed the data. A.S.C., D.K.Y.Z., and D.J.M. wrote the manuscript. All authors discussed the results and commented on the manuscript. The principal investigator is D.J.M.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Extended characterization of MSR-SLBs
(a) Representative brightfield microscopy of MSRs. Scale bar, 100 μm. (b) Size distribution of POPC liposomes as measured by DLS. (c) Representative images of lipid-coated MSRs showing the aggregation at lipid-to-MSR 1:20 (w/w). MSRs (left), fluorophore-tagged phospholipid (middle), and co-localization of MSRs and lipid (right). Scale bar, 50 μm. (d) Quantification of lipid retention onto MSRs for different phosphatidylcholines of increasing saturation. C:D are shown in the labels. Retention of different lipid coatings on MSRs over time in PBS (e) and RPMI-1640 containing 10% serum (cRPMI; f). (g) Representative overlaid fluorescence microscopy images of different lipid-coated MSRs, maintained in cRPMI under standard cell culture conditions. Scale bar, 100 μm. (h) Quantification of recombinant human IL-21 (left) and TGF-β (right) released from MSR-SLBs in vitro over time (data points) with sigmoidal curve fitted to data points (IL-21, R2 = 0.99; TGF-β, R2 = 0.97). Approximately 76 ± 4% and 75 ± 5% of the input IL-21 and TGF-β was loaded into MSR-SLBs, respectively. Physical properties of the proteins are listed. Data in (b) represents mean size distribution of three independent samples. Data in (d-f, h) represent mean ± s.d. of three experimental replicates and are representative of at least two independent experiments.
Supplementary Figure 2 Association of T cells with APC-ms
Representative microscopy images of MSR-SLBs not presenting any surface cues (-cue), or surface-presenting αCD3 and αCD28 (APC-ms; +cue), cultured with primary mouse T cells for one day. Images taken at low (left) or high (right) magnification. Cells and material are visible in brightfield images (top) and MSR-SLB lipid coatings are visible in the green channel. Merged images are shown on the bottom. Low magnification scale bar, 500 μm, high magnification scale bar, 100 μm.
Supplementary Figure 3 Extended characterization of polyclonally expanded primary mouse T cells
(a) Viability of primary mouse T cells isolated from C57BL/6J mice cultured with αCD3/αCD28 Dynabeads of varying doses (D1, D2, D3) or APC-ms of varying formulations (A1, A2, A3, A4; see Table 1), at day 7. (b) Expansion of T cells that were either untreated (mock), or cultured with free cues (50 nM αCD3, 50 nM αCD28, 30 U/ml IL-2), commercial T cell expansion beads (Dynabead, dose D1), IL-2-loaded MSR-SLBs without T cell cues presented on the bilayer surface (MSR-SLB (-cue)), or APC-ms (formulation A1). Curves for mock and free were indistinguishable from the MSR-SLB (-cue) curve. (c) Frequencies of CD4 and CD8 single positive cells (expressed as a ratio of CD4+ to CD8+ cells) isolated from C57BL/6J mice in cultures with Dynabeads or APC-ms, evaluated using FACS. (d) Quantification of CD4+ and CD8+ cells isolated from BALB/c mice after 7 days of culture with APC-ms (A4) or Dynabeads (D2). (e) Representative plots showing CD4 and CD8 expression among T cells derived from C57BL/6J or BALB/c mice cultured with APC-ms (A4) for 7 days. (f) Quantification of Granzyme B positive cells among CD8+ cells expanded with Dynabeads (D1) or APC-ms (A1). (g) Quantification of FoxP3 positive cells among CD4+ cells expanded with Dynabeads (D1) or APC-ms (A1). FACS data gated on Fluorescence Minus One (FMO) controls for each sample, at each timepoint. Data in (b, f-g) represent mean ± s.d. of three experimental replicates. Data in (a, c-d) represent mean ± s.d. of n=4 mice. Data in (a-c, f-g) were conducted using primary T cells from C57BL/6 mice; data in (d) were conducted using primary T cells from BALB/c mice. All data are representative of at least two independent experiments.
Supplementary Figure 4 Characterization of Dynabead doses and comparison to APC-ms formulations
(a) Quantification of total amount of αCD3/αCD28 presented on commercial Dynabeads via BCA assay. Approximately 0.46-0.54 ng of αCD3/αCD28 is presented per Dynabead. (b) Amount of αCD3/αCD28 presented on varying Dynabead doses and APC-ms formulations (see Table 1). Data in (a) represents mean ± s.d. of three experimental replicates and are representative of at least two independent experiments.
Supplementary Figure 5 Extended phenotypic characterization of polyclonally expanded primary human T cells
FACS quantification of (a) cell viability and (b) CD62L and CCR7 co-expression among CD3+ cells over time, in samples cultured with either Dynabeads (D1) or APC-ms of varying formulations (see Table 1). Data represents mean ± s.e.m of at least three different donor samples and is representative of at least two independent experiments.
Supplementary Figure 6 Extended characterization of primary human T cells cultured with antigen-specific APC-ms or autologous monocyte-derived dendritic cells
Antigen-specific enrichment (a) and expansion (b) of CLG- or GLC-specific CD8+ T cells cultured with either APC-ms presenting either CLG or GLC or autologous moDCs pulsed with CLG or GLC (right), quantified via FACS. Each line color denotes an individual donor (donors 1, 6, 8, 9, 10, 12), allowing visualization of intra-donor trends. APC-ms consistently promoted antigen-specific T cell expansion but significant donor-to-donor variability in how T cells responded to stimulation was observed. (c) Total expansion of all cells in cultures with APC-ms (left), or autologous moDCs (right). Data are from different donor samples and are representative of at least two independent experiments. APC-ms/CLG, n=4; APC-ms/GLC, n=6; moDC/CLG, n=2; moDC/GLC, n=4.
Supplementary Figure 7 Extended functional characterization of primary human T cells expanded with CLG- or GLC-presenting APC-ms
(a) Quantification of IFNγ secretion by CD8+ T cells cultured with APC-ms presenting either CLG or GLC, following co-culture with T2 cells that were unpulsed (-Peptide), pulsed with CLG (+CLG), or pulsed with GLC (+GLC). (b) Representative plots showing IFNγ and TNFα expression by CD8+ T cells cultured with APC-ms presenting CLG or GLC, following co-culture with T2 cells that were unpulsed (-Peptide; top), pulsed with CLG (+CLG; middle), or pulsed with GLC (+GLC; bottom). Data in (a) represent mean ± s.d. of three experimental replicates and are representative of at least two independent experiments with n=2 donor samples.
Supplementary Figure 8 Antigen-specific expansion of CD14+ cell-depleted PBMCs with APC-ms
Antigen-specific enrichment (a) and expansion (b) of CLG- or GLC-specific CD8+ T cells from CD14+ cell-depleted PBMCs cultured with APC-ms presenting CLG or GLC, quantified via FACS. Data are from different donor samples and are representative of at least three independent experiments. APC-ms/CLG, n=2; APC-ms/GLC, n=4. Only day 14 data is available for the enrichment and expansion of GLC-specific CD8+ T cells.
Supplementary Figure 9 Extended characterization of APC-ms-restimulated 19BBz CAR T cells and their efficacy in a disseminated lymphoma xenograft model
(a) Representative FACS plots showing CD4 and CD8 expression on CD3+ 19BBz T cells at the start of culture (d0), and after 14 days of culture with either Dynabeads (dose D1) or APC-ms (formulation A4). (b) Degradation of APC-ms cultured with mouse T cells, measured via ICP-AES. Si content of APC-ms was below the limit of detection of ICP-AES by day 7. (c-d) Complete IVIS image sets for luciferized Raji-inoculated NSG mice that were mock-treated (RPMI-1640; labelled “M”), or treated with 19BBz T cells expanded for 7 days (c) or 14 days (d) with Dynabeads (labelled “D”) or APC-ms (labelled “A”). The dotted line denotes the average luminescence of mice that were not administered luciferin at the first measured timepoint (d7) and represents baseline. Bioluminescent images (top) and quantification of images (bottom). One animal in these images was excluded from analysis (c, left; denoted by “X”) because it was administered a different dose of 19BBz T cells. For 7 day-expanded 19BBz T cells: mock, n=6; Dynabeads, n=6; APC-ms, n=6. For 14 day-expanded 19BBz T cells: mock, n=6; Dynabeads, n=7; APC-ms, n=7.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–9 (PDF 1815 kb)
Supplementary Table 1
Enrichment of EBV-specific CD8+ 1 T cells following a single stimulation with APC-ms presenting either CLG or GLC. (PDF 124 kb)
Formation of APC-ms in culture.
Individual rods randomlysettled and stacked to form a 3D scaffold in culture. (WMV 12616 kb)
Infiltration of APC-ms by primary T cells.
The large interparticle spaces formed by 3D scaffold were infiltrated by mouse T cells. (WMV 10742 kb)
Rights and permissions
About this article
Cite this article
Cheung, A., Zhang, D., Koshy, S. et al. Scaffolds that mimic antigen-presenting cells enable ex vivo expansion of primary T cells. Nat Biotechnol 36, 160–169 (2018). https://doi.org/10.1038/nbt.4047
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.4047
This article is cited by
-
Lyophilized lymph nodes for improved delivery of chimeric antigen receptor T cells
Nature Materials (2024)
-
Biomaterials to enhance adoptive cell therapy
Nature Reviews Bioengineering (2024)
-
In vivo human T cell engineering with enveloped delivery vehicles
Nature Biotechnology (2024)
-
Harnessing 3D in vitro systems to model immune responses to solid tumours: a step towards improving and creating personalized immunotherapies
Nature Reviews Immunology (2024)
-
Multimodal probing of T-cell recognition with hexapod heterostructures
Nature Methods (2024)